Table of Contents
Definition / general | Essential features | Terminology | ICD coding | Epidemiology | Sites | Pathophysiology | Clinical features | Diagnosis | Laboratory | Prognostic factors | Case reports | Treatment | Microscopic (histologic) description | Microscopic (histologic) images | Virtual slides | Cytology description | Peripheral smear description | Positive stains | Negative stains | Flow cytometry description | Flow cytometry images | Molecular / cytogenetics description | Molecular / cytogenetics images | Videos | Sample pathology report | Differential diagnosis | Additional references | Board review style question #1 | Board review style answer #1 | Board review style question #2 | Board review style answer #2Cite this page: Zinzuwadia S, Mittal A, Raouf MF, Gupta R, Kini AR. AML with inv(16)(p13.1;q22) or t(16;16)(p13.1;q22); CBFB::MYH11. PathologyOutlines.com website. https://www.pathologyoutlines.com/topic/leukemiainv16.html. Accessed April 24th, 2024.
Definition / general
- Acute myeloid leukemia (AML) with inv(16)(p13.1;q22) or t(16;16)(p13.1;q22); CBFB-MYH11 is a subtype of AML with recurrent genetic abnormalities characterized by myelomonocytic differentiation and presence of abnormal eosinophils
Essential features
- Inv(16)(p13.1;q22) or t(16;16)(p13.1;q22) results in the formation of an abnormal core binding factor beta subunit / myosin heavy chain 11 (CBFB-MYH11) fusion gene
- Most cases show myelomonocytic differentiation with presence of abnormal eosinophils
- Diagnosis of AML with inv(16)(p13.1;q22) or t(16;16)(p13.1;q22) can be made even when the blast percentage is less than 20%
