Table of Contents
Definition / general | Essential features | Terminology | ICD coding | Epidemiology | Sites | Pathophysiology | Etiology | Clinical features | Diagnosis | Radiology description | Radiology images | Prognostic factors | Case reports | Treatment | Clinical images | Gross description | Gross images | Frozen section description | Frozen section images | Microscopic (histologic) description | Microscopic (histologic) images | Cytology description | Cytology images | Positive stains | Negative stains | Molecular / cytogenetics description | Molecular / cytogenetics images | Sample pathology report | Differential diagnosis | Additional references | Board review style question #1 | Board review style answer #1 | Board review style question #2 | Board review style answer #2 | Board review style question #3 | Board review style answer #3Cite this page: Durowoju L, Tretiakova M. Invasive urothelial carcinoma. PathologyOutlines.com website. https://www.pathologyoutlines.com/topic/bladderurothelialinvasivegen.html. Accessed April 25th, 2024.
Definition / general
- Urothelial carcinoma that has penetrated the basement membrane and invaded into the lamina propria or deeper
Essential features
- Histologic characterization and depth of invasion are the most important factors for determining prognosis (Adv Anat Pathol 2015;22:102)
- Urothelial carcinoma is morphologically heterogenous with many variants and subtypes (Surg Pathol Clin 2018;11:713)
- Invasive urothelial carcinoma involving the lamina propria (T1) is often treated with conservative intravesical therapy and mucosal resection (Ann Diagn Pathol 2007;11:395)
