Table of Contents
Definition / general | Essential features | Terminology | ICD coding | Epidemiology | Sites | Pathophysiology | Etiology | Clinical features | Diagnosis | Prognostic factors | Case reports | Treatment | Gross description | Gross images | Microscopic (histologic) description | Microscopic (histologic) images | Virtual slides | Positive stains | Negative stains | Electron microscopy description | Molecular / cytogenetics description | Sample pathology report | Differential diagnosis | Additional references | Practice question #1 | Practice answer #1Cite this page: Lechner A, Parra-Herran C. Adenosarcoma. PathologyOutlines.com website. https://www.pathologyoutlines.com/topic/cervixadenosarcoma.html. Accessed August 31st, 2025.
Definition / general
- Rare, mixed lesion with malignant mesenchymal and benign glandular components
Essential features
- Leaf-like glands composed of bland epithelium and condensed periglandular stroma with atypia and mitotic activity
- Most are of low malignant potential with good probability of disease free and overall survival
- 3 most important prognostic factors are: (1) presence of sarcomatous overgrowth, (2) histologic grade and (3) depth of myometrial invasion
- Stromal cells may lose CD10 and PR when sarcomatous overgrowth is present; other markers may be gained in cases of heterologous differentiation
- Recurrence may consist solely of sarcomatous component
Terminology
- Also called Müllerian adenosarcoma
ICD coding
Epidemiology
