Table of Contents
Definition / general | Essential features | Embryology | Histology | Menstrual cycle related changes | Pregnancy / lactation | Menopause | Neonatal breast | Male breast | Transsexual breast | Clinical features | Gross description | Gross images | Microscopic (histologic) images | Positive stains | Negative stains | Electron microscopy images | Videos | Additional references | Board review style question #1 | Board review style answer #1 | Board review style question #2 | Board review style answer #2Cite this page: Agarwal I, Blanco L. Histology. PathologyOutlines.com website. https://www.pathologyoutlines.com/topic/breastnormal.html. Accessed April 24th, 2024.
Definition / general
- Mammary glands located on the upper anterior thorax that produce and secrete milk in females
Essential features
- Adult female breast consists of branching ducts and acini; the acini group together to form lobules
- Ductal - lobular system: composed of ducts and acini lined by a dual inner (luminal) epithelial cell layer and outer (basal) myoepithelial cell layer, resting on a basement membrane, enveloped by stroma
- Exhibits physiologic histomorphologic changes during menstrual cycle, pregnancy / lactation, menopause, exogenous hormone therapy
Embryology
- Considered an epidermal gland, as it develops as diverticula of epidermis into dermis
- Mammary gland development begins at week 5
- Week 5:
- Ectodermal thickenings called mammary ridges develop along ventral embryo, extending from axilla to medial thigh
- Ridges eventually disappear, except at fourth intercostal space where mammary gland develops
- Week 15 - 20:
- Mesenchyme condenses around epithelial stalk under testosterone influence
- Solid epithelial columns develop within mesenchyme
- Week 20 - 32:
- Differentiation of mesenchyme into fat within collagenous stroma
- Stroma of reticular dermis forms suspensory ligaments of Cooper, attaching breast to skin and subdividing breast into lobes
- Areola formed by ectoderm
- Week 32 - 40:
- Epithelial cords canalize and branch, forming lobuloalveolar structures
- Depression in epidermis forms at the convergence of lactiferous ducts: mammary pit
- Near term birth: nipple formed by evagination of mammary pit
- Reference: Mills: Histology for Pathologists, 4th Edition, 2012
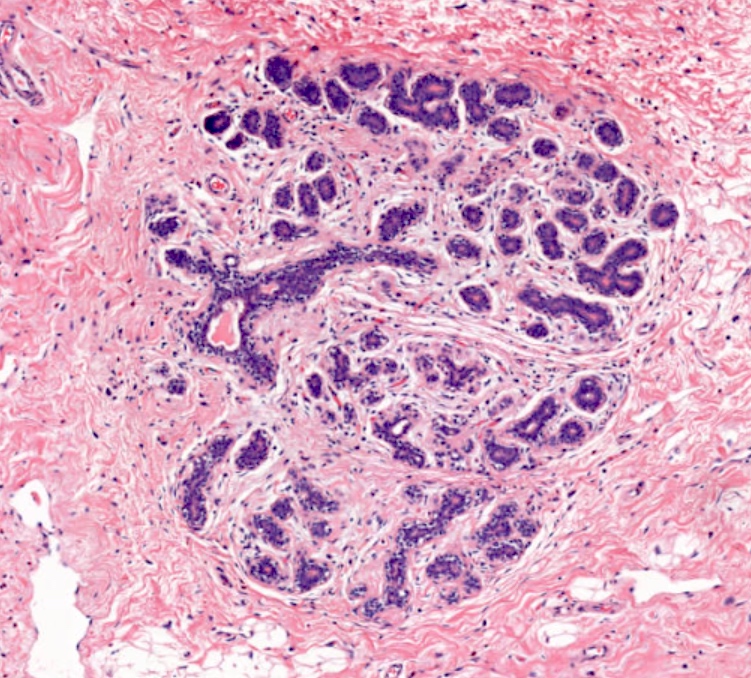
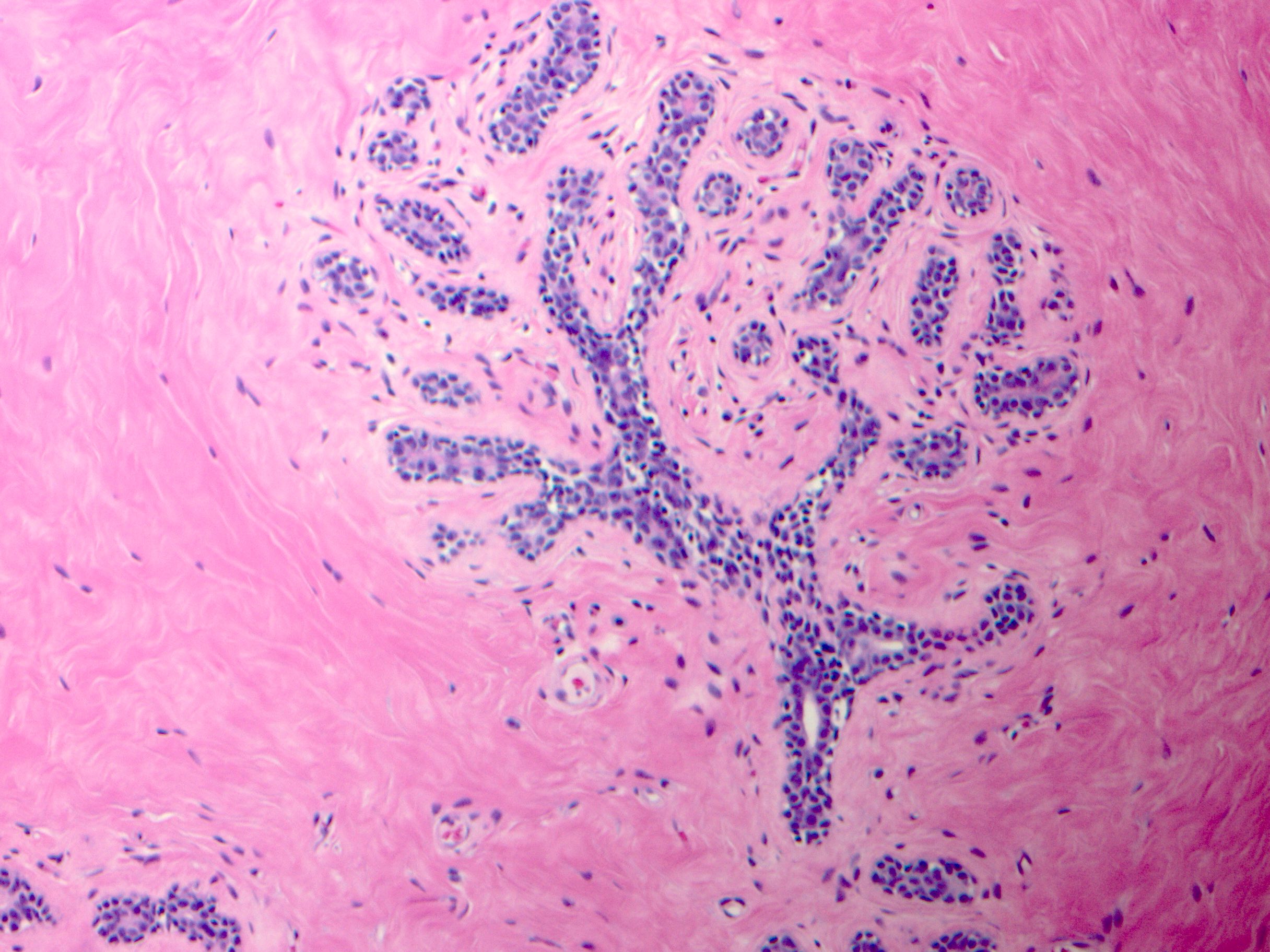
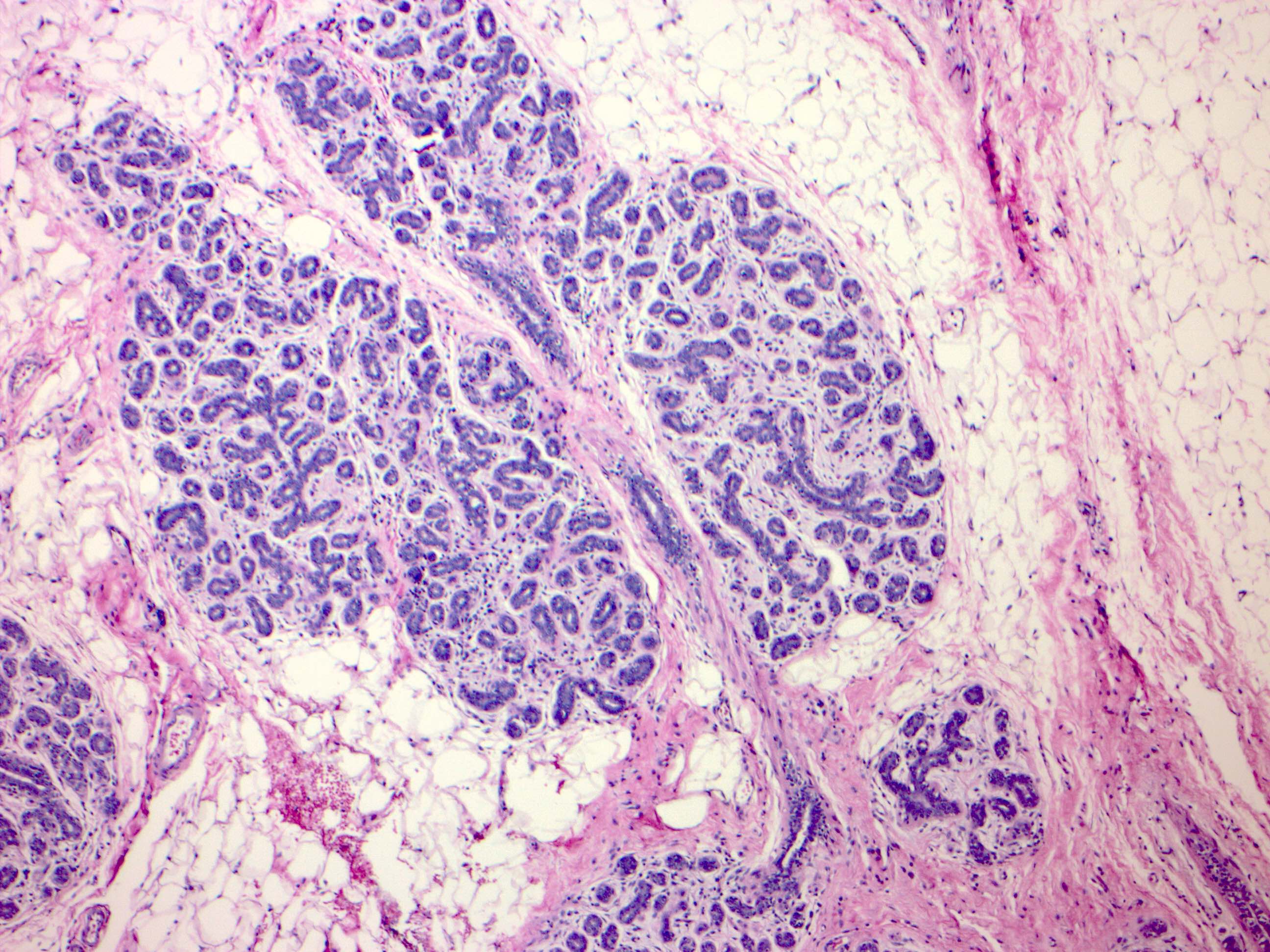
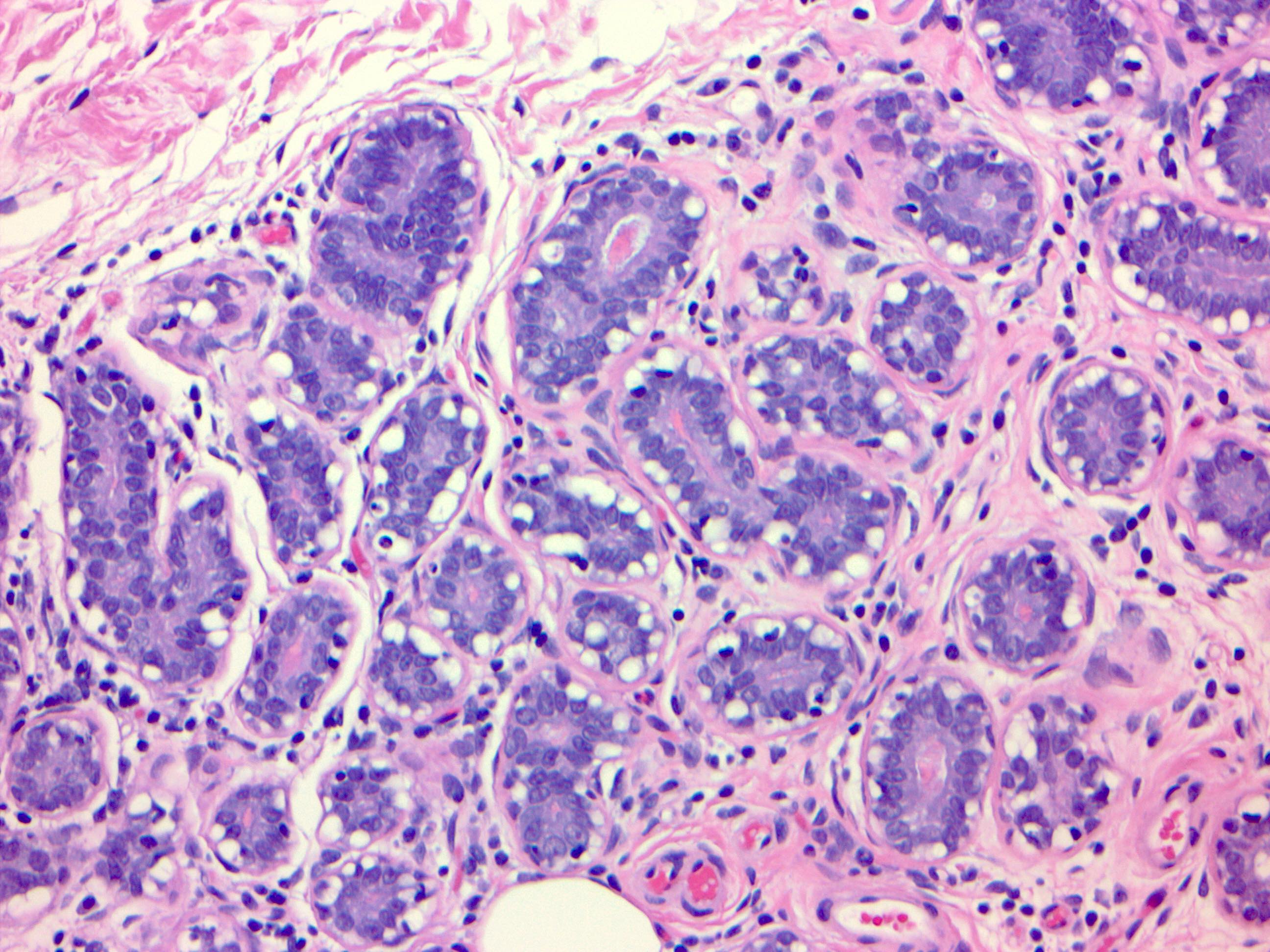
Histology
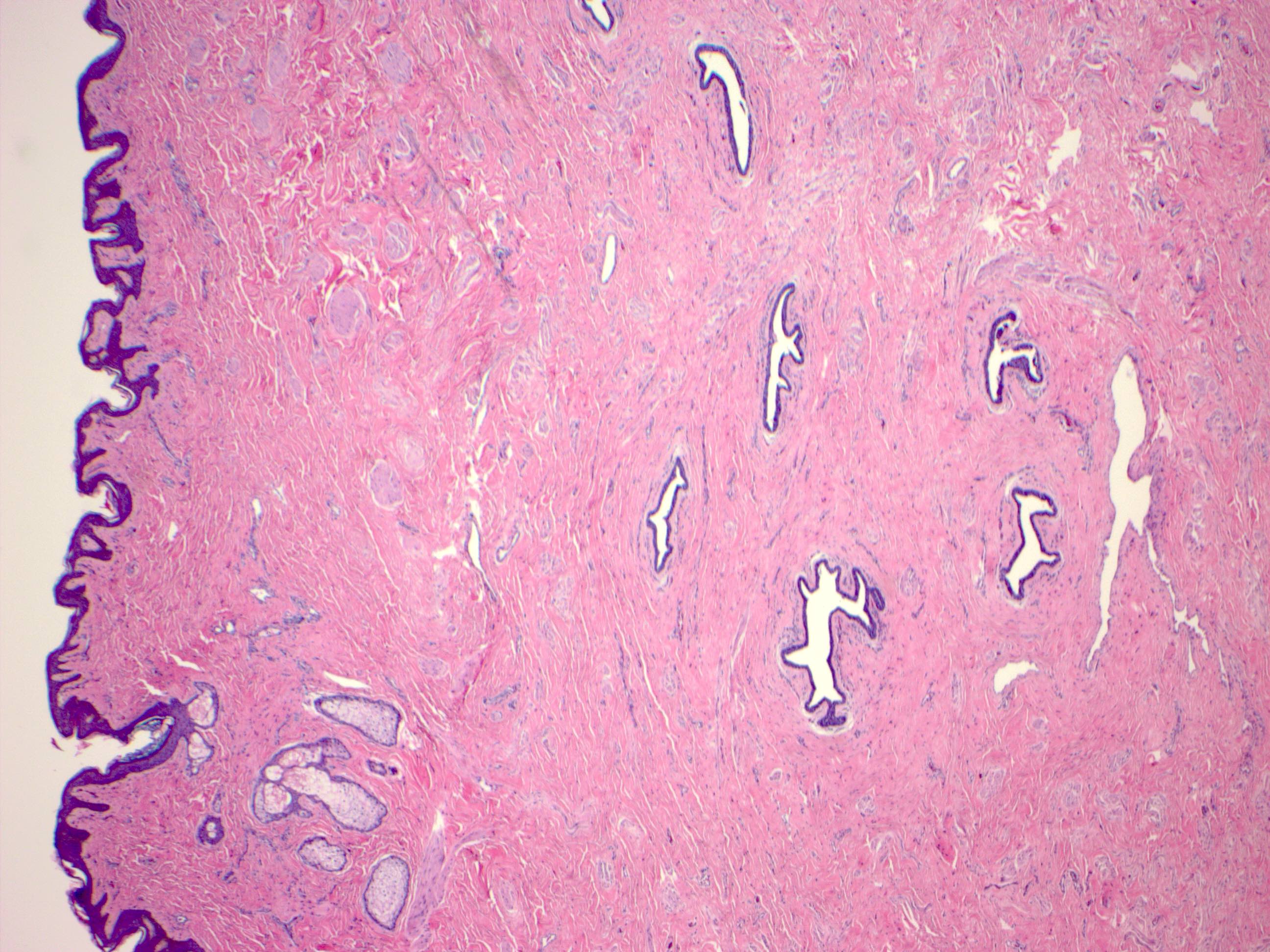
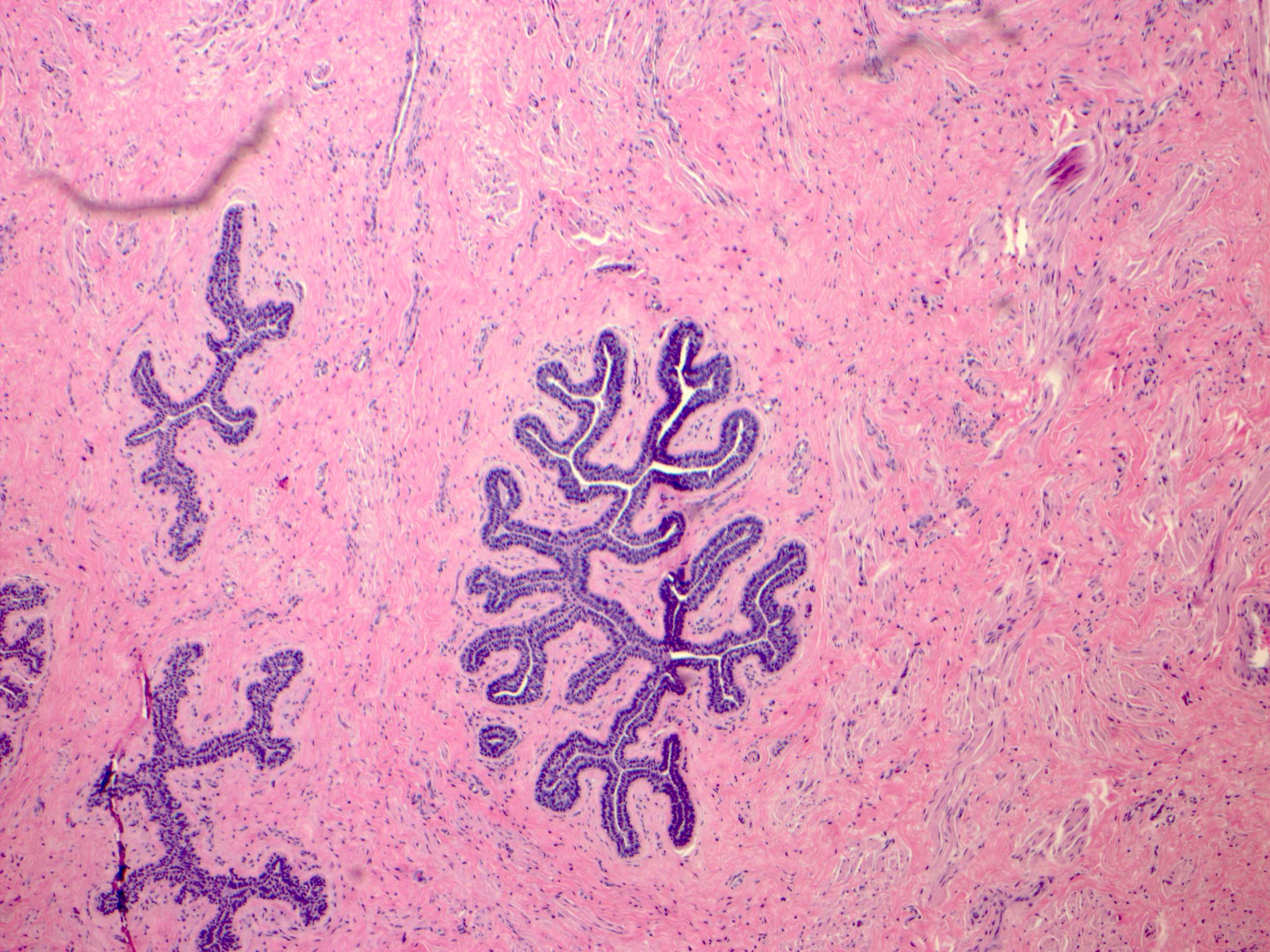
- Ductal - lobular system:
- Consists of large lactiferous ducts that open to the surface of the nipple via lactiferous sinuses, which branch into ducts and terminal ductules which terminate in acini
- Acini are grouped together in clusters to form lobules (terminal duct lobular unit)
- Ductal - lobular system is arranged in series of segments (5 - 10)
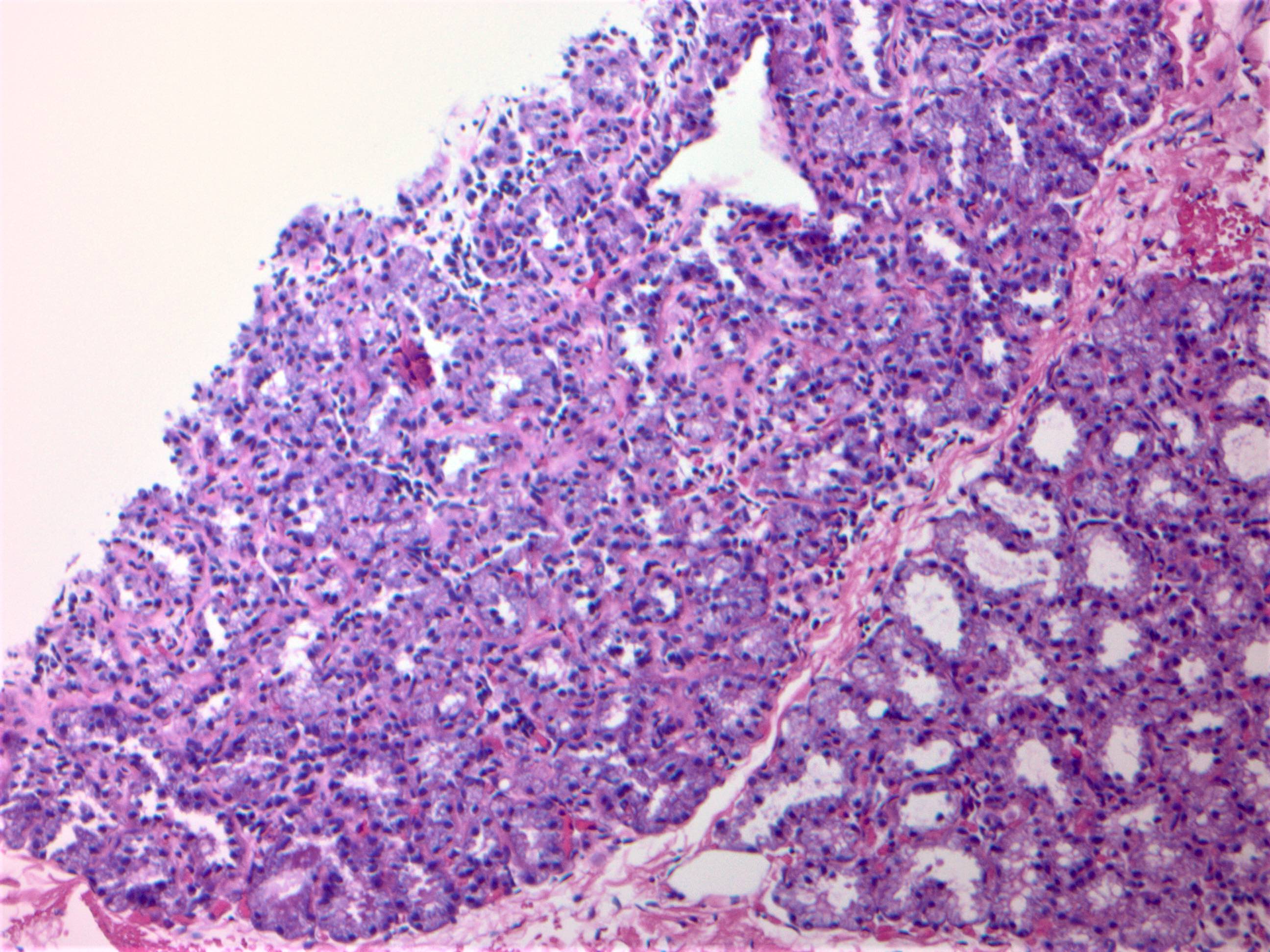
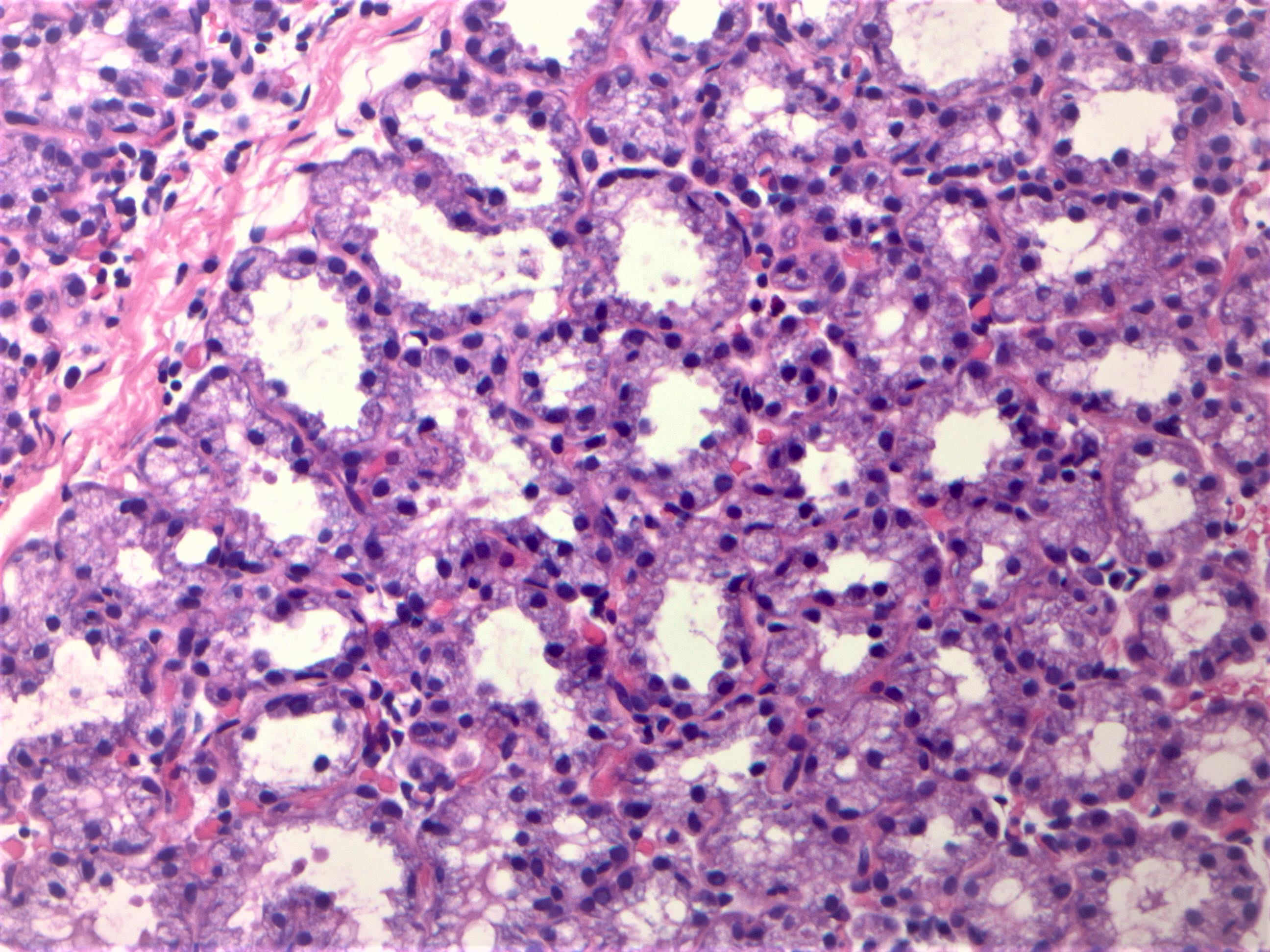
- Cellular lining of the ductal - lobular system is bilayered and consists of:
- Inner (luminal) epithelial cells: cuboidal to columnar epithelium with pale eosinophilic cytoplasm
- Outer (basal) myoepithelium: variably distinctive, varies in appearance from flattened cells with compressed nuclei to prominent epithelioid cells with abundant clear cytoplasm, can sometimes have myoid appearance
- Both epithelial and myoepithelial cells thought to arise from a bipotent progenitor cell (Oncotarget 2017;8:10675)
- Basement membrane surrounds mammary ducts, ductules and acini (Virchows Arch A Pathol Anat Histopathol 1992;421:323)
- Basement membrane contains type IV collagen and laminin, demarcates ductal - lobular system from the surrounding stroma
- Transgression of the myoepithelial layer and basement membrane by tumor cells defines invasion
- Stroma:
- Consists of varying amounts of fibrous tissue and adipose tissue and can be divided into:
- Interlobular stroma:
- Surrounds large ducts and terminal duct lobular units (TDLUs), denser, more collagenous than intralobular connective tissue
- Multinucleated giant cells of unknown significance are sometimes present (Virchows Arch 2001;439:768)
- Intralobular stroma: surrounds acini within TDLUs, loose, contains fibroblasts, scattered inflammatory cells and histiocytes
- Interlobular stroma:
- Consists of varying amounts of fibrous tissue and adipose tissue and can be divided into:
- Nipple - areolar complex: skin variably pigmented, contains numerous sebaceous glands
- Independent of hair follicles, except at periphery of areola; contains dense fibrous stroma with bundles of smooth muscle
- Montgomery tubercles: sebaceous glands sharing ostium with a lactiferous duct, more prominent during pregnancy and lactation
- Epidermis may contain occasional clear cells, either clear keratinocytes or Toker cells (epidermally located mammary duct epithelium)
- Toker cells found in 10% of normal nipple (Histol Histopathol 2009;24:367)
- Stratified squamous epithelium extends into duct lumens for 1 - 2 mm (Clin Radiol 2012;67:e42)
Pregnancy / lactation
- Dramatic increase in the number of lobules, as well as acinar units
- Increased epithelial cell proliferation and lobuloalveolar differentiation
- Decrease in intralobular and interlobular stroma
- Second and third trimester:
- Increase in size of epithelial cells and cytoplasmic vacuolization, myoepithelial cells difficult to discern
- Lactation:
- Abundant secretory material and prominent cytoplasmic vacuoles, epithelial cells may have bulbous or hobnail appearance
- Myoepithelial cells remain inconspicuous
- Rarely areas of infarction (Exp Ther Med 2015;10:1888)
Menopause
- Reduction of estrogen and progesterone causes involution and atrophy of TDLUs
- Reduction in size and complexity of acini, loss of specialized intralobular stroma
- Hyalinized stroma with relative increase in fat
- Reference: Mills: Histology for Pathologists, 4th Edition, 2012
Neonatal breast
- Identical in males and females at birth
- Breast enlargement due to neonatal prolactin production, caused by falling maternal estrogen
- Associated with production of colostrum
- Extramedullary hematopoiesis may be seen (J Pediatr Hematol Oncol 2012;34:229)
Male breast
- Composed of ductal structures only within collagenized stroma, with no / rare acini
Transsexual breast
- Female to male conversion results from prolonged androgen administration, starting prior to mastectomy
- Reduction in glandular tissue and prominence of stroma (J Histochem Cytochem 2006;54:905)
- Calcifications more frequent (J Pathol 1993;170:37)
- Male to female conversion involves prolonged estrogen therapy and chemical castration with progestational agents
- Changes include well developed lobular structures and pregnancy related changes
Clinical features
- Gross anatomy:
- Lies on the anterior chest wall over the pectoralis major muscle, extends from second to sixth rib vertically and from sternal edge to midaxillary line horizontally
- Also projects into the axilla as the tail of Spence
- Microscopic foci of glandular tissue may extend through the pectoral fascia
- Clinical relevance: total mastectomy does not remove all glandular breast tissue
- Arterial supply, lymphatic flow:
- Principal arterial supply by the internal mammary and lateral thoracic arteries
- Lymphatic drainage: > 90% to axillary lymph nodes, remaining to internal mammary and posterior intercostal nodes
- Relevance to staging, as metastasis to internal mammary node is higher nodal stage
- Breast tissue is typically less nodular, late follicular phase is the optimal time for mammography (Radiol Phys Technol 2018;11:20)
- Loss of myoepithelium is usually considered evidence of invasion, with the exception of microglandular adenosis, encapsulated papillary carcinoma and solid papillary carcinoma
- Transsexual breast may develop any benign or malignant process seen in nontranssexual breast (Hoda: Rosen's Breast Pathology, 4th Edition, 2014)