- Invasive urothelial carcinoma involving the muscularis propria (T2) is often treated with radical cystectomy (Ann Diagn Pathol 2007;11:395)
Terminology
- Invasive transitional cell carcinoma (historical term)
- WHO classification 2016:
- Infiltrating urothelial carcinoma
- Urothelial carcinoma with divergent differentiation
- With squamous differentiation
- With glandular differentiation
- With trophoblastic differentiation
- Nested urothelial carcinoma
- Including large nested variant
- Microcystic urothelial carcinoma
- Micropapillary urothelial carcinoma
- Lymphoepithelioma-like urothelial carcinoma
- Plasmacytoid / signet ring / diffuse urothelial carcinoma
- Sarcomatoid urothelial carcinoma
- Giant cell urothelial carcinoma
- Poorly differentiated urothelial carcinoma
- Lipid rich urothelial carcinoma
- Clear cell (glycogen rich) urothelial carcinoma
ICD coding
- ICD-O:
- ICD-10: C67.9 - malignant neoplasm of bladder, unspecified
Epidemiology
- Fourth most common malignancy in U.S. men
- Eight most common malignancy associated with death in U.S. men
- Median age: 69 years in men and 71 years in women (Adv Anat Pathol 2015;22:102)
- Incidence:
- 4x higher in men than women
- 2x higher in white men than in black men
Sites
- Lower tract urothelial carcinoma: bladder and urethra represent 90 - 95% (Eur Urol 2015;68:868)
- Upper tract urothelial carcinoma: ureter and renal pelvis represent 5 - 10% (Eur Urol 2015;68:868)
Pathophysiology
- Smoking or other chemical exposures start the process of carcinogenesis by altering the DNA of the bladder mucosa; these genetic alterations lead to unregulated cell growth (Adv Anat Pathol 2019;26:251)