- Prognosis is good compared with other AMLs
Terminology
- Acute myelomonocytic leukemia with abnormal eosinophils
- French American British (FAB) M4Eo
- Acute myeloid leukemia with CBFB-MYH11
ICD coding
Epidemiology
- 7% of AMLs in the pediatric age group (J Clin Oncol 2010;28:2674)
- < 5% of AMLs in older adults (Blood 2006;108:3280)
Sites
- Most cases present with involvement of the blood and bone marrow
- Occasional cases present as myeloid sarcoma (Am J Clin Pathol 2020;153:333)
Pathophysiology
- Both inv(16)(p13.1;q22) and t(16;16)(p13.1;q22) result in a core binding factor beta subunit / myosin heavy chain 11 (CBFB-MYH11) fusion gene encoding an abnormal CBFB-MYH11 fusion protein (Semin Hematol 2015;52:215)
- CBFB-MYH11 fusion protein causes aberrant self renewal and suppresses differentiation
- Additional mutations promote leukemogenesis
Clinical features
- Fatigue resulting from anemia
- Easy bruising from thrombocytopenia
- Mass effects of myeloid sarcoma, such as intestinal obstruction
- Higher white blood cell count at diagnosis of AML with inv(16)(p.13.1q22) and t(16;16)(p.13.1;q22) compared with other AMLs (J Clin Oncol 2005;23:5705)
Diagnosis
- Diagnosis is based on a combination of morphology, flow cytometry, conventional cytogenetics, real time PCR and FISH analysis
Laboratory
- Anemia and thrombocytopenia
Prognostic factors
- Overall prognosis is good compared to other AMLs (Blood 2003;102:462)
- KIT and FLT3 mutations are associated with worse prognosis
- Trisomy 22 is associated with a more favorable outcome
- High levels of minimal residual disease after induction are associated with worse outcomes (Am Soc Clin Oncol Educ Book 2018;38:555)