Gross description
- Mastectomy attempts to remove all breast tissue
- Different types of mastectomies: simple, modified radical, prophylactic, skin sparing, nipple sparing and radical
- Axillary dissections performed for the staging of invasive carcinoma
- Number of lymph nodes with metastases is the most important prognostic factor for breast carcinoma
- Reference: Lester: Manual of Surgical Pathology, 3rd Edition, 2010
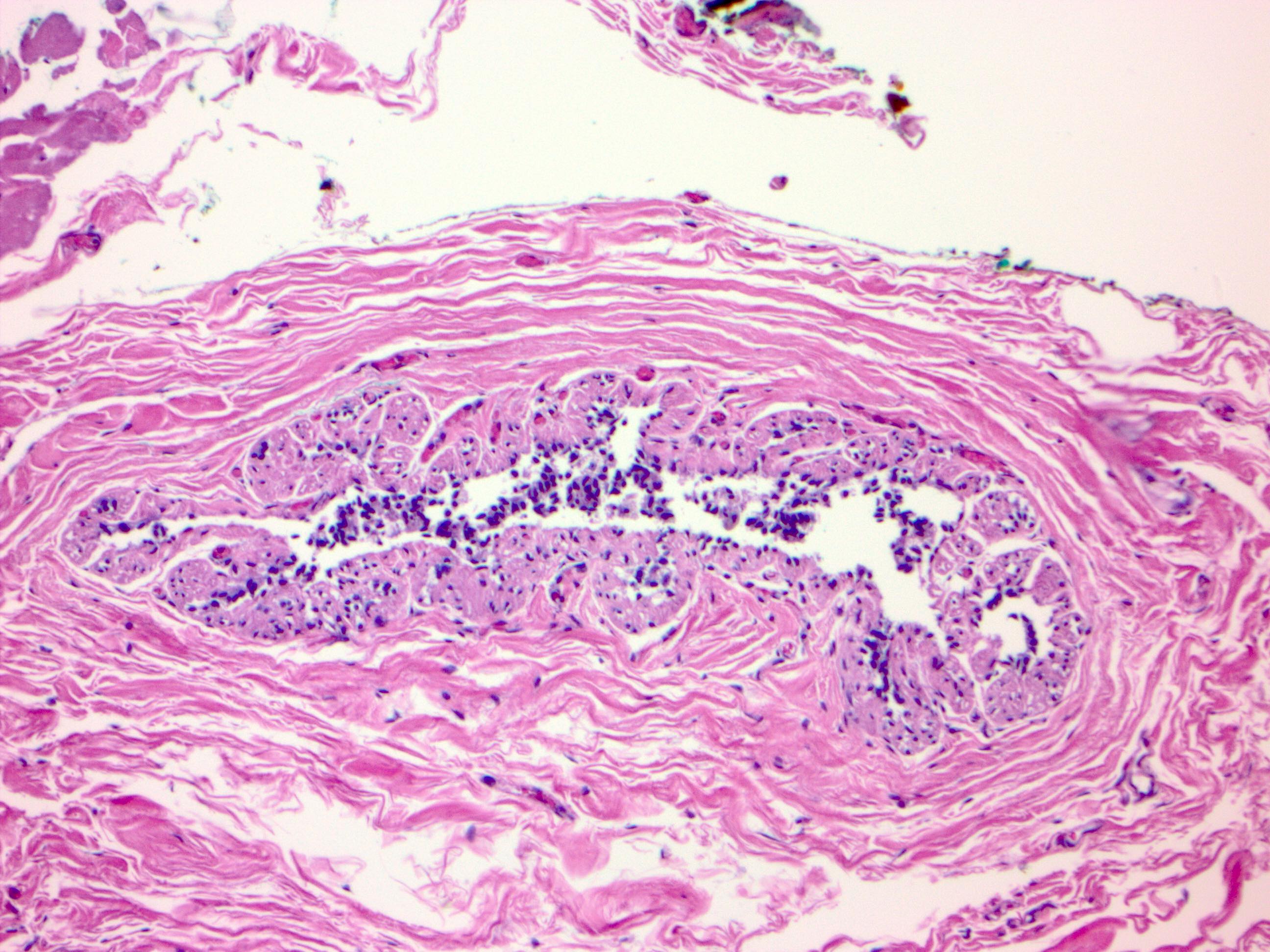
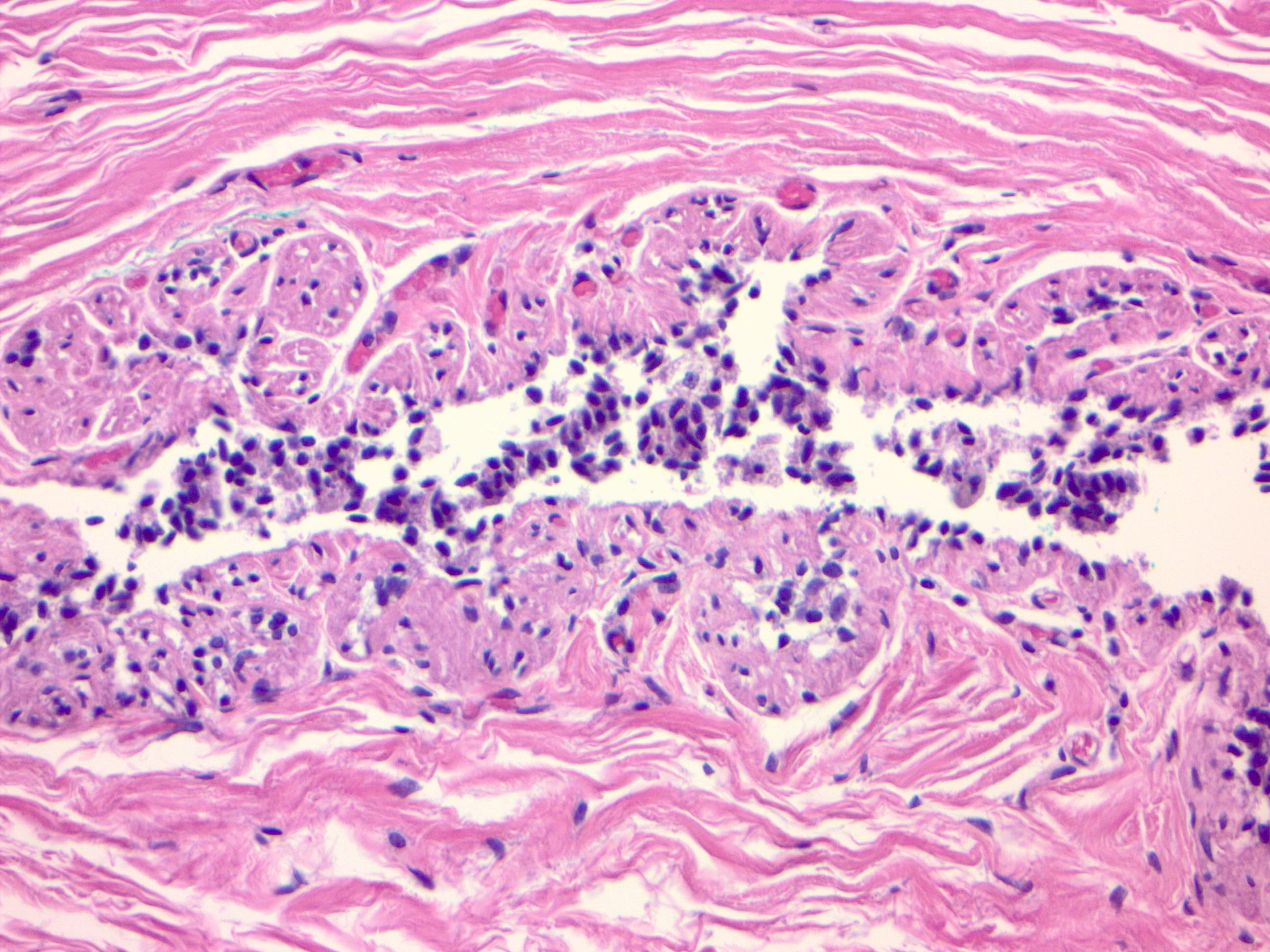
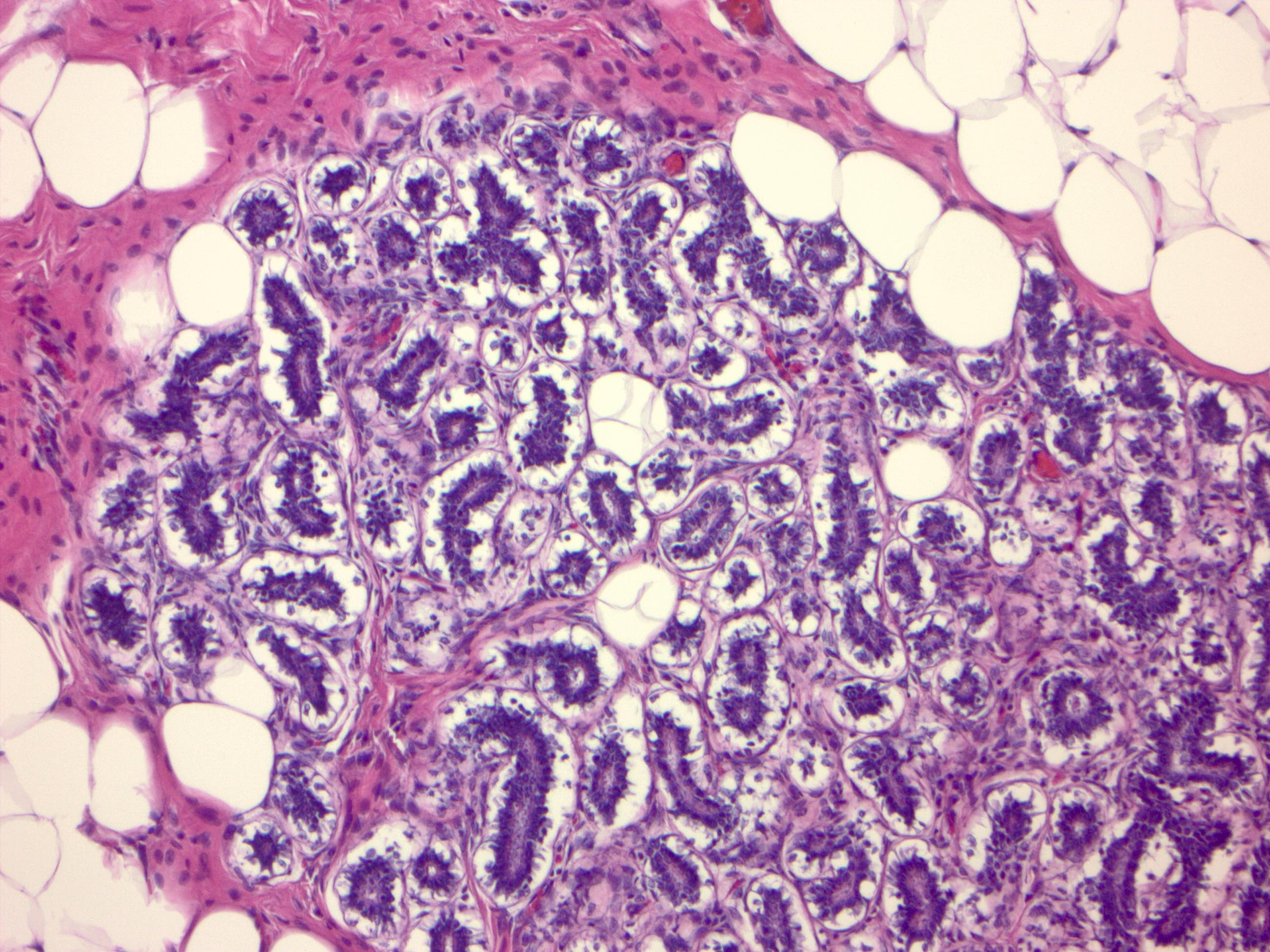
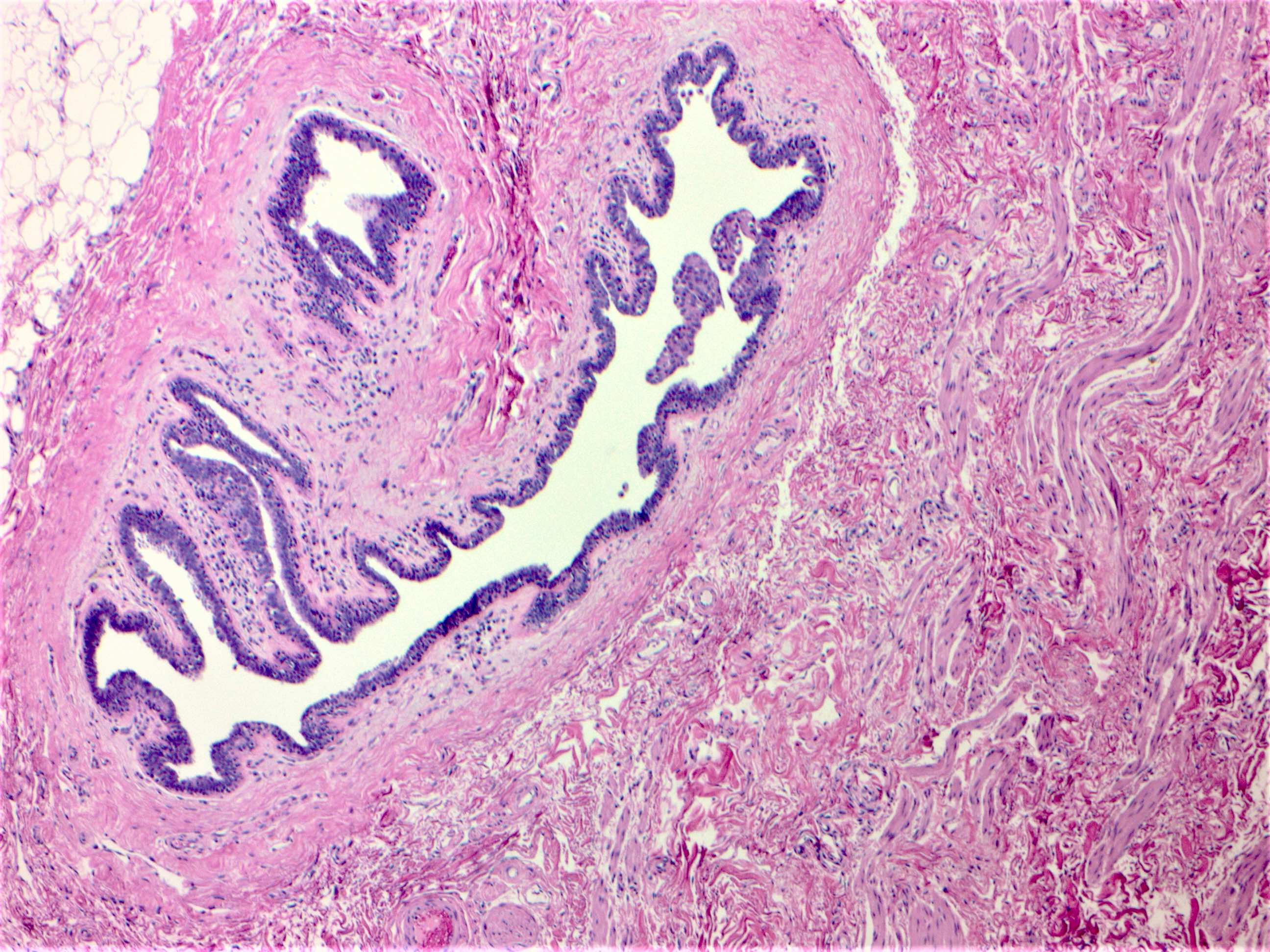
Microscopic (histologic) images
Positive stains
- Basement membrane: laminin, type IV collagen and reticulin
- Epithelium / luminal cells:
- Usually express luminal cytokeratins: LMWCK, CK7, CK8, CK18, CK19 (J Pathol 2004;203:661)
- Also express mammaglobin, gross cystic disease fluid protein (GCDFP)
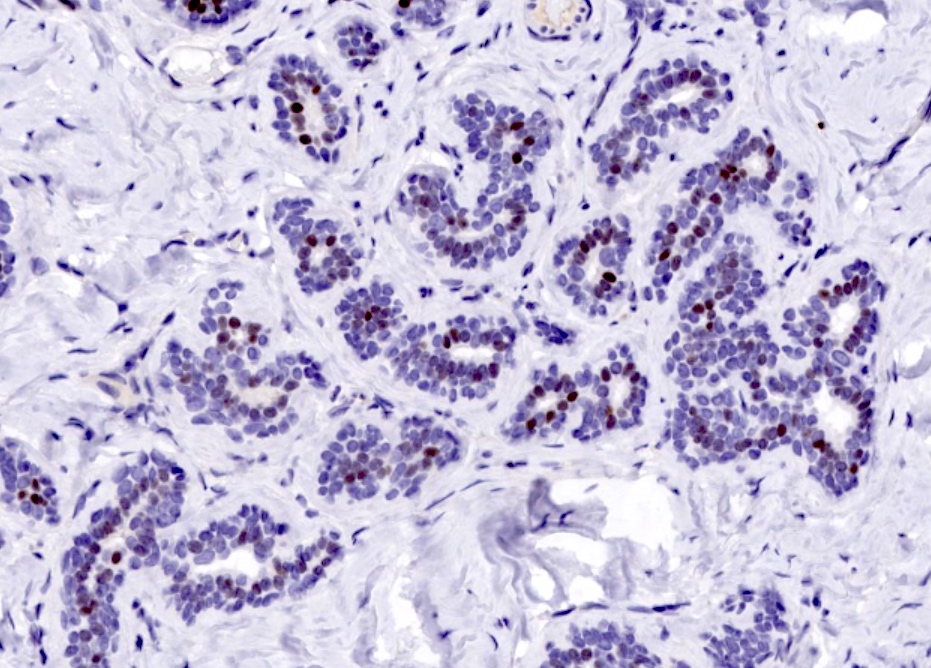
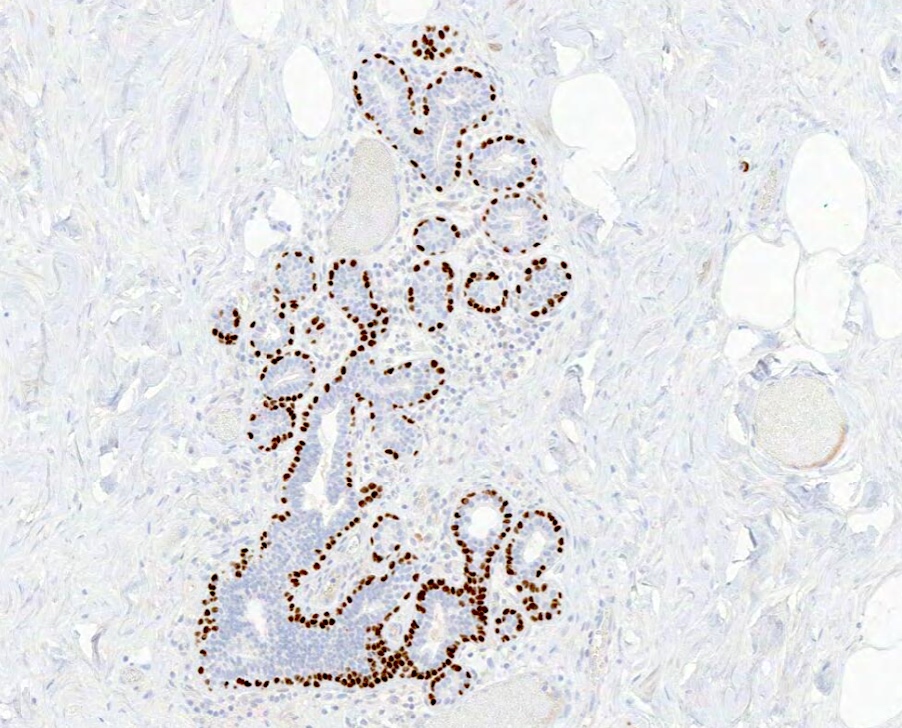
- Most express ER alpha or PR in a heterogeneous pattern
- E-cadherin, p120, beta catenin in membranous pattern
- Myoepithelial cells:
- Basal cytokeratins: HMWCK, CK5/6, CK14 (Front Biosci (Landmark Ed) 2010;15:226)
- P-cadherin, calponin, smooth muscle myosin heavy chain (SMMHC), smooth muscle actin, CD10, S100 (Arch Pathol Lab Med 2008;132:349)