- Müllerian adenosarcoma accounts for 5.5 - 7% of uterine sarcomas (Cancer 1993;71:1702, Histopathology 2009;54:355)
- Most patients with cervical adenosarcoma are young (a third were under 15 years of age in one series) with median age of 37 - 39 years at presentation (Int J Gynecol Pathol 1995;14:223)
Sites
- Müllerian adenosarcoma can occur in multiple sites:
- Uterine corpus > cervix > ovary / pelvis
- Extrauterine adenosarcoma may show associated endometriosis
- 10% occur in the cervix (Gynecol Oncol 2016;143:636)
- Patients with cervical primaries are younger whereas corpus and ovarian primaries typically affect postmenopausal patients (Gynecol Oncol 2016;143:636)
Pathophysiology
Etiology
- Multiple small series have implicated hyperestrogenism (e.g., in the setting of tamoxifen therapy or ovarian thecoma) as a risk factor for uterine sarcomas including adenosarcoma (Int J Gynecol Pathol 1996;15:222, Gynecol Oncol 1985;21:135)
- Due to a small population, these associations may be coincidental
- Prior pelvic radiation therapy may increase risk
Clinical features
- Common presenting features include (Adv Anat Pathol 2010;17:122):
- Abnormal vaginal bleeding (most common)
- Pelvic pain
- Abdominal mass
- Vaginal discharge
- Lesion is frequently interpreted as an endometrial or endocervical polyp on clinical and radiologic evaluation
- Recurrence is usually composed of solely sarcomatous component
Diagnosis
Prognostic factors
- Most uterine adenosarcomas are of low malignant potential with favorable prognosis:
- 83% are FIGO stage I at time of diagnosis with 63 - 84% 5 year overall survival (Gynecol Oncol 2010;119:305)
- Low grade histology, absence of myometrial invasion or sarcomatous overgrowth all confer good prognosis (Oncol Rep 1998;5:939)
- Presence of a tumor stalk is an independent protective factor for both disease free and overall survival (Front Oncol 2019;9:237)
- Cervical primary is associated with improved disease free survival compared to uterine corpus primary (Front Oncol 2019;9:237)
- Adverse prognostic factors are:
- Myometrial and vascular space invasion: myometrial invasion is seen in 14% of cases and is associated with adverse outcome (Hum Pathol 1990;21:363, Int J Gynecol Pathol 1992;11:75)
- Sarcomatous overgrowth: highly associated with extrauterine spread at presentation; high rates of recurrence and death (Am J Surg Pathol 1989;13:28)
- High grade sarcomatous component: highly associated with extrauterine spread, recurrences and metastases; usually associated with overgrowth, even if the high grade component is minor (Am J Surg Pathol 2017;41:1513)
- Presence of heterologous elements confers a worse outcome (Front Oncol 2019;9:237)
- Recurrence of uterine adenosarcoma (up to 46%) with mean time to recurrence of 18.3 months (Gynecol Oncol 2014;135:455)
Case reports
- 14 year old girl with a vaginal mass (Gynecol Oncol Rep 2019;32:100525)
- 15 year old girl whose tumor had sarcomatous overgrowth and heterologous elements (J Gynecol Oncol 2010;21:125)