Etiology
- Tobacco smoking: population attributable risk for ever smoking is 50% for both men and women in U.S. (Eur Urol 2013;63:234)
- Occupational exposures: aromatic amines, chlorinated hydrocarbons and polycyclic aromatic hydrocarbons including benzidine (Eur Urol 2013;63:234)
- Genetic predisposition (Eur Urol 2013;63:234)
- Unlike squamous cell carcinoma, not due to indwelling catheters
Clinical features
- Hematuria, irritation / pain during urination, increased urinary frequency or urgency (Urology 2006;67:3)
Diagnosis
- Urine cytology on voided samples (Eur Urol 2013;63:4)
- Cystoscopy with biopsy or transurethral resection (Eur Urol 2013;63:4)
Radiology description
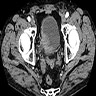
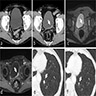
- CT image findings include urothelial thickening / hyperenhancement, asymmetric collecting duct system, urothelial calcifications or the presence of a nodule / mass (Urol Clin North Am 2018;45:389)
- MRI T2 weighted images demonstrate hypointense thickening of bladder wall (Abdom Radiol (NY) 2019;44:3858)
Radiology images
Prognostic factors
- 5 year relative survival: 69% with local disease, 37% with regional disease and 6% with distant disease (American Cancer Society: Survival Rates for Bladder Cancer [Accessed 6 April 2021])
- Stage, with histologic characterization and involvement of the muscularis propria (detrusor muscle) is an important factor for determining prognosis (Adv Anat Pathol 2015;22:102)
- Morphological variants associated with poor prognosis, possibly due to late stage diagnosis and aggressive behavior include:
- Urothelial carcinoma with divergent differentiation (squamous or glandular)
- Micropapillary urothelial carcinoma
- Plasmacytoid urothelial carcinoma
- Nested urothelial carcinoma (Adv Anat Pathol 2015;22:102)
Case reports
- 53 year old woman with IgG4 related kidney disease from the renal pelvis that mimicked urothelial carcinoma (BMC Urol 2015;15:44)
- 56 year old man with testicular metastasis (Case Rep Urol 2014;2014:759858)
- 62 year old man with lymphadenopathies diagnosed with adenocarcinoma of unknown origin (Acta Med Okayama 2019;73:279)
- 65 year old man with recurrence of urothelial carcinoma in an orthotopic neobladder (BMJ Case Rep 2017;2017:bcr2017221052)
- 65 year old man with plasmacytoid urothelial carcinoma and unique clinicopathologic profile on rapid autopsy (Diagn Pathol 2019;14:113)
- 80 year old woman with invasive urothelial carcinoma with chordoid features (Int J Clin Exp Pathol 2015;8:15344)
Treatment
- Conservative therapy is often offered for invasion into the lamina propria (T1) (Ann Diagn Pathol 2007;11:395)
- Conservative therapy includes Bacillus Calmette-Guérin (BCG) intravesical immunotherapy (American Cancer Society: Intravesical Therapy for Bladder Cancer [Accessed 8 April 2021])
- Radical cystectomy is usually required for invasion into the muscularis propria (T2) (Ann Diagn Pathol 2007;11:395)
- Locally advanced cancers are typically treated with chemotherapy, immunotherapy and occasionally radiation (J Urol 2018;199:1129)
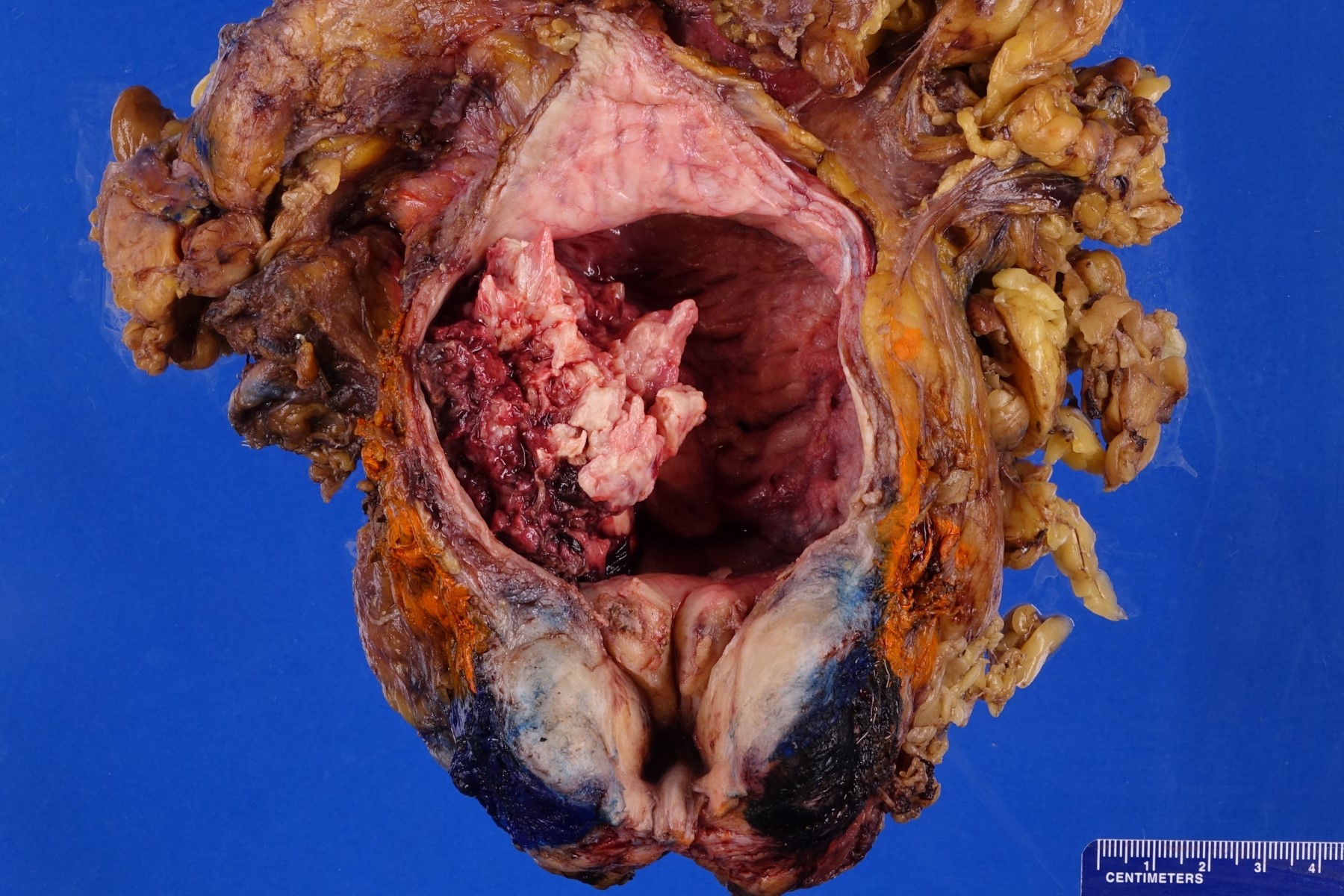
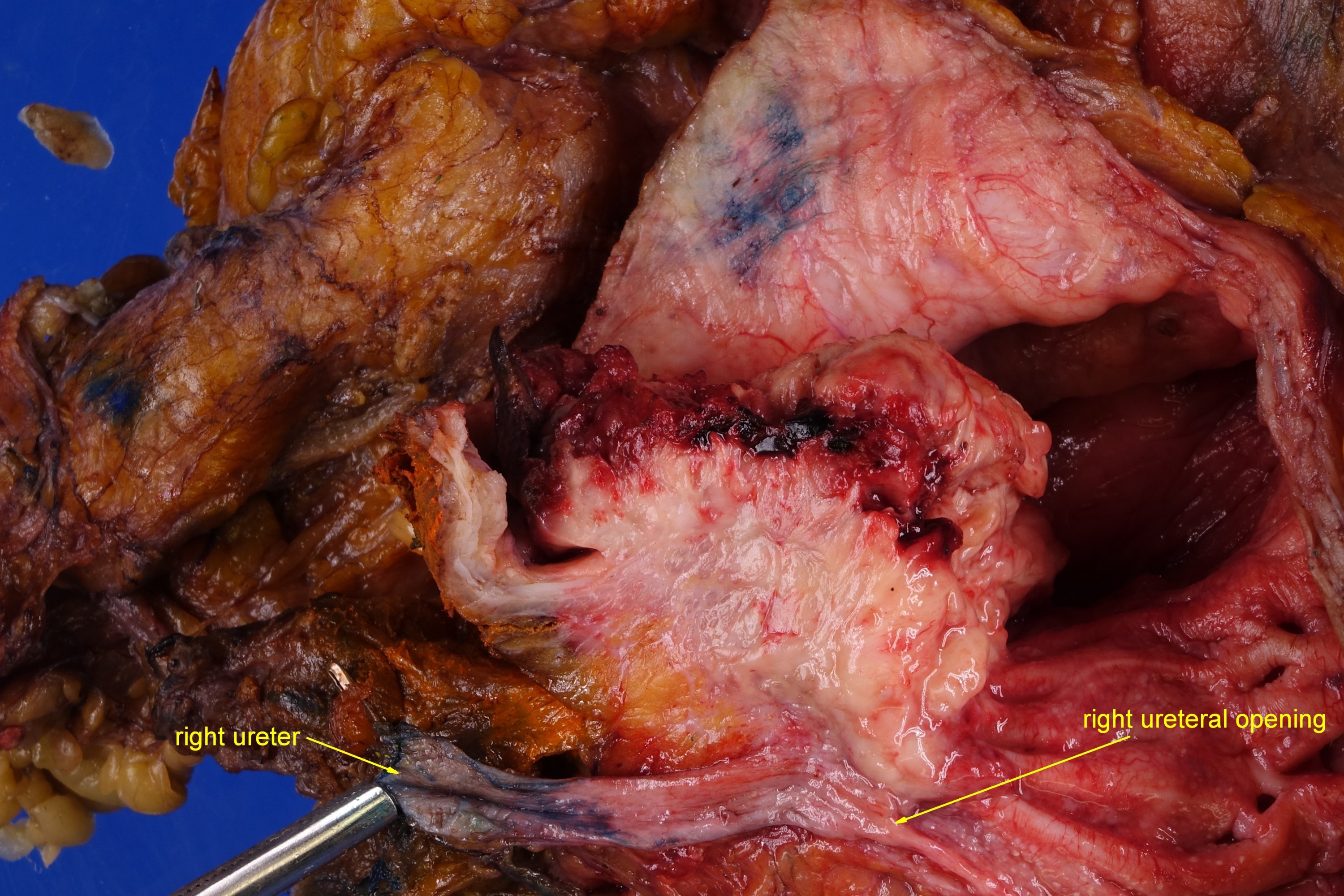
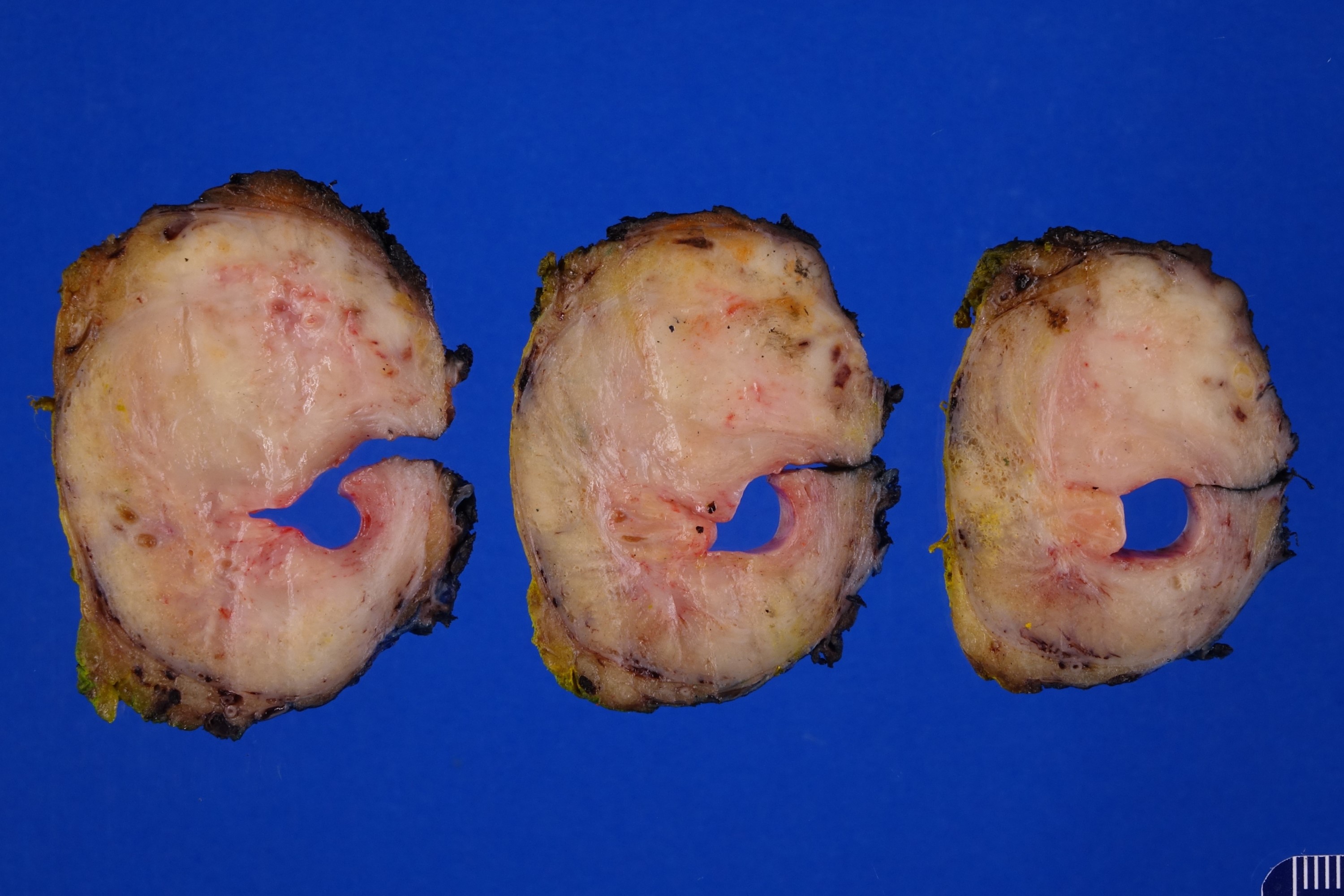
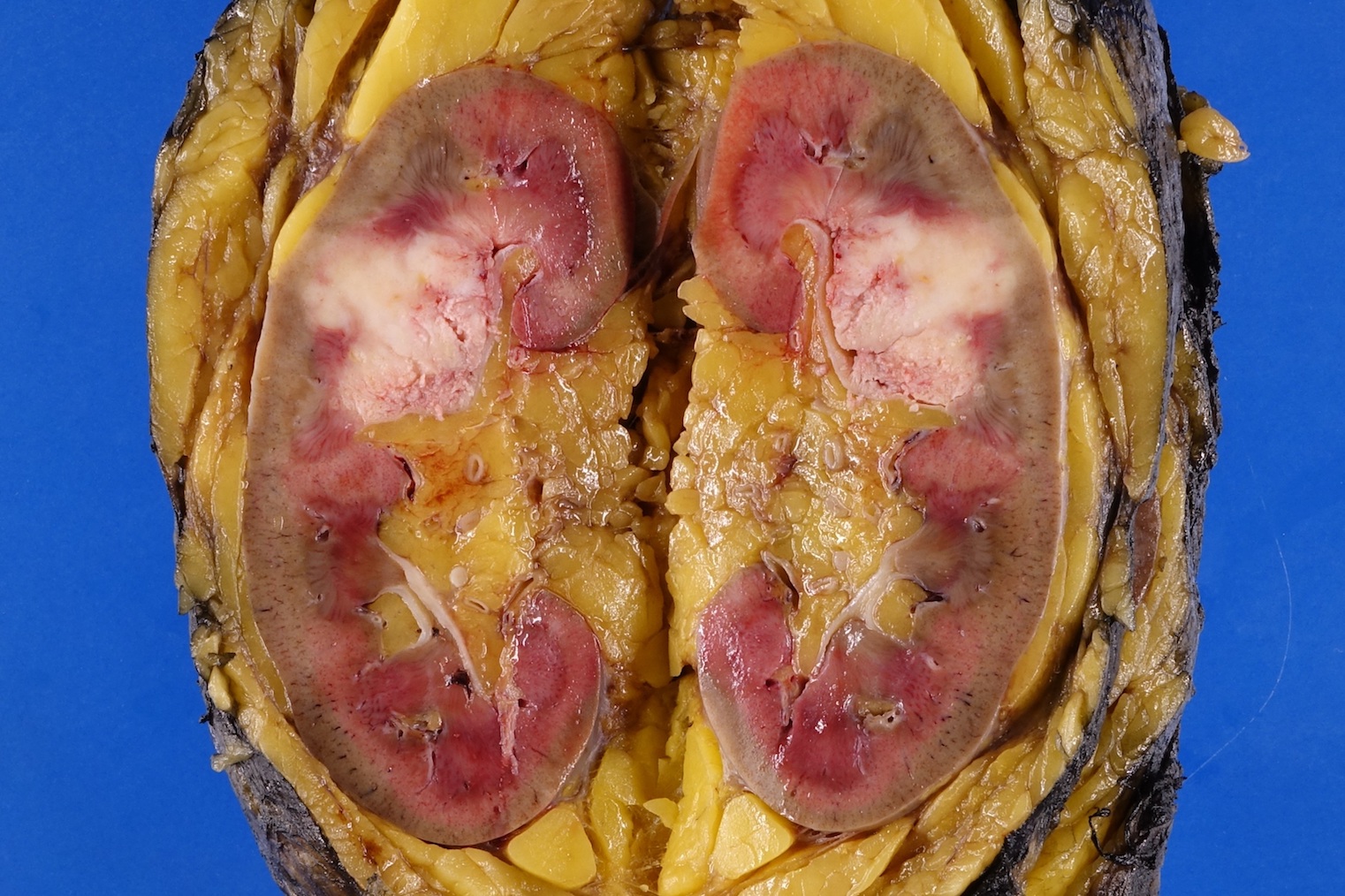