Case reports
- 30 year old man presented to emergency department with 2 weeks history of fever, abdominal pain and fatigue (Clin Med Insights Blood Disord 2017;10:1179545X17700858)
- 30 year old woman presented with a 2 week history of fever, cough with dyspnea and chest pain, generalized weakness and erythematous papules over the dorsum of her right hand and bleeding per vaginum (Indian J Pathol Microbiol 2016;59:104)
- 32 year old woman was first seen for gingivitis, hypertrophic gingiva, metrorrhagia and purpura (Hematol Oncol 2017;35:385)
- 47 year old man presented with intermittent low grade fever and progressive fatigue (Mol Cytogenet 2020;13:4)
Treatment
- Induction chemotherapy with 3 days of anthracycline and 7 days of cytarabine (7+3) (Am Soc Clin Oncol Educ Book 2018;38:555)
- Cytarabine or HDAC inhibitors are used for consolidation therapy
- Gemtuzumab ozogamicin (toxin conjugated monoclonal antibody to CD33) may improve outcomes when combined with standard chemotherapy
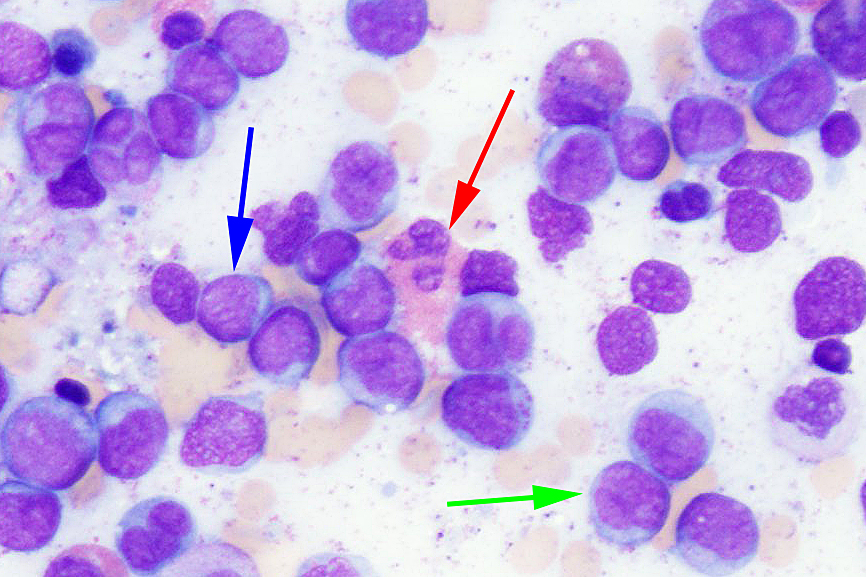
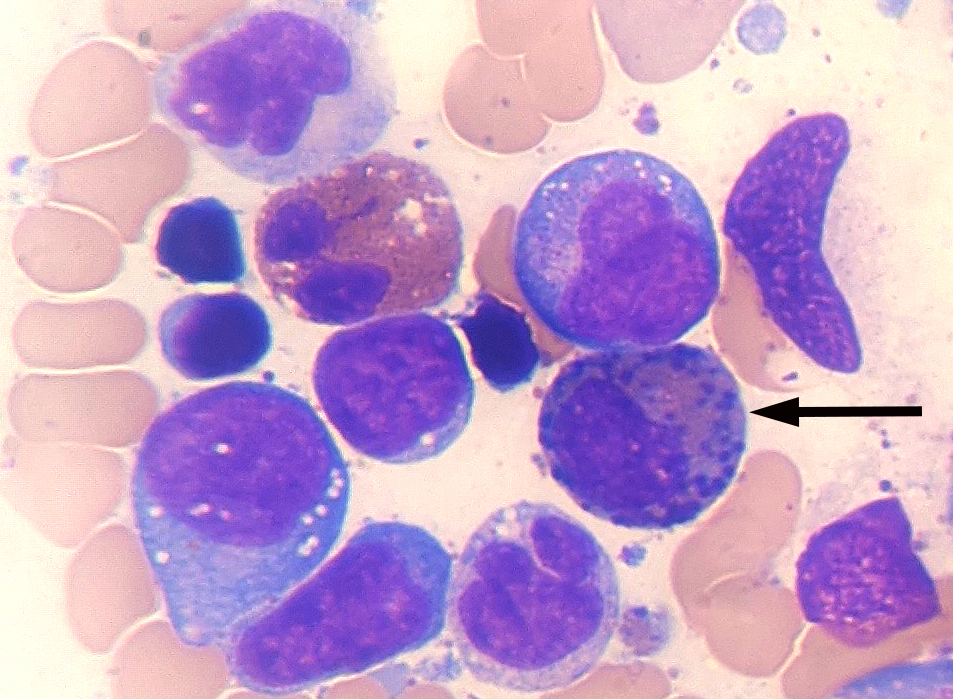
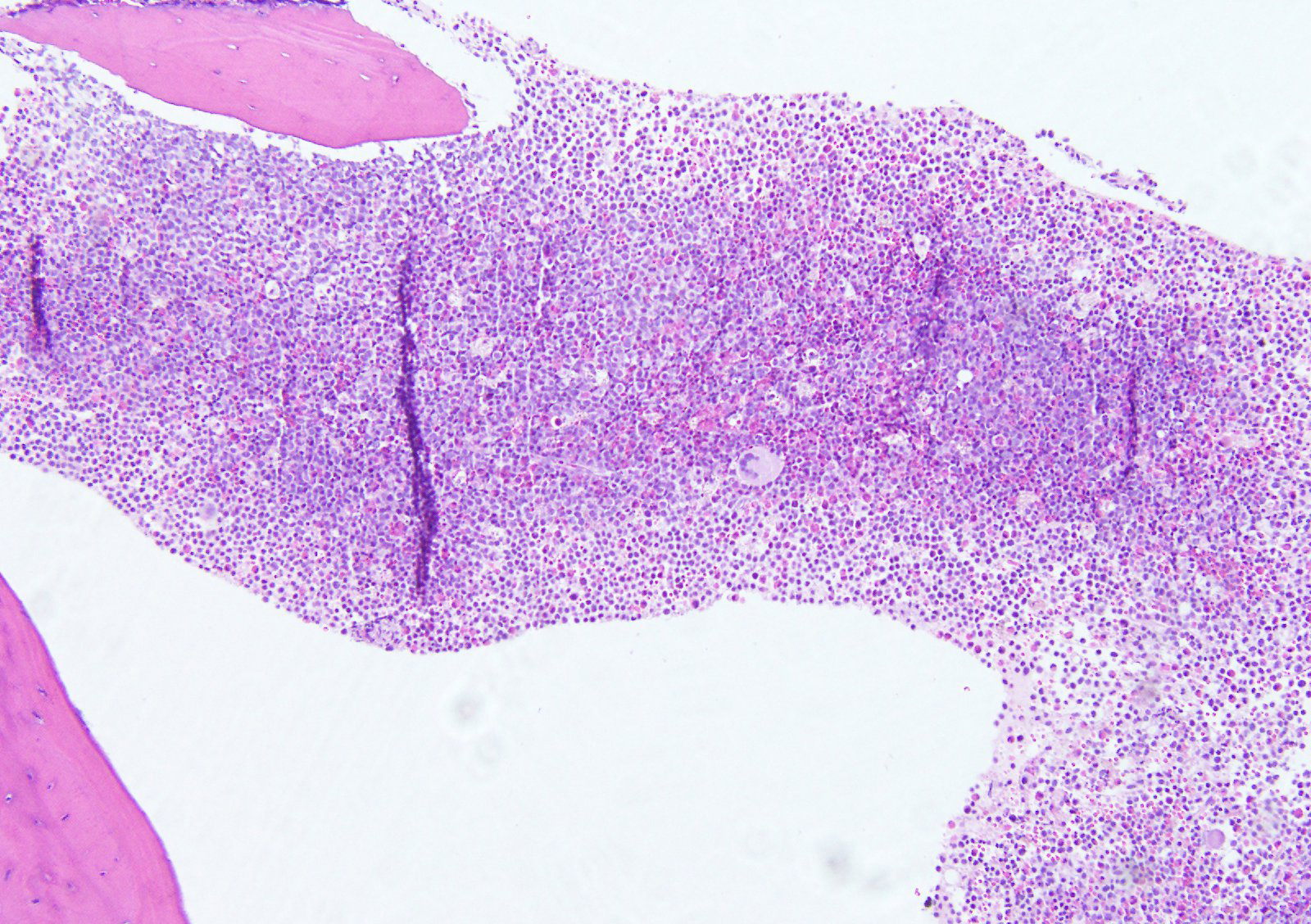
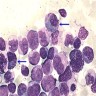
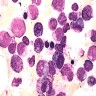
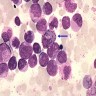
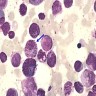
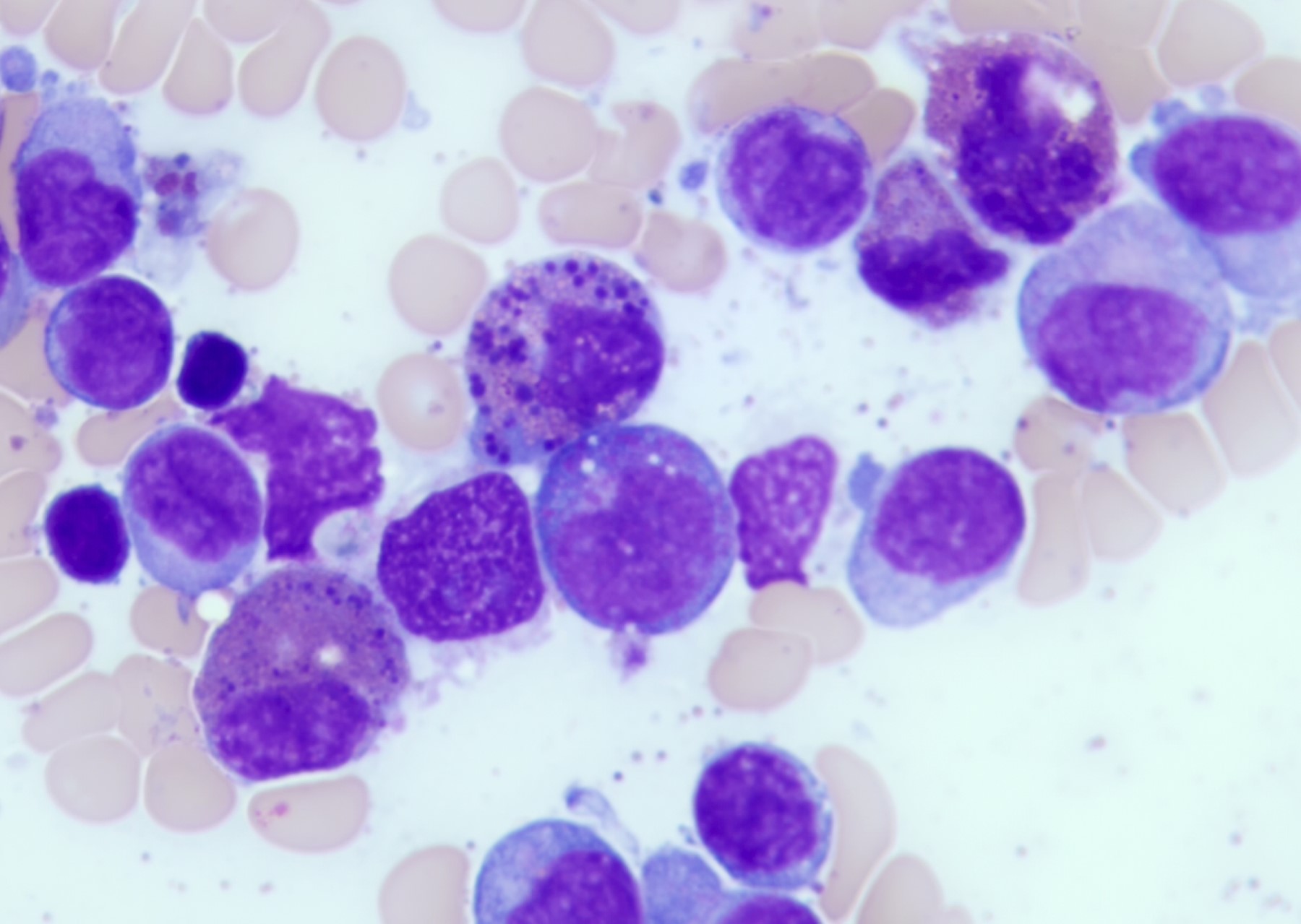
Microscopic (histologic) description
- Hypercellular bone marrow core biopsy and aspirate clot sections with presence of immature cells; increased eosinophilic cells can be seen
Microscopic (histologic) images
Contributed by Ameet R. Kini, M.D., Ph.D. and Maryam F. Raouf, M.D.