- At least 2 markers, 1 with a nuclear staining pattern (e.g. p63) and 1 with cytoplasmic staining (e.g. SMMHC) recommended
- Toker cells: CK7, EMA and usually positive for ER and PR
Negative stains
- HER2 / neu overexpression or amplification not seen in normal breast, restricted to carcinomas (J Clin Oncol 2000;18:267)
Videos
Shotgun histology inactive breast
Additional references
Board review style question #1
Board review style answer #1
B. Lactational changes. During lactation, epithelial cells are enlarged and have a bulbous or hobnail appearance with attenuated myoepithelial cells. These florid changes seen can be alarming and can be mistaken for atypia or malignancy.
Comment Here
Reference: Breast - Histology
Comment Here
Reference: Breast - Histology
Board review style question #2
Myoepithelial cells are always retained in which of the following?
- Atypical ductal hyperplasia
- Encapsulated papillary carcinoma
- Invasive tubular carcinoma
- Microglandular adenosis
Board review style answer #2
A. Atypical ductal hyperplasia. Myoepithelium is retained in atypical ductal hyperplasia and most benign and in situ lesions, with the exception of microglandular adenosis and a few in situ lesions, e.g. encapsulated papillary carcinoma.
Comment Here
Reference: Breast - Histology
Comment Here
Reference: Breast - Histology