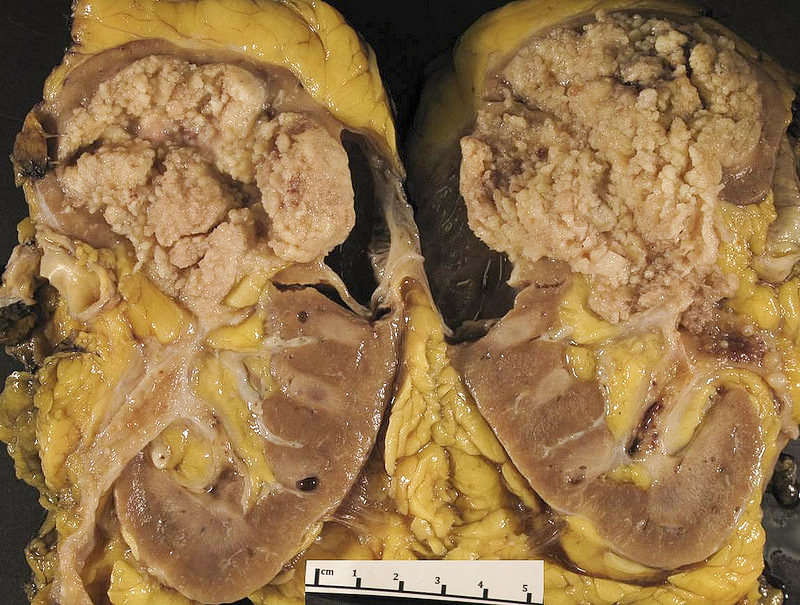
Gross description
- Macroscopic findings range from subtle bladder wall thickening to obvious exophytic mass (Adv Urol 2014;2014:192720)
Gross images
Frozen section description
- Positive margin is defined by the presence of in situ or invasive urothelial carcinoma on frozen section (Ann Diagn Pathol 2015;19:107)
- Ureteric intraoperative frozen sections have significant clinical limitations due to skip lesions, which often occur in multifocal urothelial carcinoma in situ (J Clin Pathol 2010;63:475)
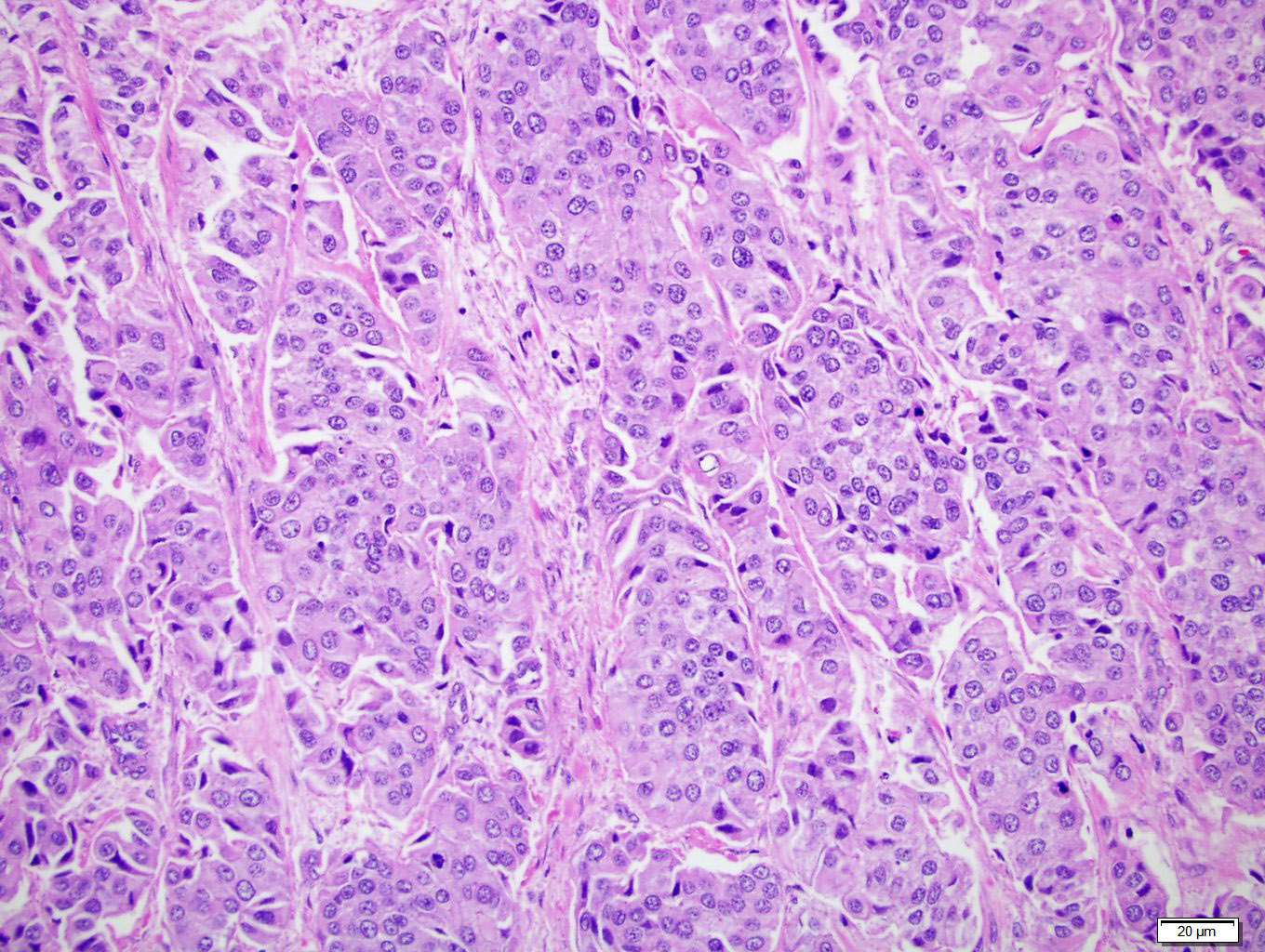
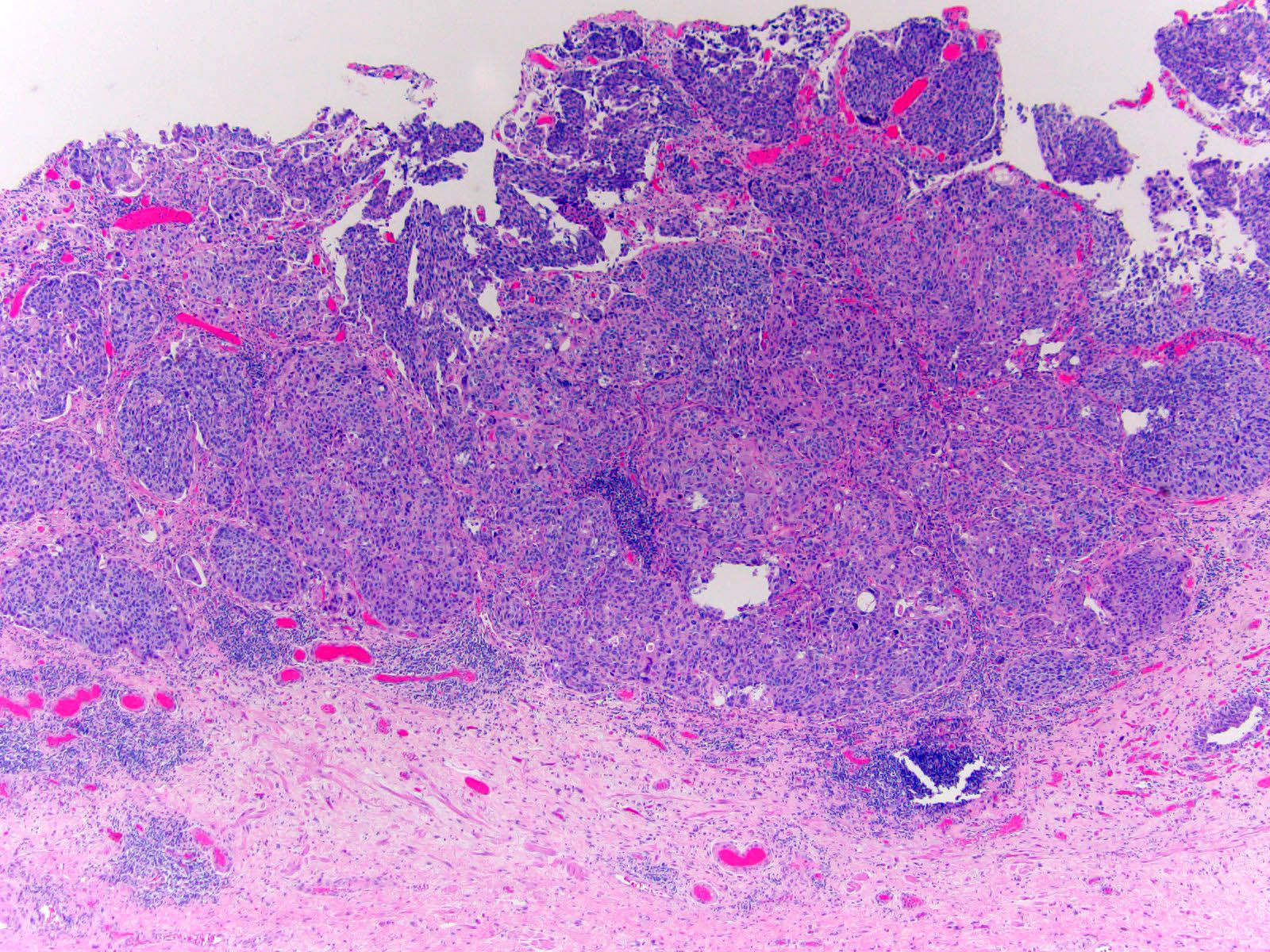
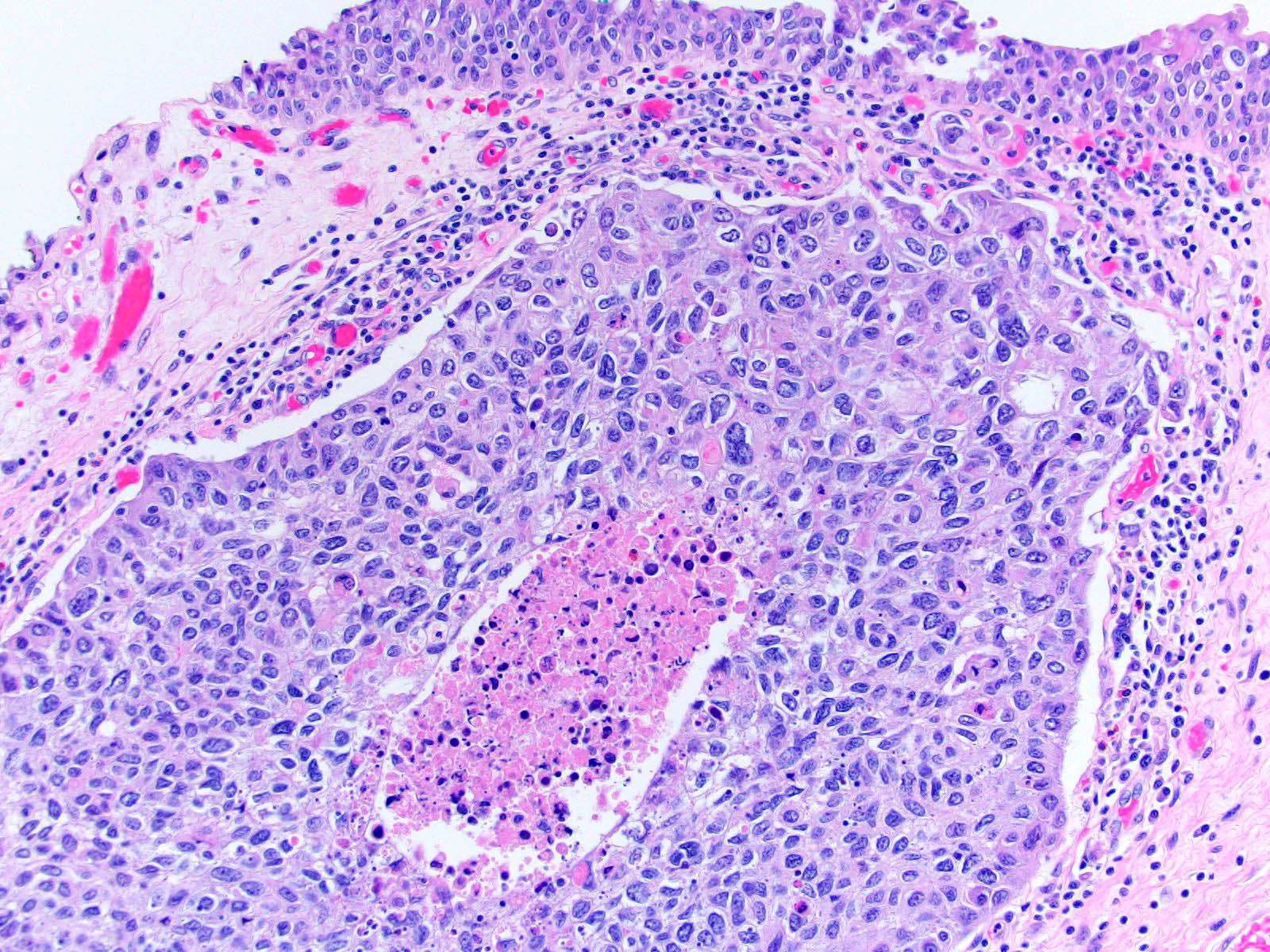
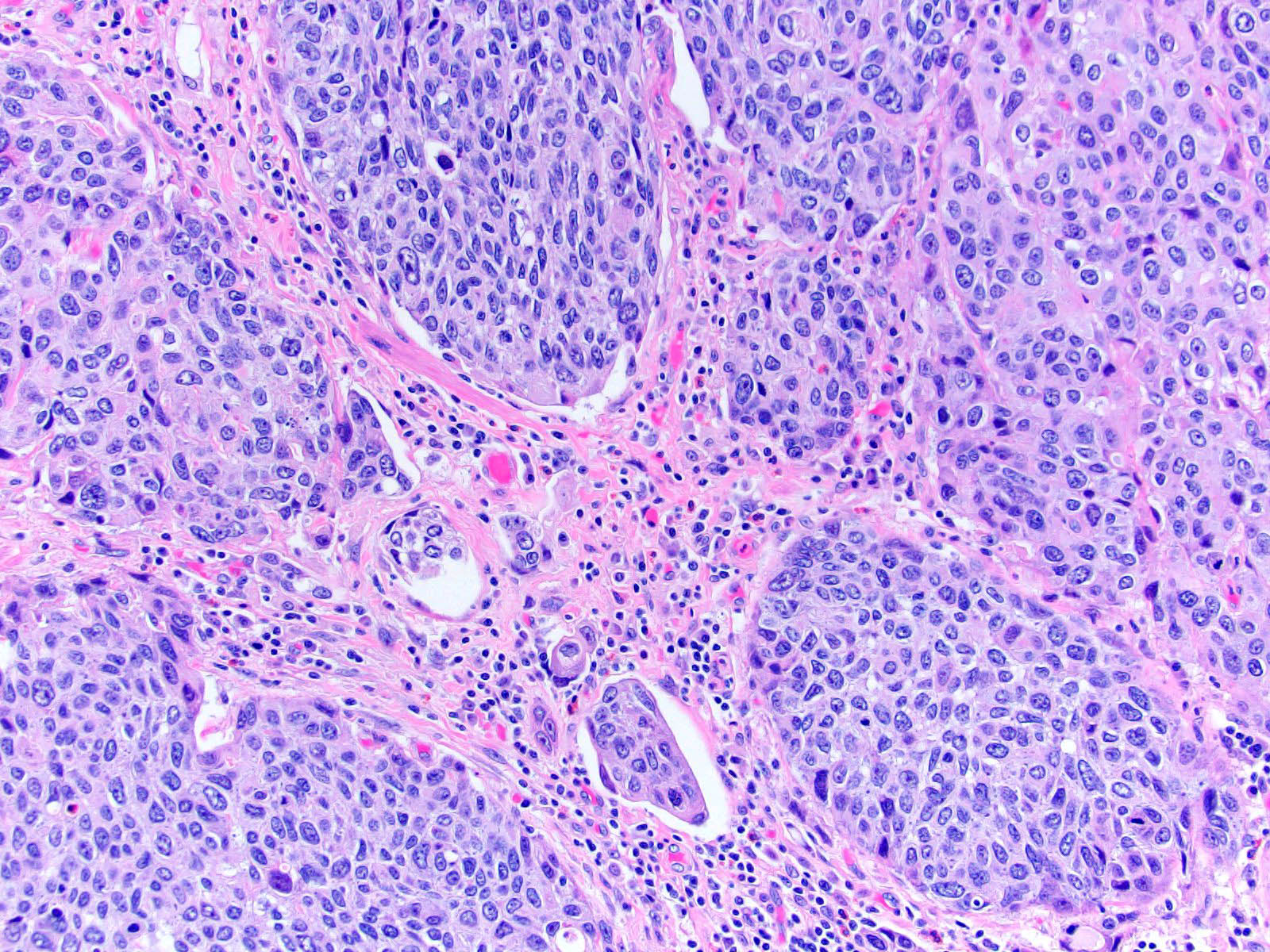
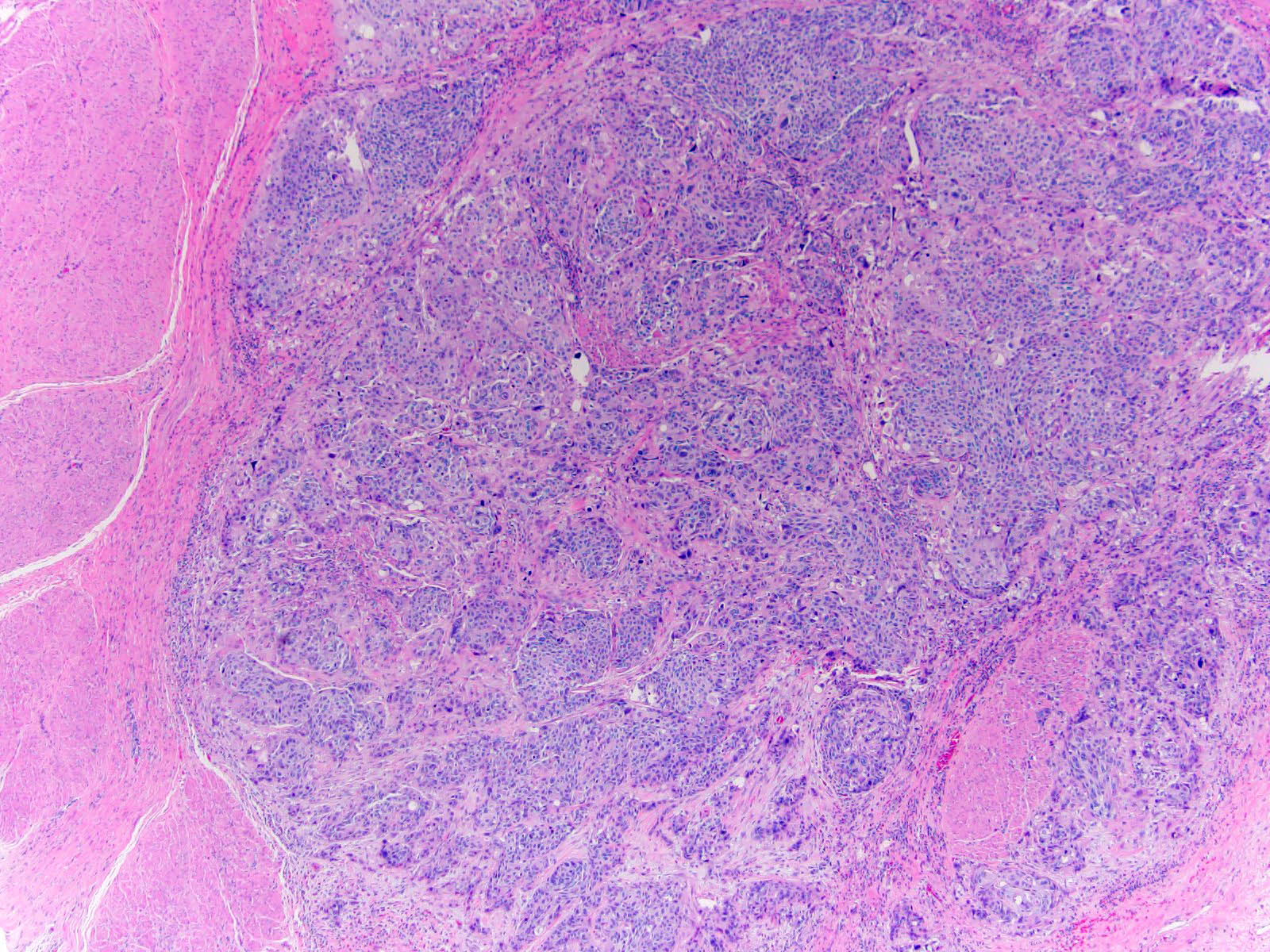
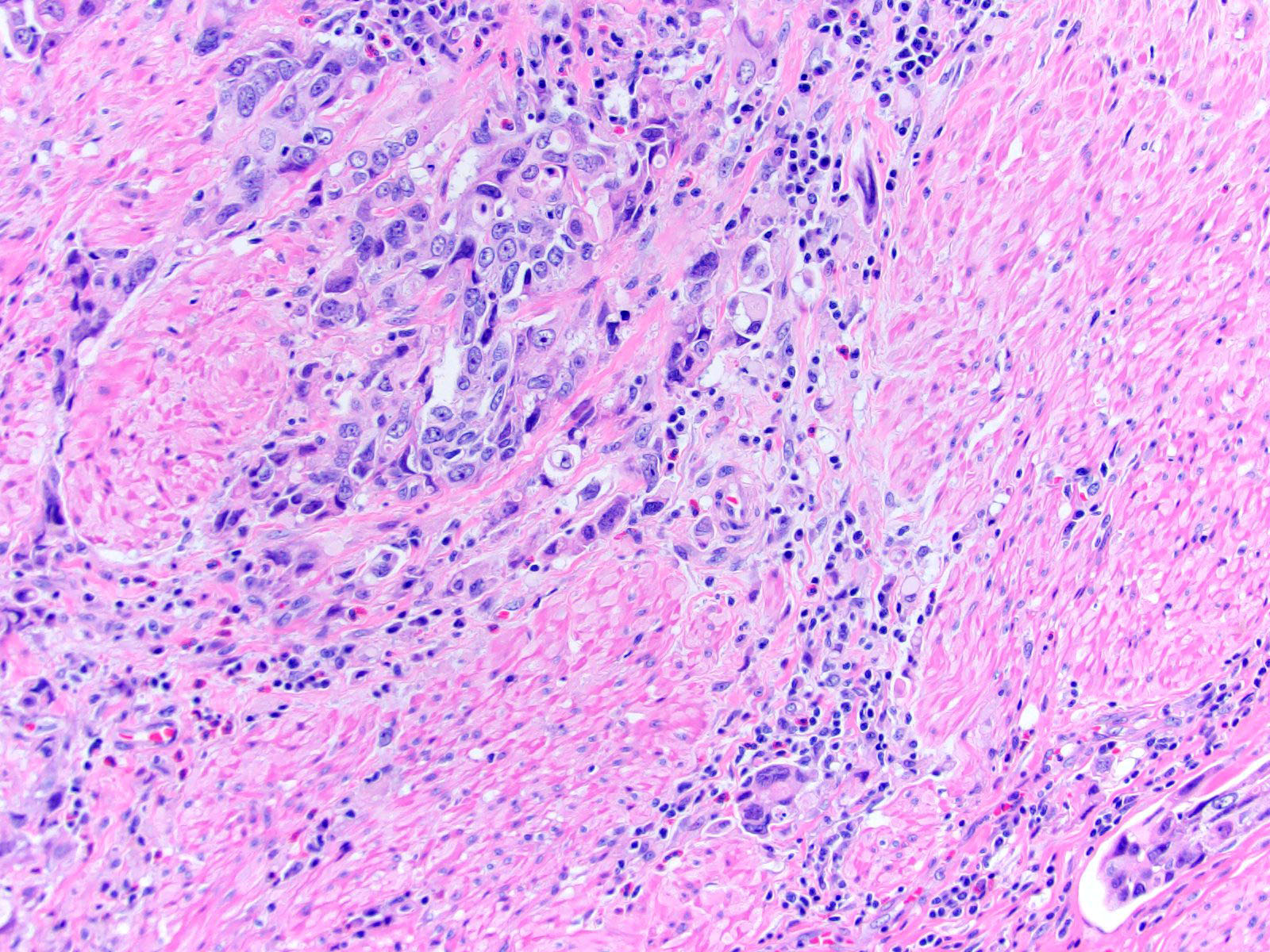
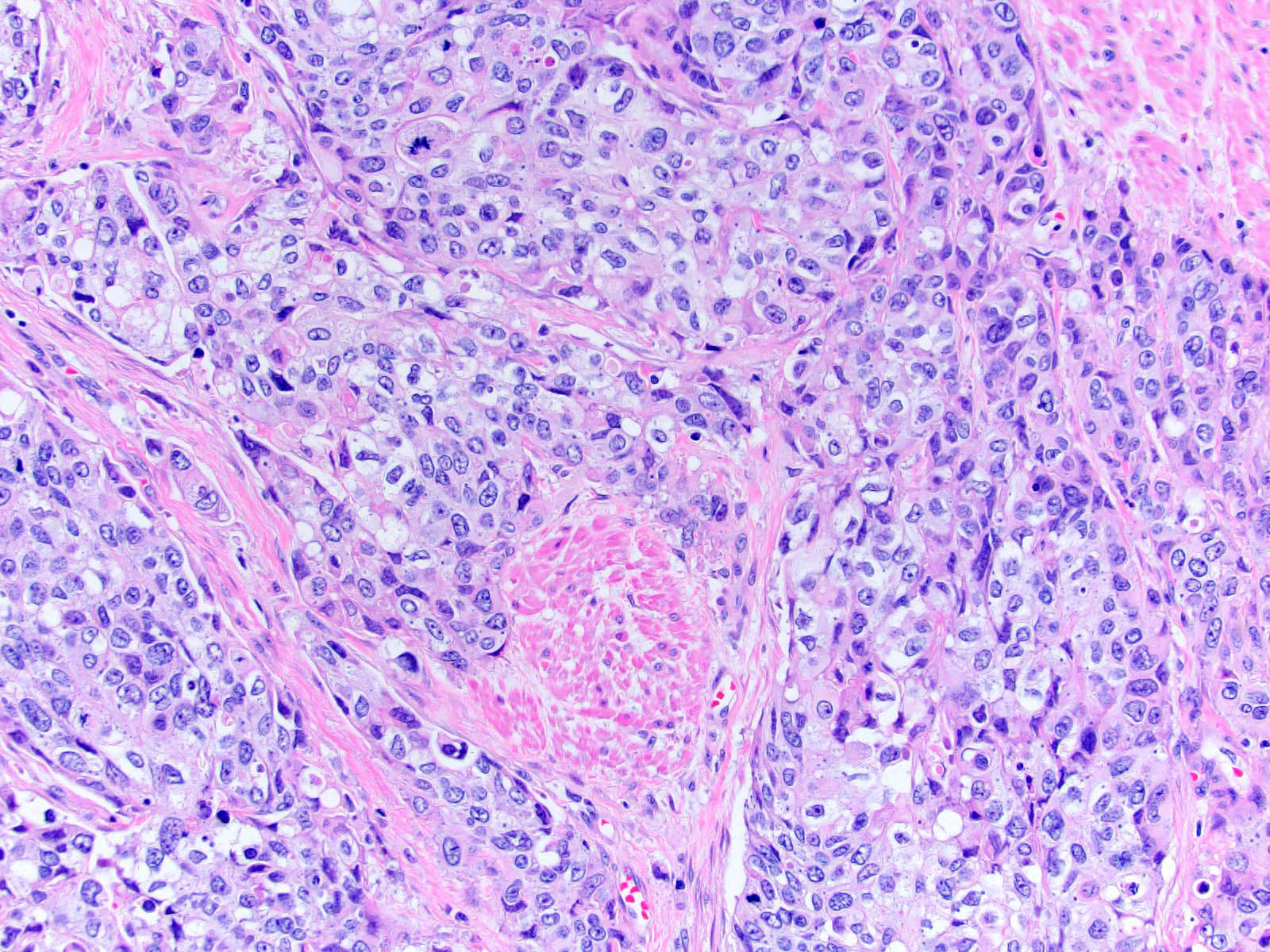
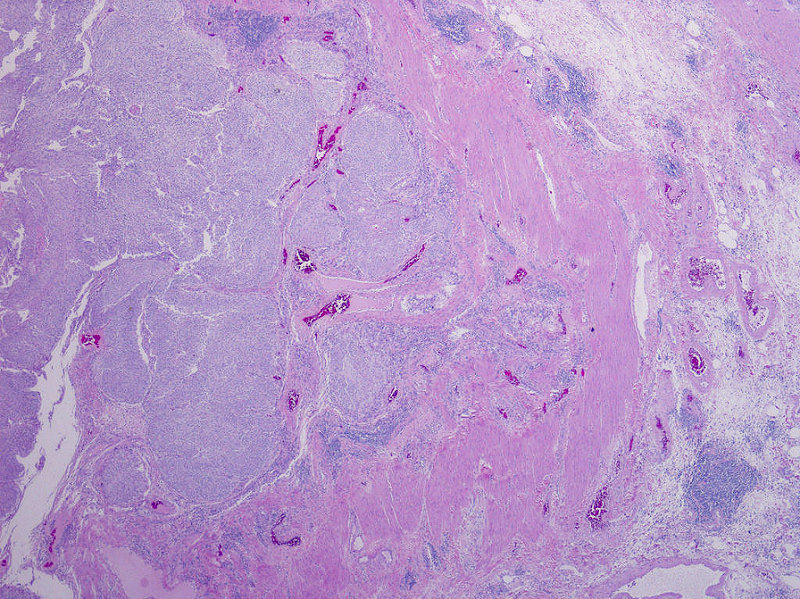
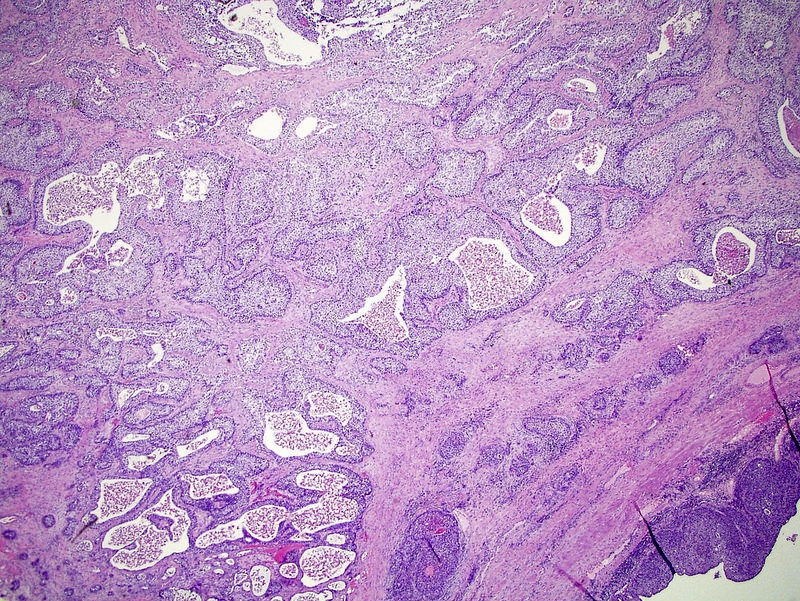
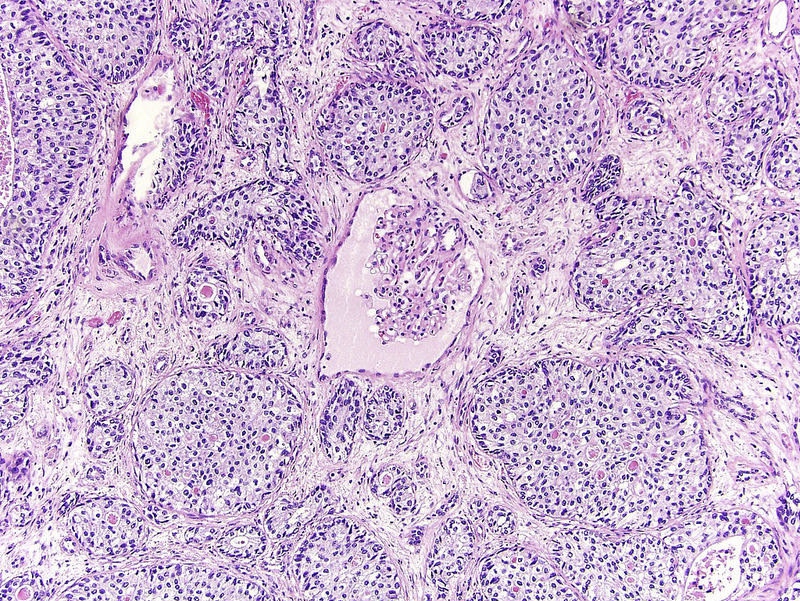
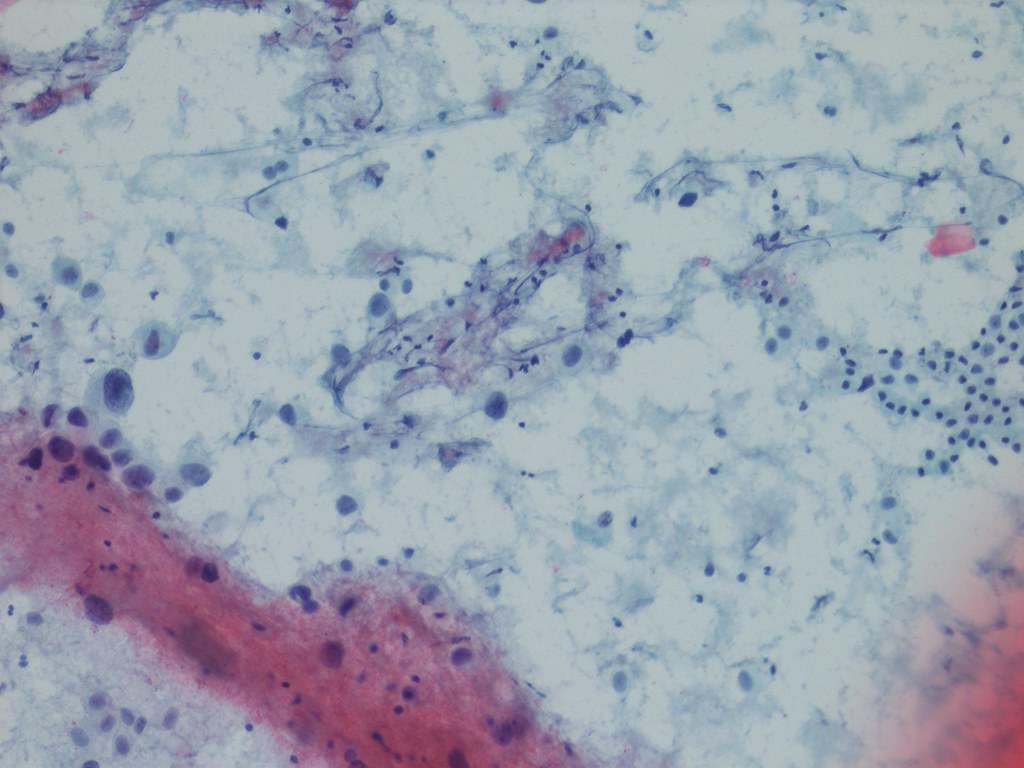
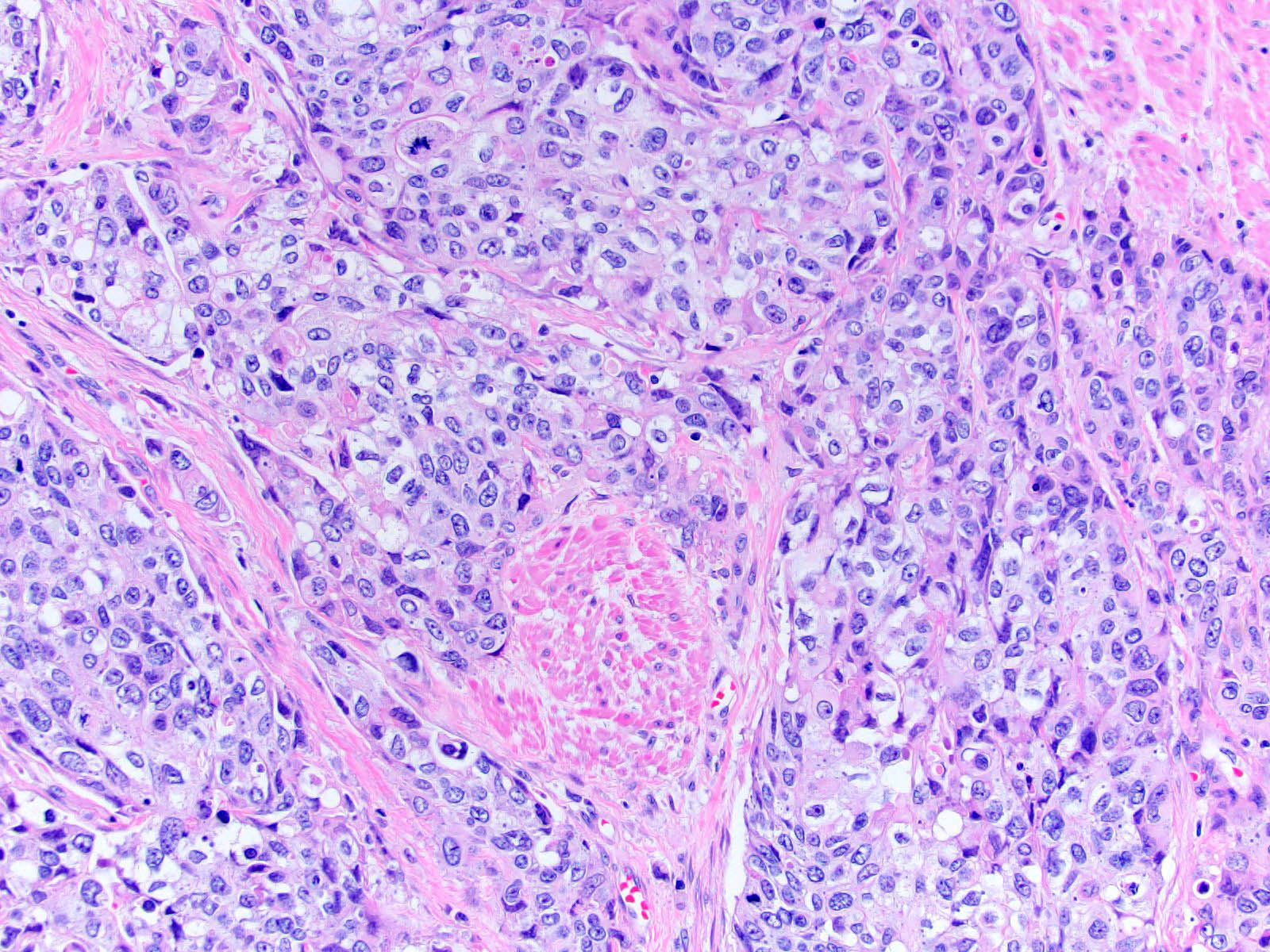
Microscopic (histologic) description
- Neoplastic cells arranged in irregular nests or single cells invading the lamina propria and muscularis propria
- Retraction artifact is often seen and can mimic vascular invasion (Am J Surg Pathol 2001;25:356)
- High grade nuclear features: nuclear pleomorphism, hyperchromasia, high N/C ratio with frequent mitotic figures (Am J Surg Pathol 2001;25:356)
- Note: nested urothelial carcinoma demonstrates bland, low grade cytology (Hum Pathol 2019;94:11)
Microscopic (histologic) images
Contributed by Maria Tretiakova, M.D., Ph.D., Andrey Bychkov, M.D., Ph.D. and Nicole K. Andeen, M.D.