Images hosted on other servers:
Cytology description
- Increased blasts showing nuclei with fine chromatin and high nuclear to cytoplasmic ratio
- Some blasts display Auer rods
- Blasts are usually > 20% but the diagnosis is made even when blasts are < 20%, provided inv(16)(p13.1;q22) or t(16;16)(p13.1;q22) is seen
- Increased eosinophils at all stages of maturation, with the earlier stages displaying dual granulation (Haematologica 2002;87:886)
- Increased immature monocytic cells (promonocytes)
Peripheral smear description
- Presence of blasts and immature monocytic cells
- Eosinophils are not usually increased (Blood 1986;68:1242)
Positive stains
- Abnormal eosinophils are faintly positive for naphthol AS-D chloroacetate esterase
- Myeloblasts are positive for myeloperoxidase
- Monocytic cells are positive for alpha naphthyl butyrate esterase (nonspecific esterase)
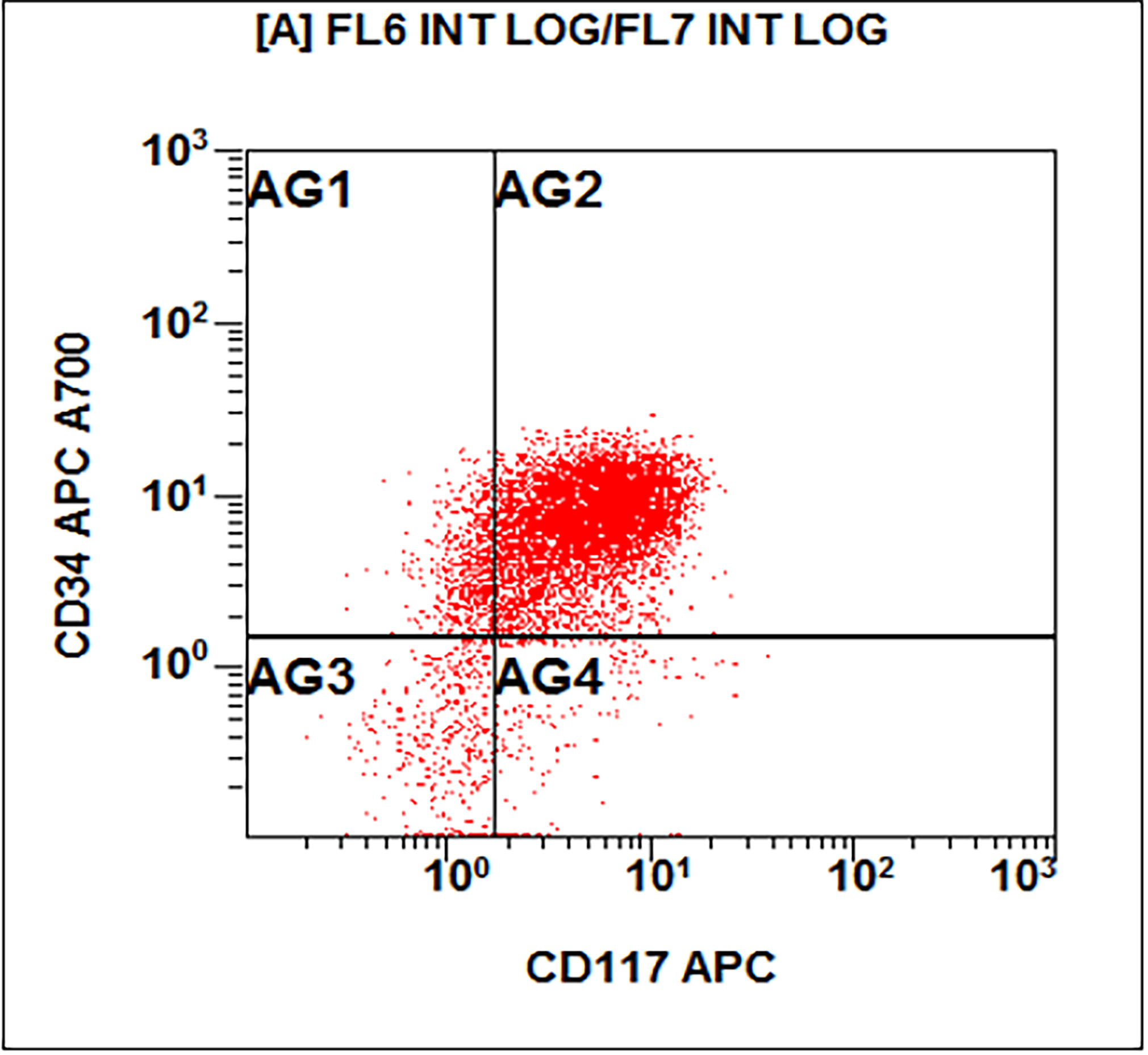
- Immunohistochemical stains show positivity for CD34 and CD117 in the blasts
Negative stains
- Negative for nonhematopoietic markers and most lymphoid markers