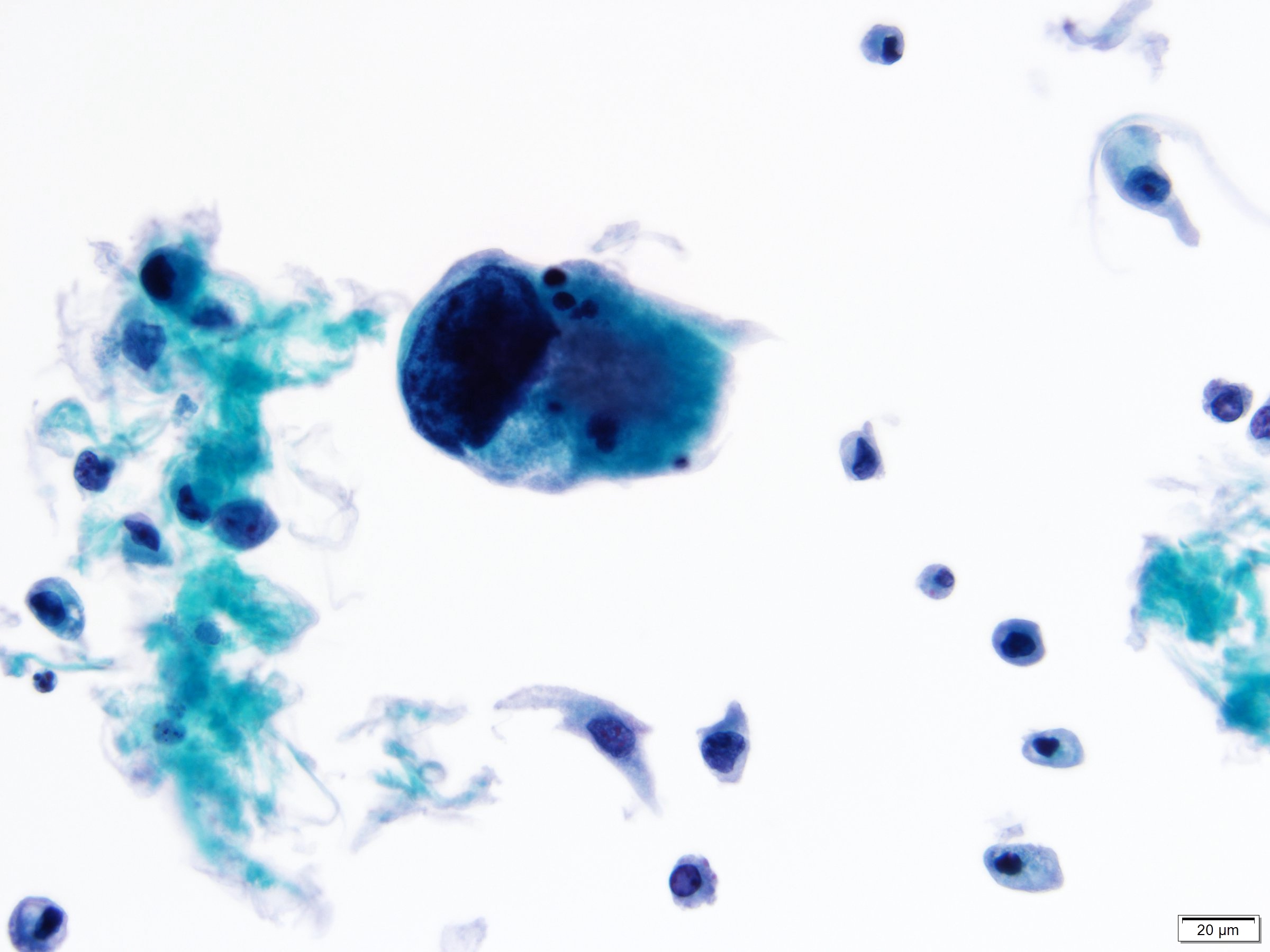
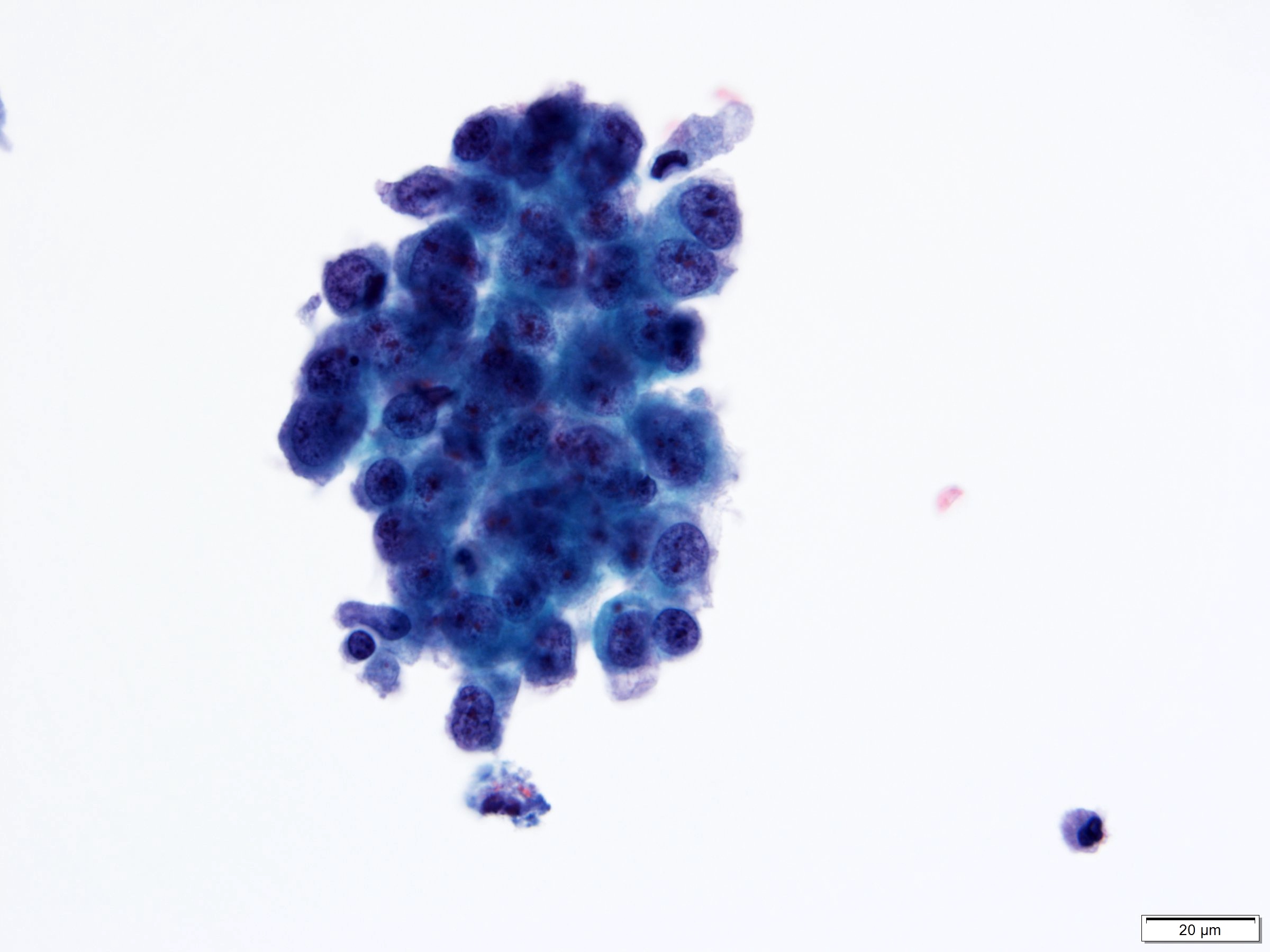
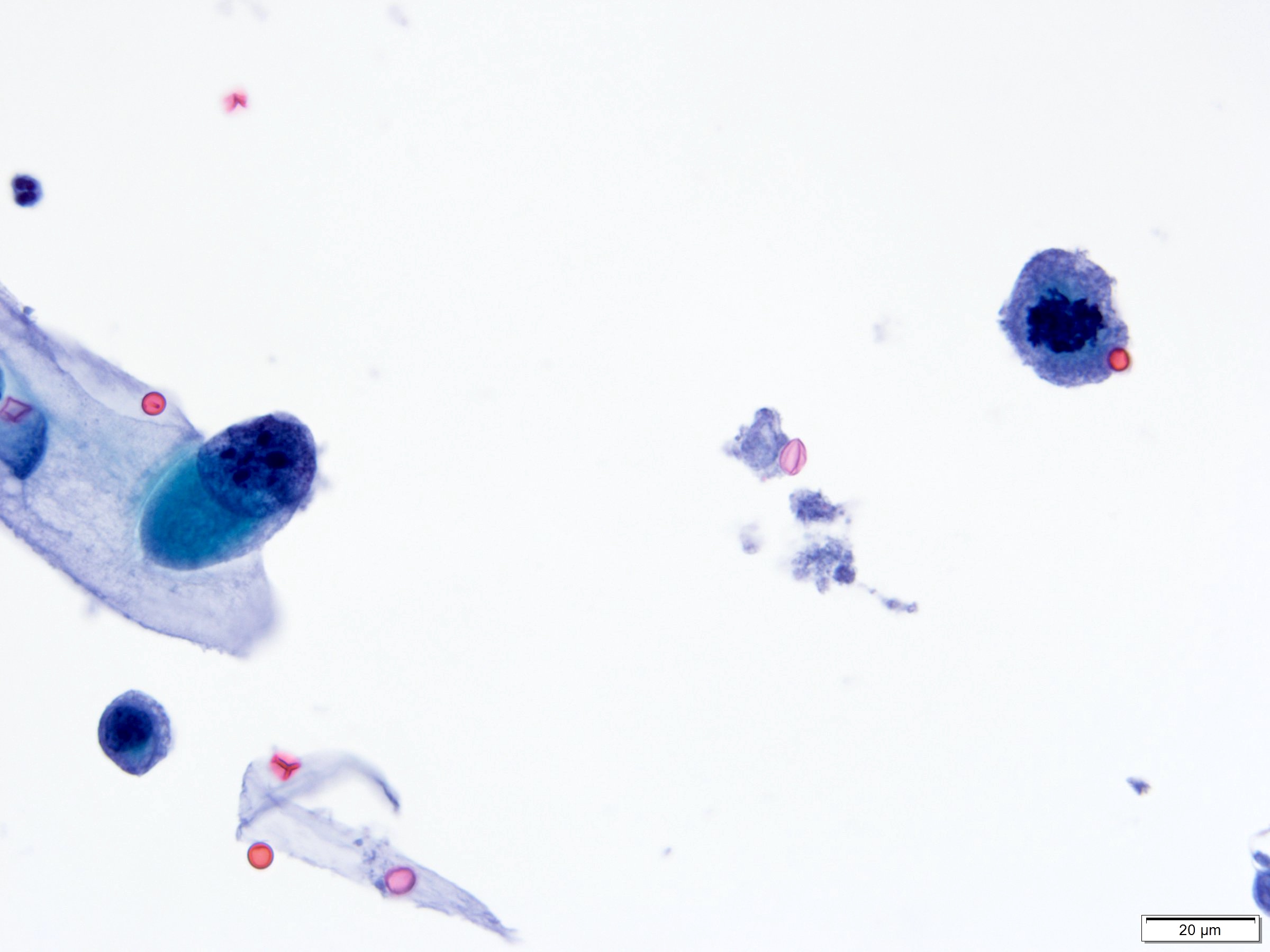
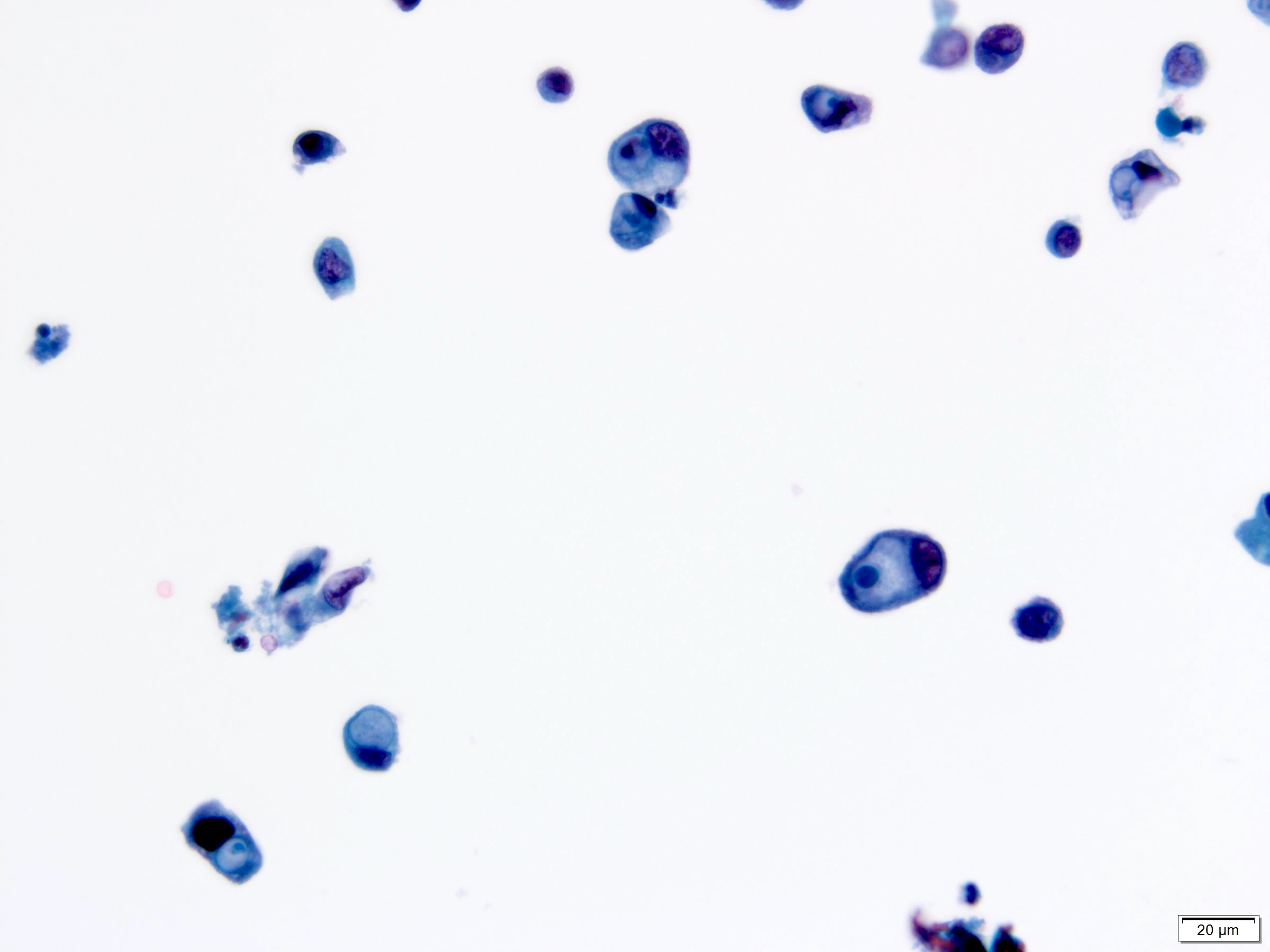
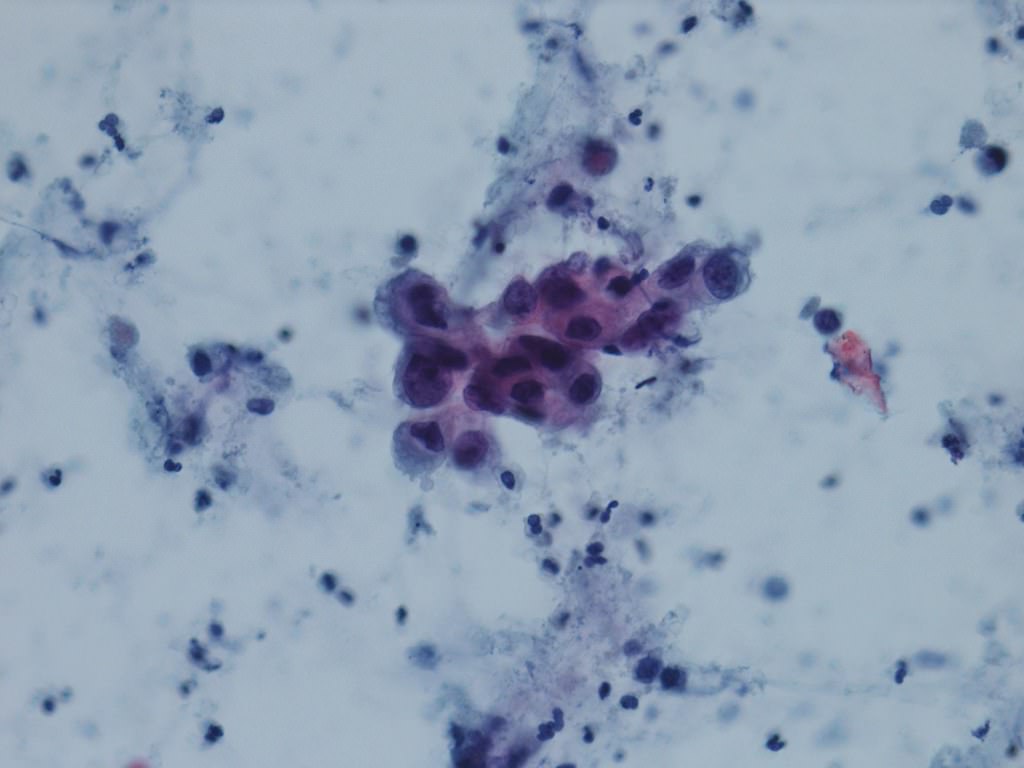
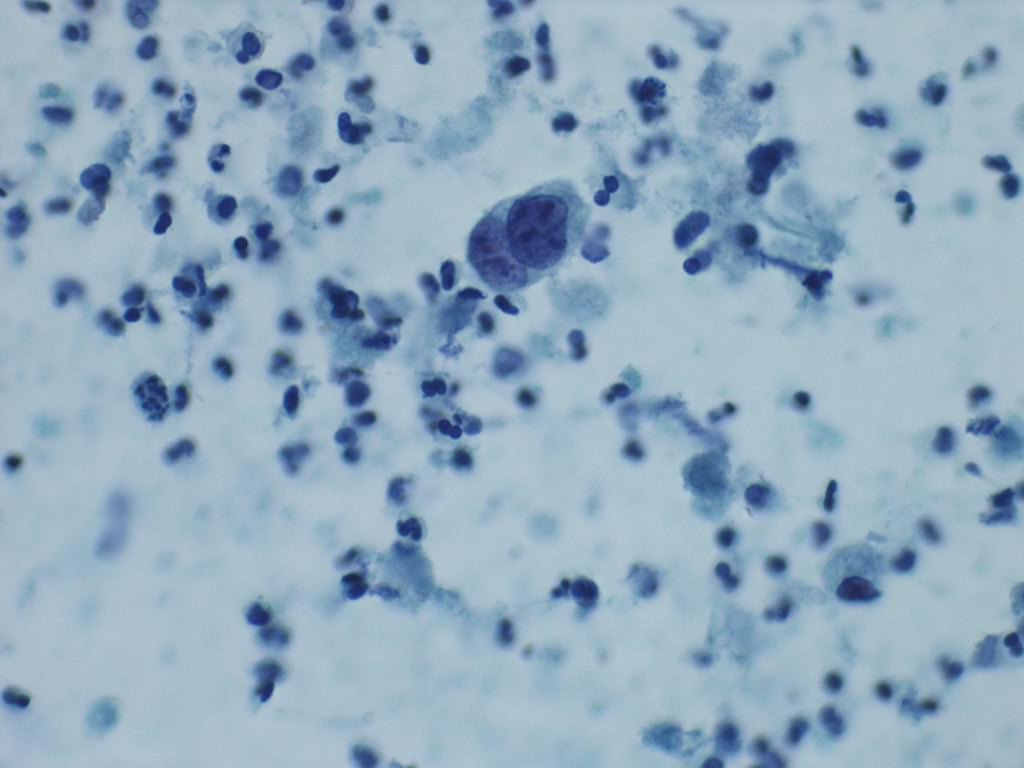
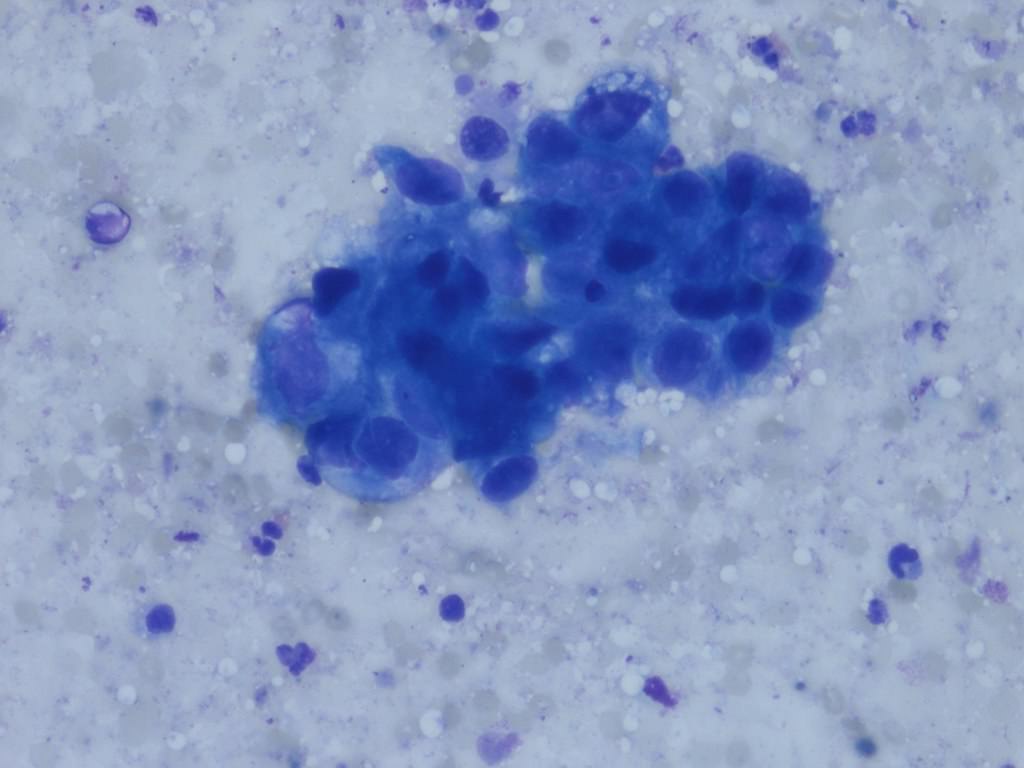
Cytology description
- Urine cytologic detection of urothelial carcinoma has sensitivity and specificity of 13 - 75% and 85 - 100%, respectively (PLoS One 2015;10:e0134940)
- Diagnostic categories are based on The Paris System for Reporting Urinary Cytology
- Cytologic diagnosis of high grade urothelial carcinoma requires > 10 cells with high N/C ratio, irregular chromatin pattern and hyperchromatic nuclei (Cancer Cytopathol 2018;126:207)
Cytology images
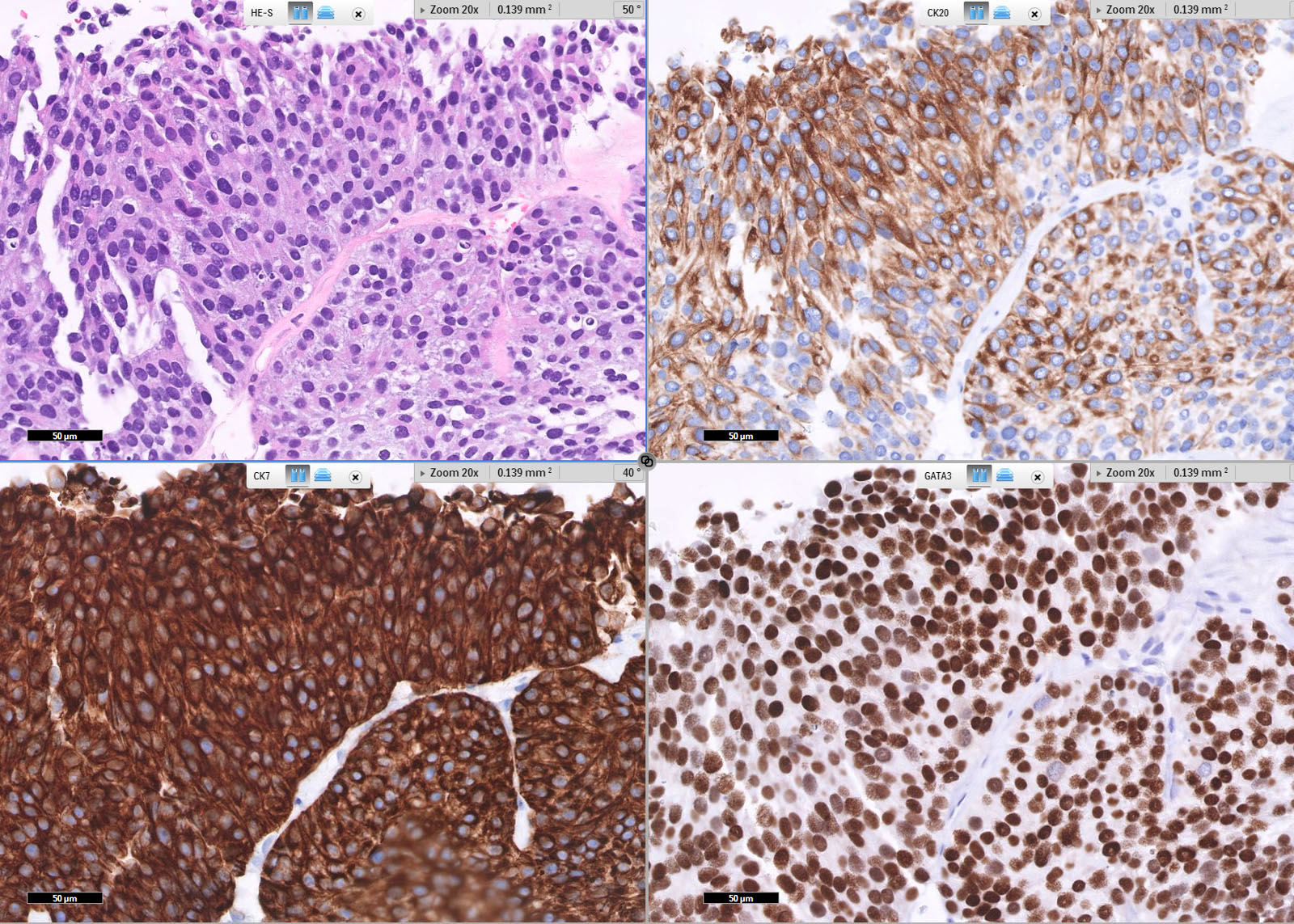
Positive stains
- GATA3, CK7, CK20, p63, p40, uroplakins II and III, HMWCK, thrombomodulin (Pathology 2016;48:543, Am J Surg Pathol 2012;36:1472)
- PDL1 in tumor cells characterized by ≥ 1% is expressed in 13.3 - 46.7% cases (Hum Pathol 2018;81:184, Mod Pathol 2018;3:623)
- Immunostaining can distinguish luminal and basal phenotypes (Sci Rep 2020;10:9743)
Negative stains
Molecular / cytogenetics description
- Most commonly mutated genes are associated with cell cycle progression, including TP53, PIK3CA, RB1 and FGFR3 (Nature 2014;507:315)
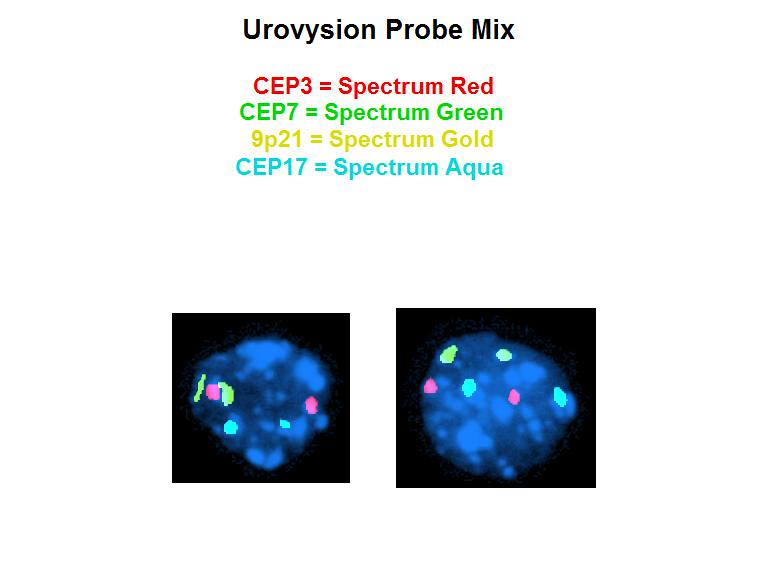
- UroVysion is an FDA approved fluorescent in situ hybridization (FISH) that detects aneuploidy in chromosomes 3, 7 and 17 and loss of 9p21 locus (72% sensitivity and 83% specificity) (BJU Int 2013;112:E372, Curr Opin Urol 2019;29:203)
- Nuclear matrix proteins (NMP22): group of proteins produced by cancerous cells
- NMP22 BladderChek test is an FDA approved qualitative assay for surveillance and detection of bladder cancer (50% sensitivity and 87% specificity) (Curr Opin Urol 2019;29:203)
- Bladder tumor antigen (BTA): antigen produced by cancerous cells
- BTA Trak (quantitative) and BTA Stat (qualitative) are FDA approved assays for surveillance (68 - 77.5% sensitivity and 50 - 75% specificity) (Curr Opin Urol 2019;29:203)
Molecular / cytogenetics images
Sample pathology report
- Bladder, TURB:
- Urothelial carcinoma, high grade (see comment)
- Type / grade comment: micropapillary (20%)
- Associated lesions: carcinoma in situ
- Extent: lamina propria invasion, extensive (pT1)
- Angiolymphatic invasion: present
- Muscularis propria: present, uninvolved
Differential diagnosis
- Inverted urothelial papilloma:
- Smooth or dome shaped surface with monotonous cells growing in an endophytic pattern without the presence of lamina propria invasion (Hum Pathol 2019;92:18)
- Prostatic adenocarcinoma:
- Paraganglioma:
- Similar nested architecture, prominent vascular network and distinguishable by immunohistochemistry
- Positive for S100 (sustentacular pattern), synaptophysin and chromogranin (Histopathology 2019;74:77)
- Negative for AE1 / AE3 and CK7
- Potential pitfall: also positive for GATA3 (J Mod Pathol 2013;26:1365)
- Nephrogenic adenoma:
- Rare nuclear atypia and distinguishable by immunohistochemistry: positive for PAX8, PAX2 and AMACR (Histopathology 2019;74:77)
- Cystitis cystica / glandularis:
- Lacks cytologic atypia, necrosis, stromal reaction and invasion into lamina propria and muscularis propria (Histopathology 2019;74:77, Ann Diagn Pathol 2019;38:11)
- Renal cell carcinoma (for renal pelvis):