Flow cytometry description
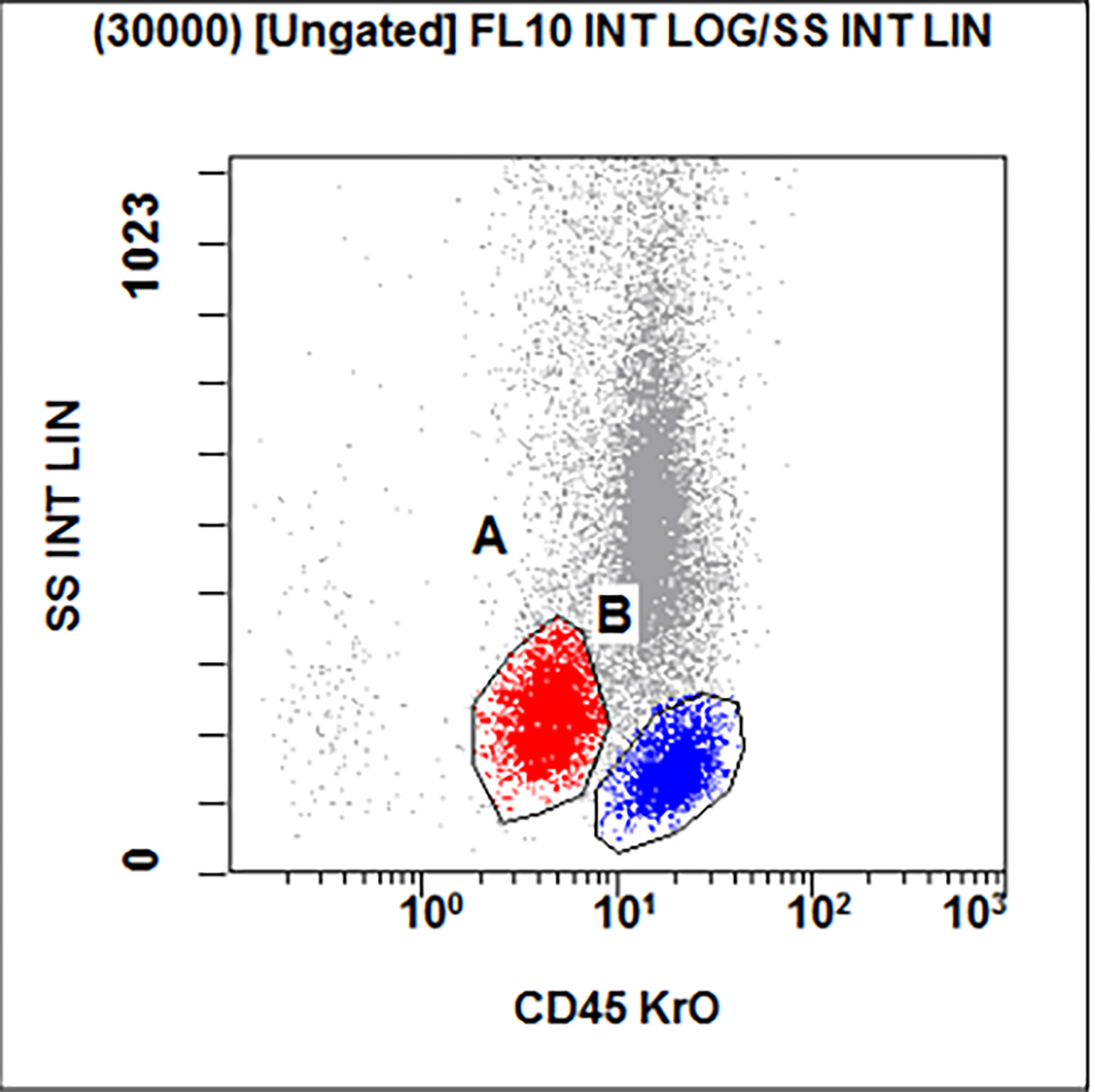
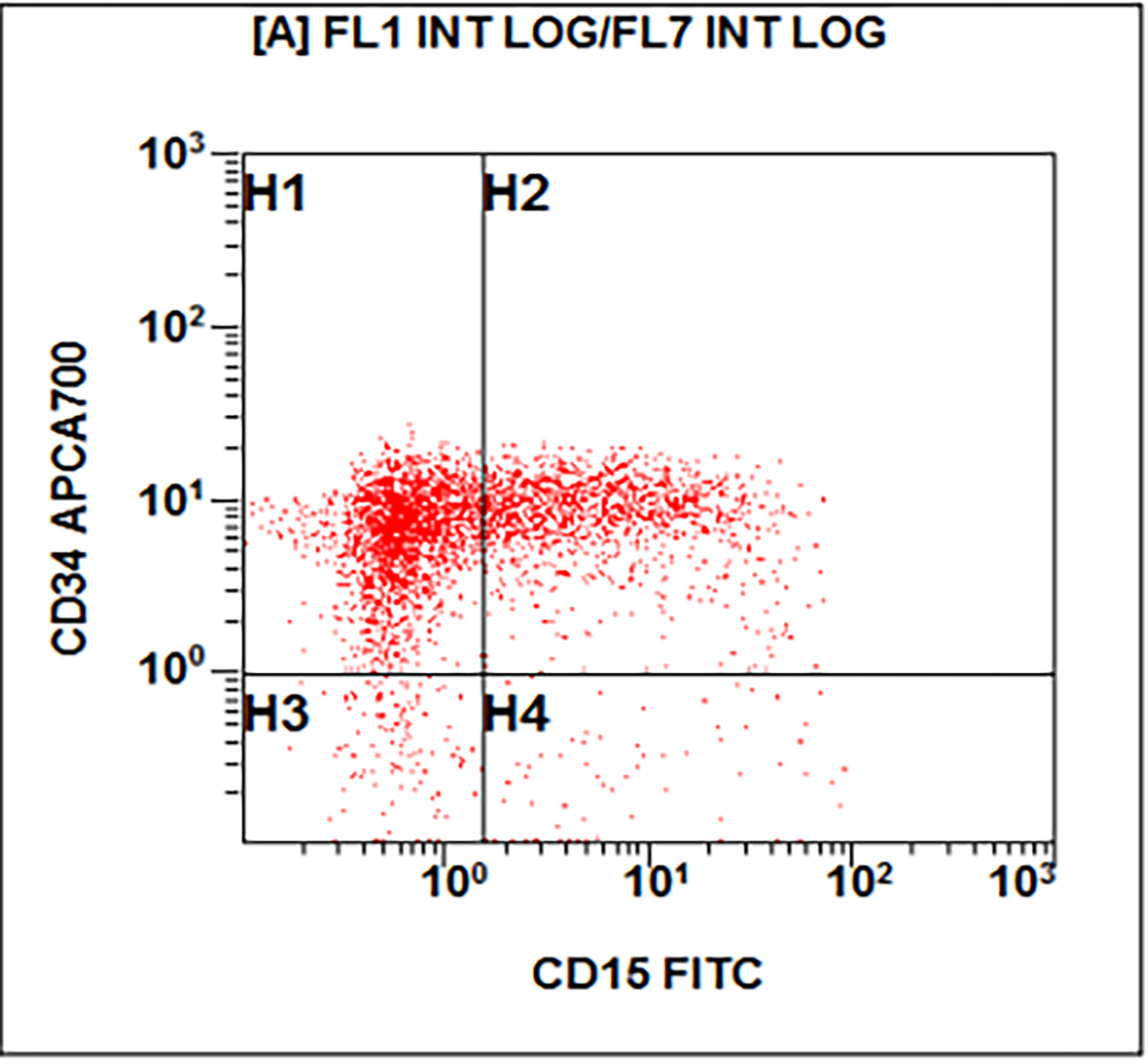
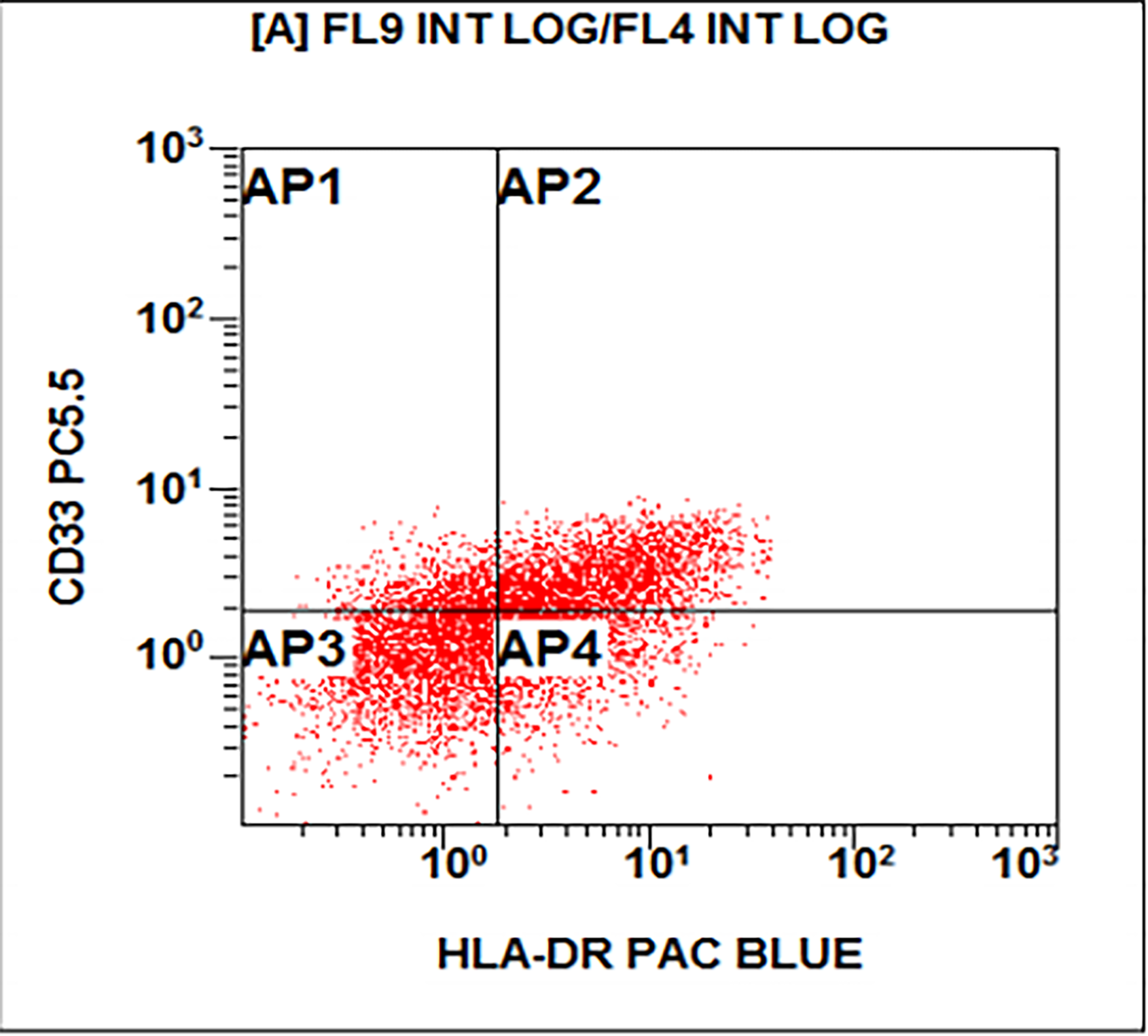
Flow cytometry images
Molecular / cytogenetics description
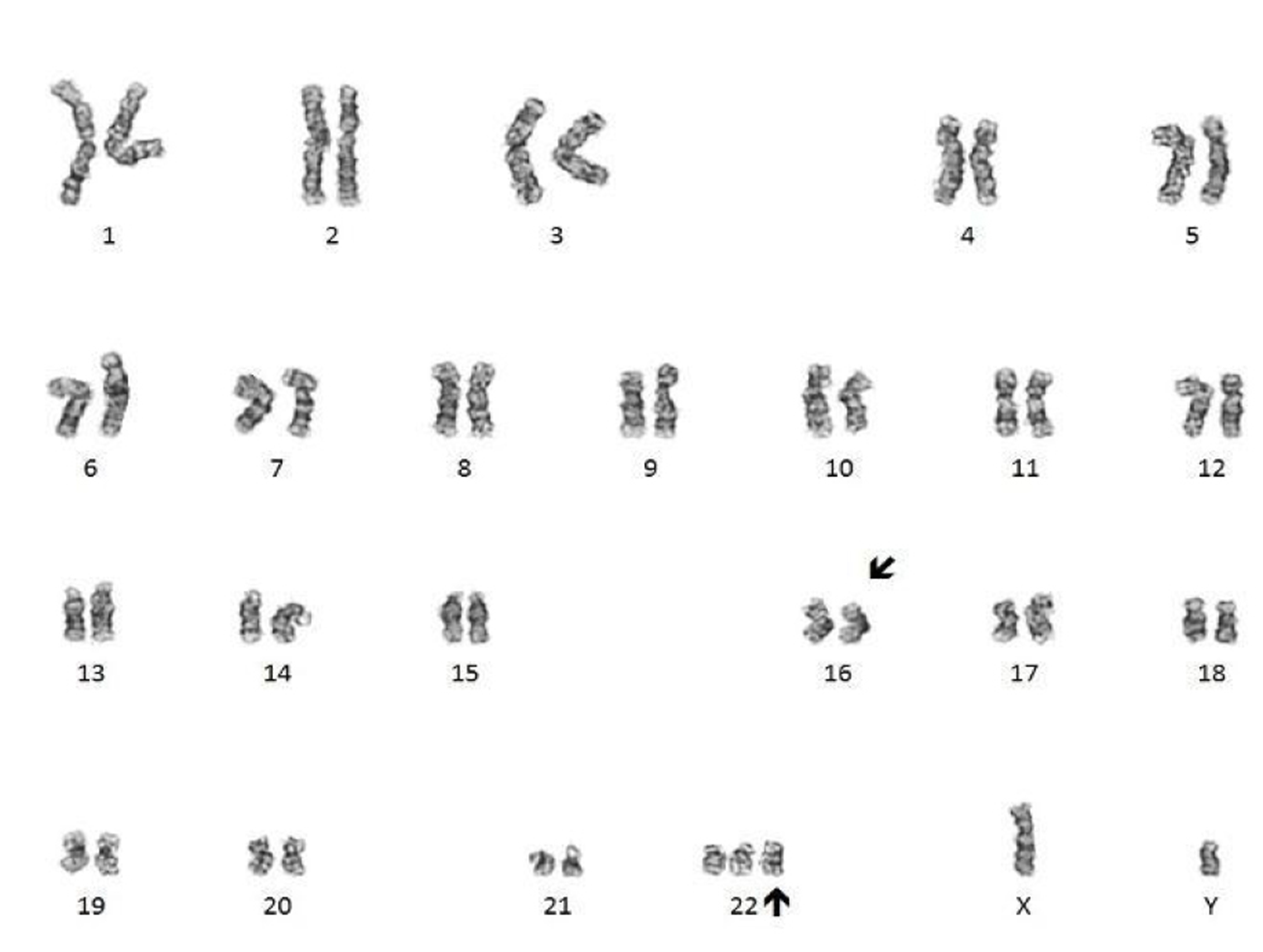
- Conventional cytogenetics shows presence of inv(16)(p13.1;q22) or t(16;16)(p13.1;q22)
- FISH analysis may be required to detect cryptic cases and shows the CBFB-MYH11 fusion gene (Genes Chromosomes Cancer 1998;22:87)
- In rare cases, real time PCR can detect the CBFB-MYH11 transcript when cytogenetics and FISH analyses are negative
- Real time PCR is used to assess minimal / measurable residual disease (MRD)
Molecular / cytogenetics images
Videos
AML diagnosis
Targeted therapy for inv(16)
AML diagnosis and treatment
MRD in AML
Sample pathology report
- Bone marrow, right posterior iliac crest, core biopsy, clot section, aspirate smears and touch imprint hypercellular bone marrow (95%) with involvement by acute myeloid leukemia with inv(16)(p13.1;q22) (see comment)