- Lack of urothelial carcinoma in situ is the most helpful distinguishing factor
- Positive PAX8, negative p63 and negative GATA3 immunoprofile favors renal cell carcinoma; however, stain interpretation and immunohistochemical profile overlap can create diagnostic challenges (Adv Anat Pathol 2018;25:387)
Additional references
Board review style question #1
A 76 year old man with gross hematuria was found to have invasive urothelial carcinoma on transurethral biopsy. Which of the following is not a risk factor for urothelial carcinoma but is a risk factor for developing squamous cell carcinoma?
- Tobacco smoke
- Polycyclic aromatic hydrocarbon exposure
- Benzidine exposure
- Indwelling catheter
Board review style answer #1
Board review style question #2
Which of the following histopathologic subtypes of urothelial carcinoma is most likely to be associated with a worse prognosis?
- Clear cell urothelial carcinoma
- Microcystic urothelial carcinoma
- Conventional high grade urothelial carcinoma
- Plasmacytoid urothelial carcinoma
Board review style answer #2
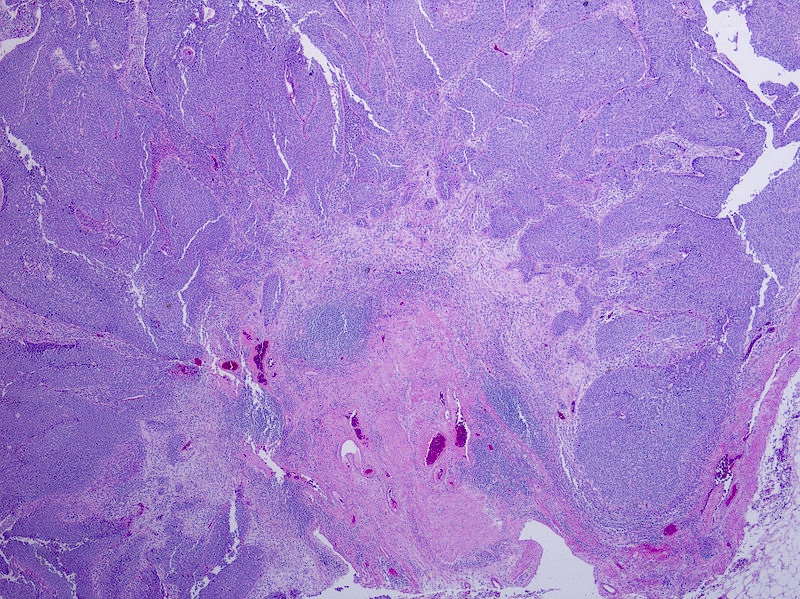
Board review style question #3
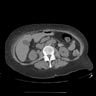
An 85 year old man has a mass involving the prostate and bladder shown above. Which of the following findings is most supportive of invasive urothelial carcinoma over prostatic adenocarcinoma?
- Sheets of monotonous cells with amphiphilic cytoplasm and prominent nucleoli
- Negative for PSA and CK7
- Positive for GATA3 and HMWCK
- Focus of squamous differentiation
Board review style answer #3
C. Positive for GATA3 and HMWCK immunohistochemical stains
Comment Here
Reference: Invasive urothelial carcinoma
Comment Here
Reference: Invasive urothelial carcinoma