- Comment: Bone marrow core biopsy is hypercellular for age (95%) Immature cells are increased. Erythroid precursors are decreased. Mature myeloid cells are decreased. Megakaryocytes are adequate and appear normal in morphology.
- Aspirate clot section is adequate and shows features similar to the core biopsy.
- Bone marrow aspirate smear is adequate. Blasts are increased (30%) and show a high nuclear to cytoplasmic ratio with fine chromatin. Immature monocytic cells (promonocytes) are also increased. Scattered eosinophils and eosinophil precursors are seen throughout the aspirate smear. Some of the eosinophilic cells show dual granulation. Megakaryocytes are adequate in number and show normal morphology. Cytochemical stains show that many of the blasts are positive for myeloperoxidase. Alpha naphthyl butyrate esterase (nonspecific esterase) is positive in the immature monocytic cells.
- Touch preparation findings are similar to the core biopsy.
- Flow cytometry shows a large abnormal myeloid blast population with expression of CD34, CD15 (subset), CD33, CD13, HLA-DR and CD117. There is also an immature monocytic population showing characteristic expression of CD64, CD11b and CD11c, and possible absence of CD14. Gating on the lymphocytes shows no evidence of a monoclonal B cell population or T cell abnormality based on the markers assayed.
- Cytogenetic analysis shows presence of a pericentric inversion of chromosome 16. FISH analysis using a dual color break apart probe shows rearrangement of CBFB, consistent with inv(16)(p13.1;q22).
Differential diagnosis
- Chronic eosinophilic leukemia:
- Blasts are < 20%
- inv(16)(p13.1;q22) or t(16;16)(p13.1;q22) is absent
- Myeloid / lymphoid neoplasms with eosinophilia and gene rearrangement:
- Acute myeloid leukemia with t(8:21)(q22;q22.1); RUNX1-RUNX1T1:
- Eosinophils do not usually display morphologic or cytochemical abnormalities
- inv(16)(p13.1;q22) or t(16;16)(p13.1;q22) is absent
Additional references
Board review style question #1
An otherwise healthy 15 year old child presents to clinic with fatigue. The patient’s mother states her child has decreased energy and seems to bruise easily. CBC shows anemia and thrombocytopenia. The bone marrow biopsy is hypercellular. The bone marrow aspirate smear (figure) shows the presence of blasts and increased abnormal eosinophils. Cytochemical stains show a dual population of blasts with a subset expressing myeloperoxidase and another subset nonspecific esterase. What is the most likely diagnosis?
- Acute lymphoblastic leukemia
- Acute megakaryoblastic leukemia
- Acute myeloid leukemia with inv(16)
- Acute promyelocytic leukemia with t(15;17)
- Hypereosinophilic syndrome
Board review style answer #1
C. Acute myeloid leukemia with inv(16)
Comment Here
Reference: AML with inv(16)(p13.1;q22) or t(16;16)(p13.1;q22)
Comment Here
Reference: AML with inv(16)(p13.1;q22) or t(16;16)(p13.1;q22)
Board review style question #2
The inv(16)(p13.1;q22) or t(16;16)(p13.1;q22) results in the formation of which abnormal fusion gene?
- BCR-ABL1
- CBFB-MYH11
- KMT2A-MLLT3
- PML-RARA
- RUNX1-RUNX1T1
Board review style answer #2