- 32 year old woman whose tumor metastasized to the ovary (Turk J Obstet Gynecol 2017;14:195)
- 37 year old woman with clinical endocervical polyp (Int J Gynecol Cancer 2004;14:1024)
- 45 year old woman with vaginal bleeding and prior pelvic irradiation (Pathologica 2020;112:219)
Treatment
- Hysterectomy with bilateral salpingectomy oophorectomy is the standard of treatment and is curative in most cases
- Radiation therapy is considered in patients with advanced stage (FIGO stage II or more) or after recurrence
- Fertility sparing surgery (FSS) via cervical conization may be an option for a subset of patients:
- Recent data show no decrease in disease free or overall survival after FSS for FIGO stage IA tumors (Front Oncol 2019;9:237)
- Older reports do not support this finding; however, patients from these groups were of higher clinical stage (e.g., FIGO IB)
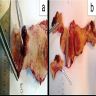
Gross description
- Broad based or sessile polypoid mass on gross examination
- Cut surface displays predominantly solid tumor with numerous cysts
- Reference: Adv Anat Pathol 2010;17:122
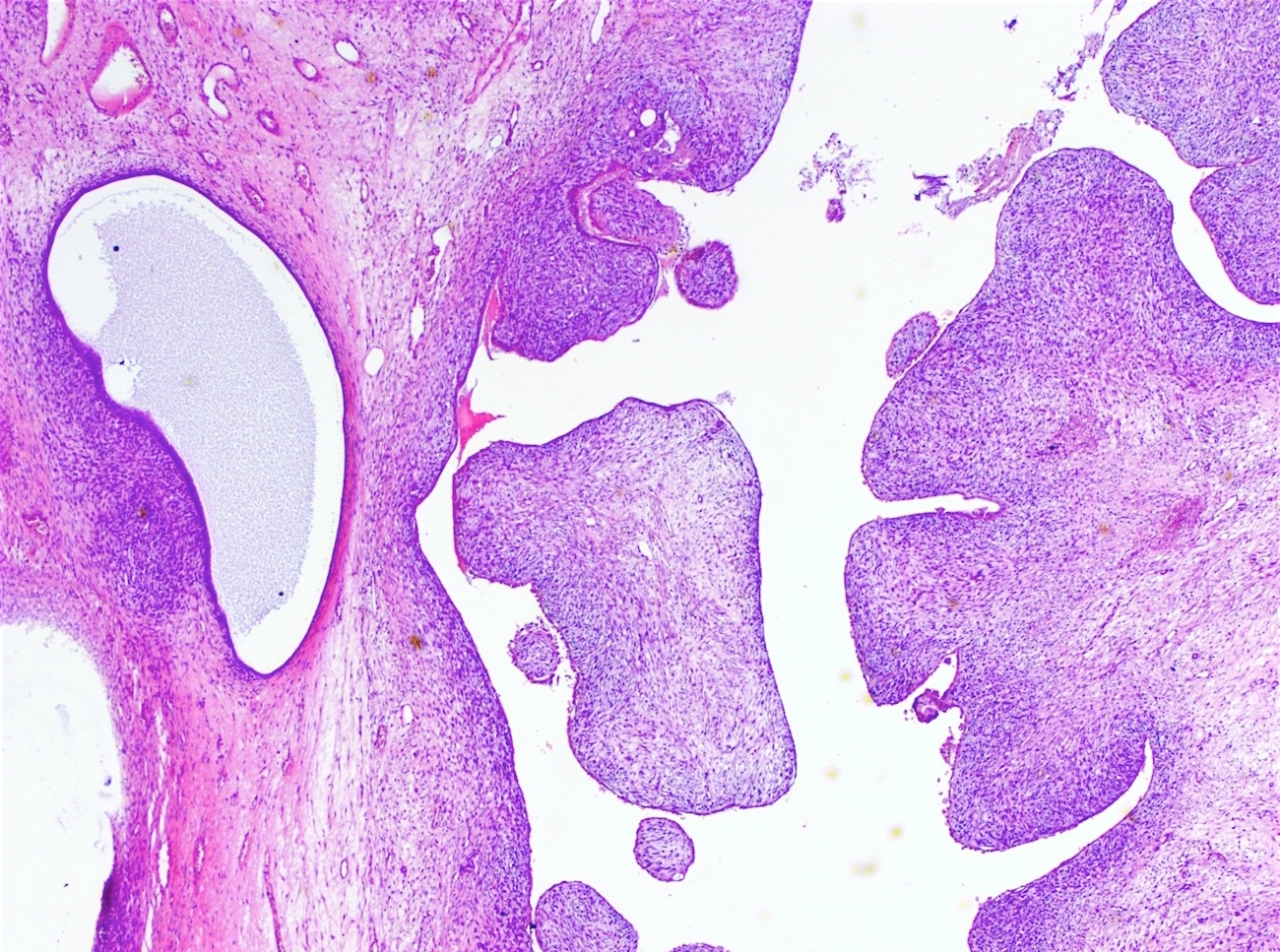
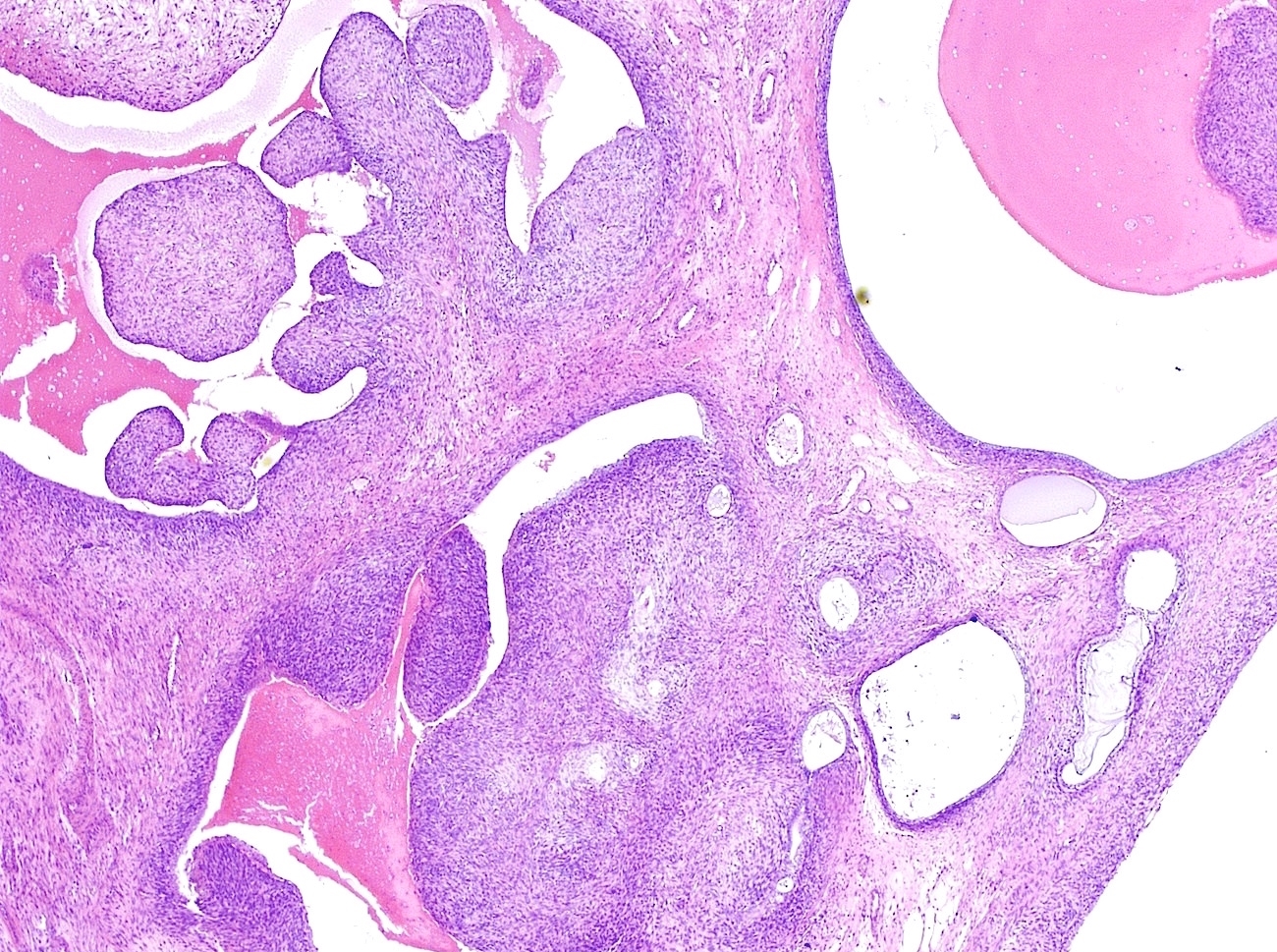
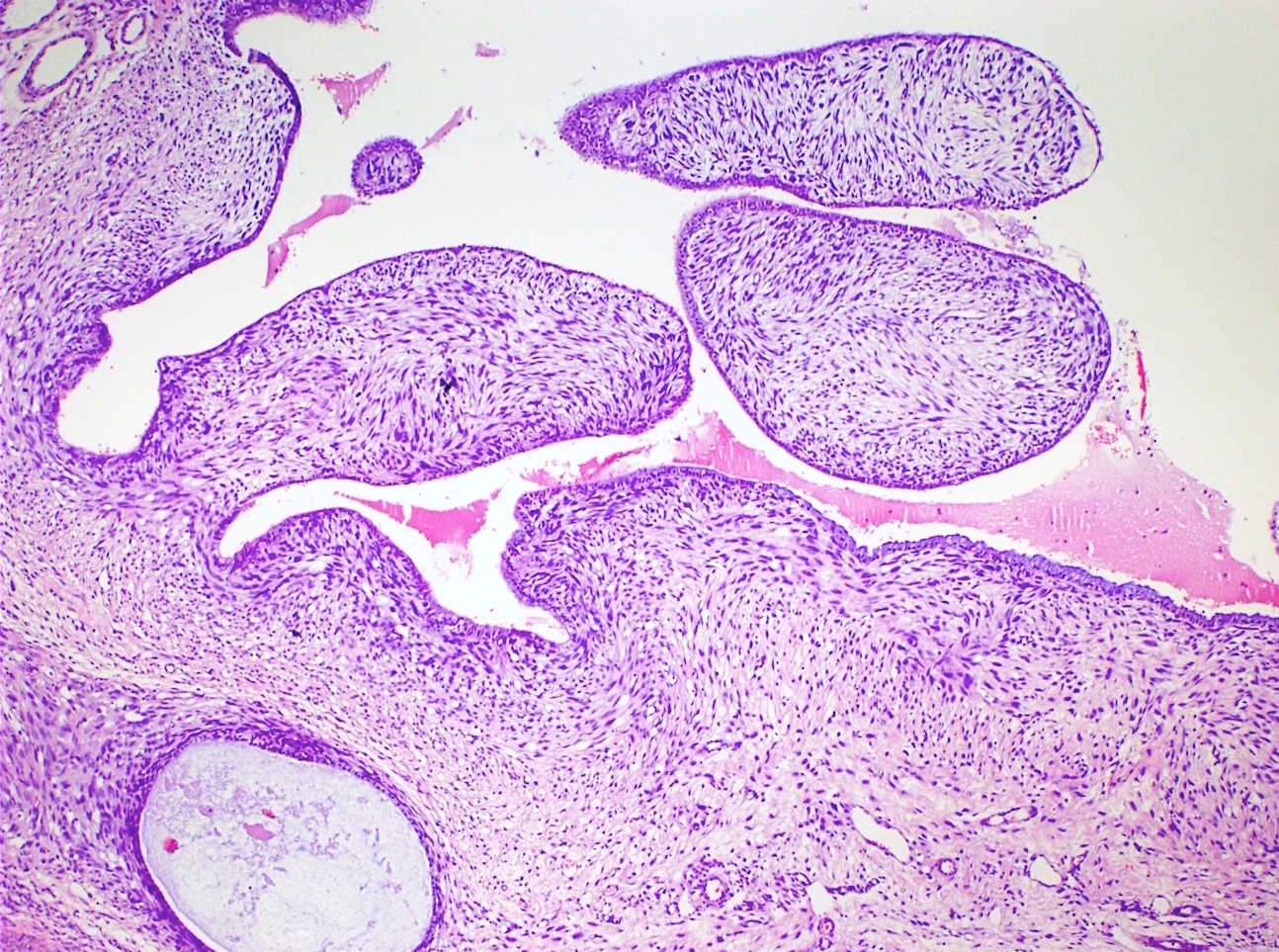
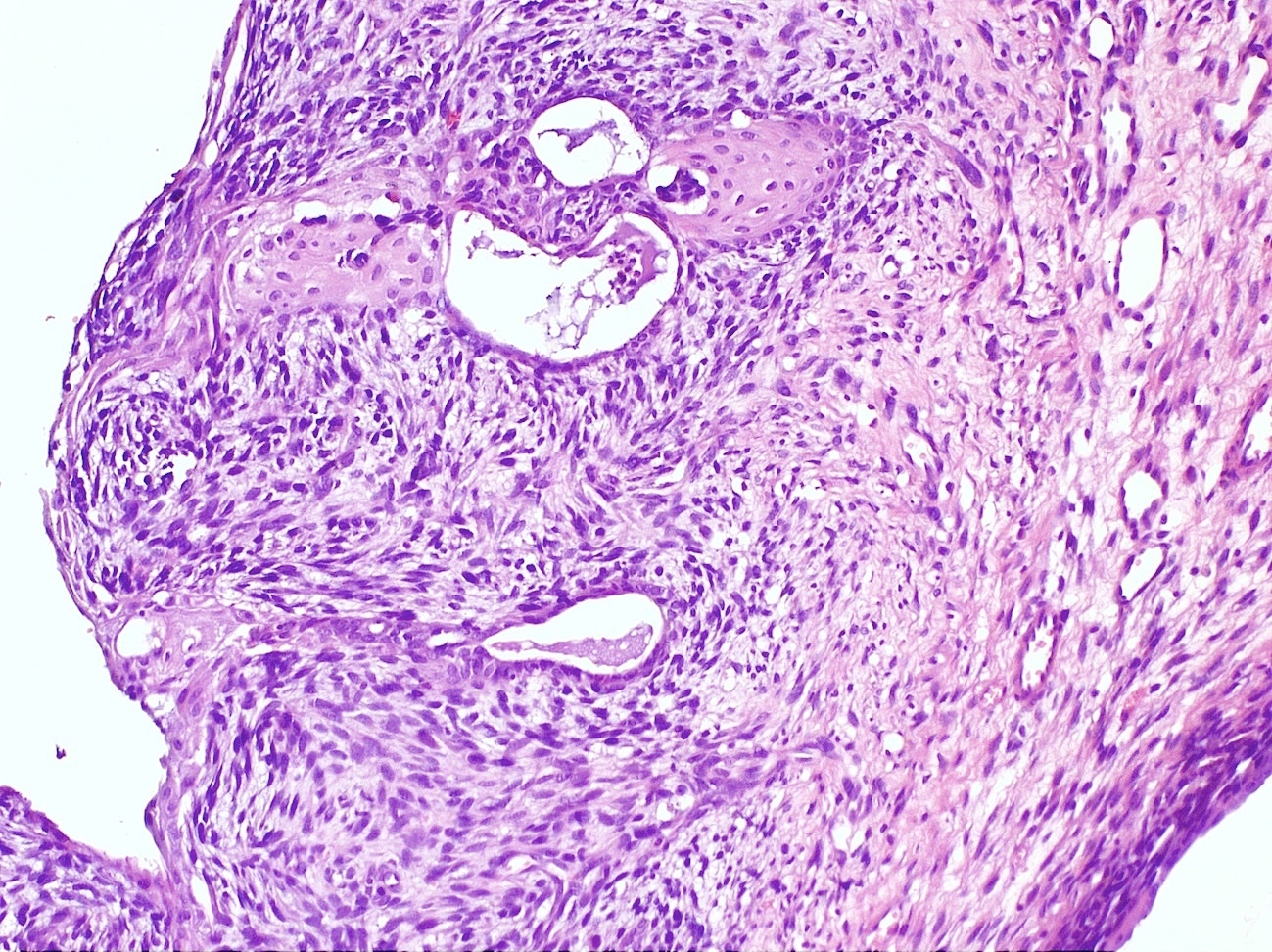
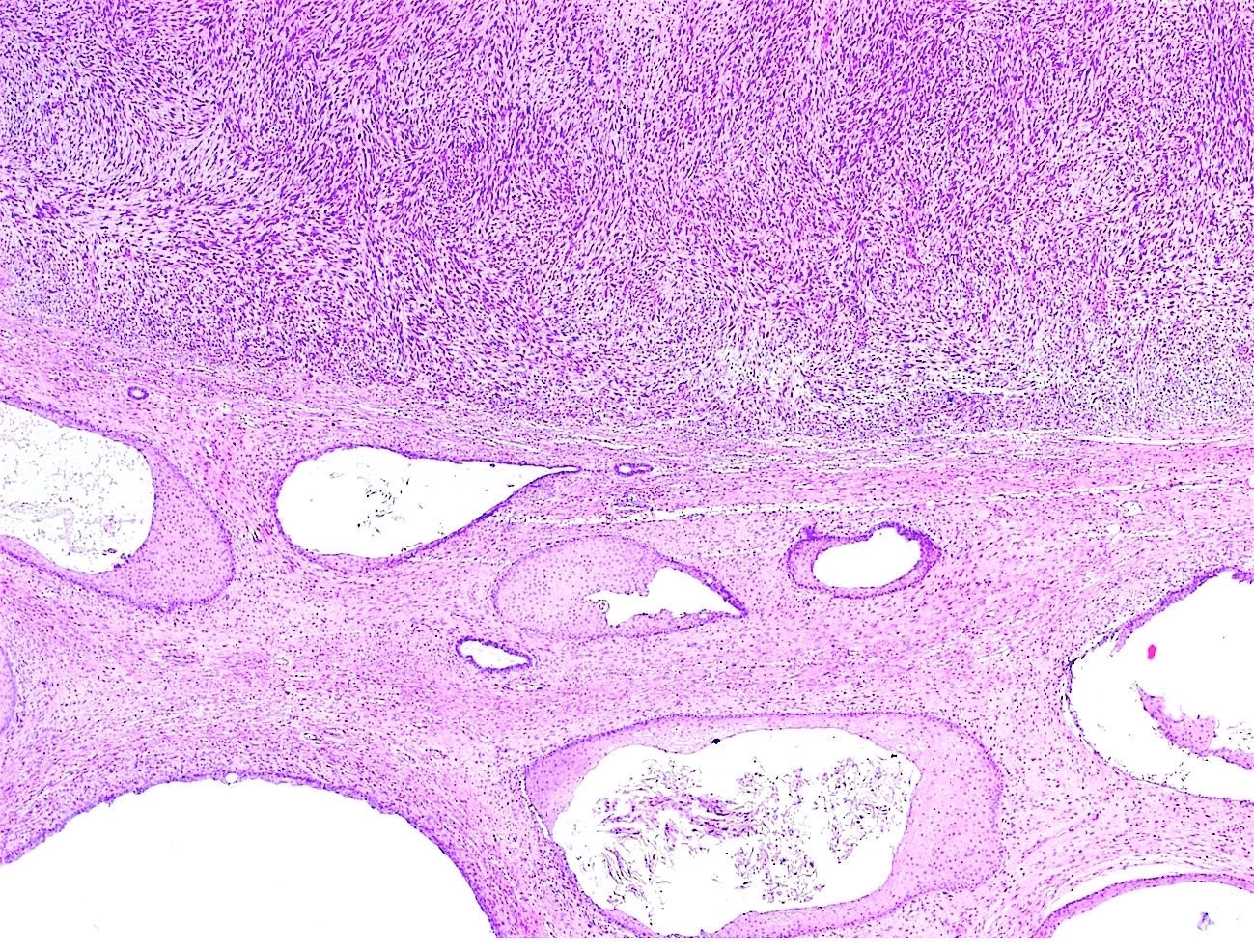
Microscopic (histologic) description
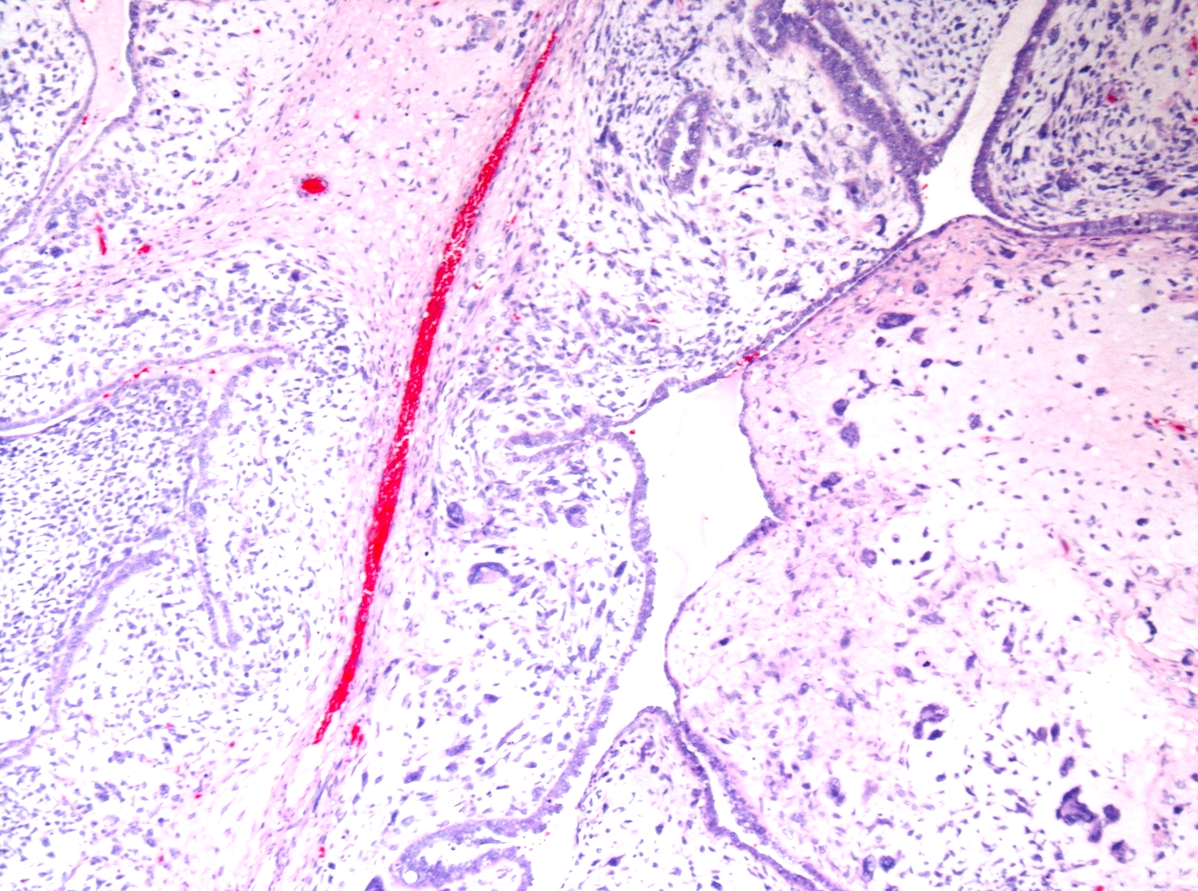
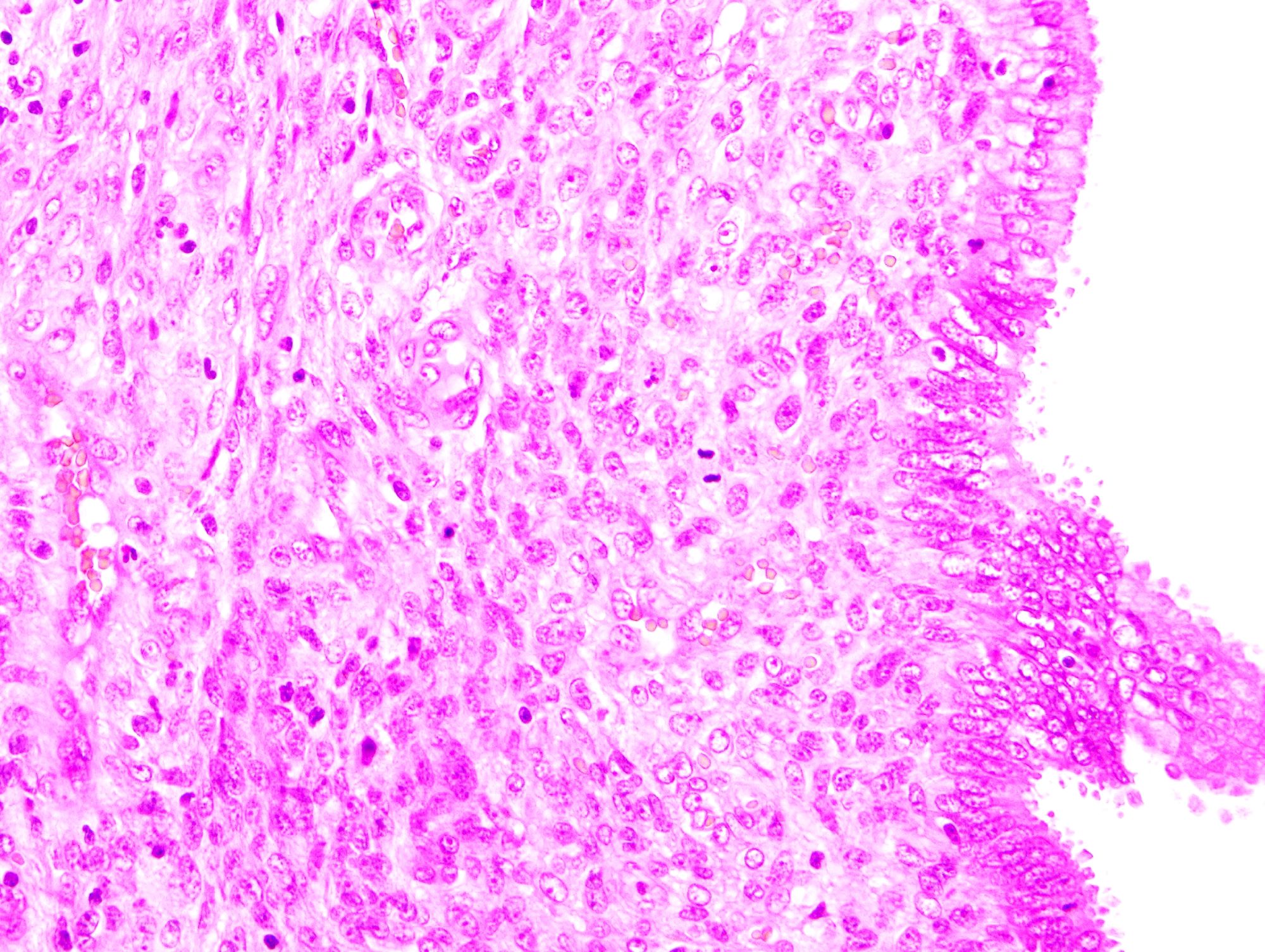
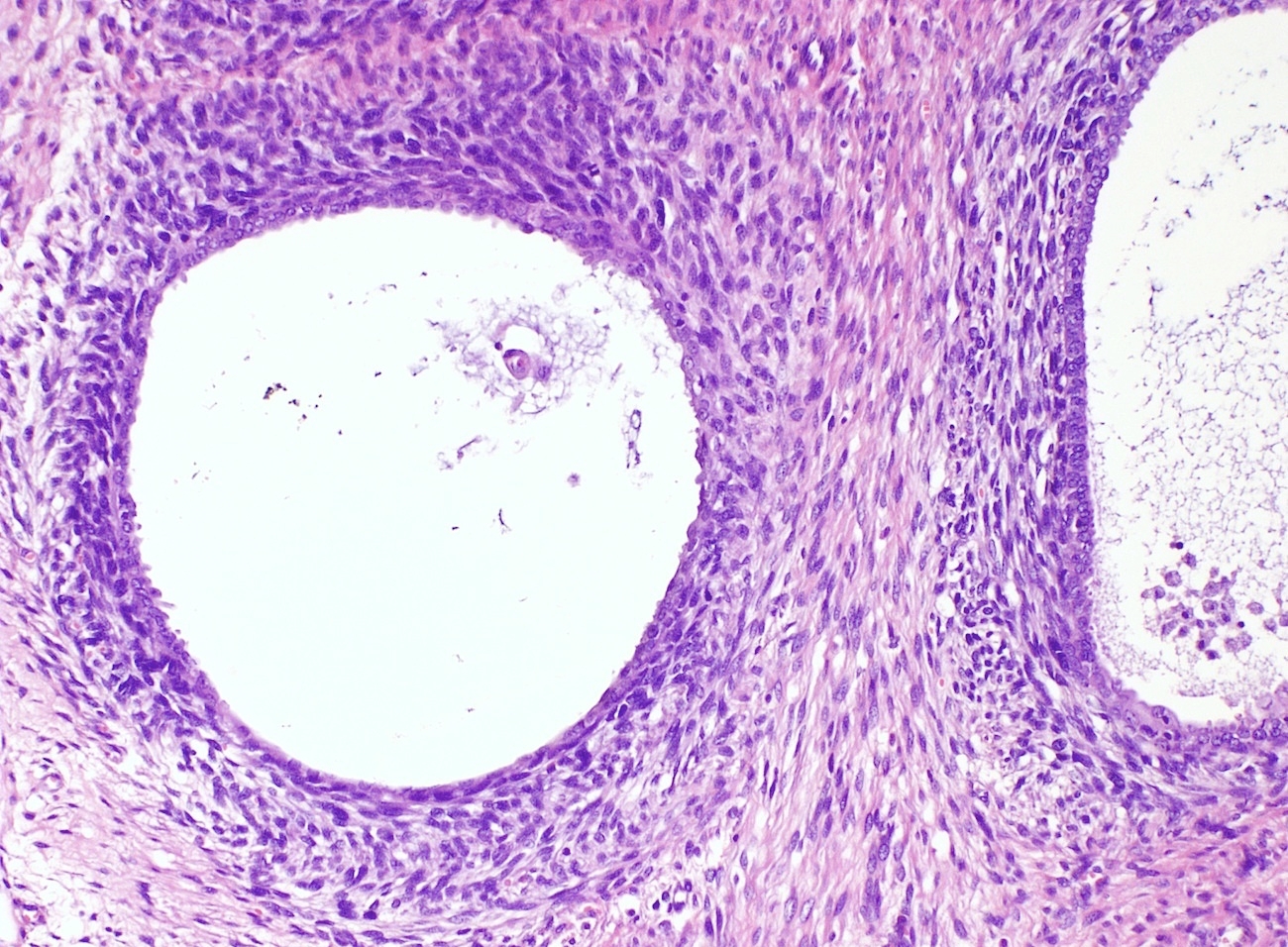
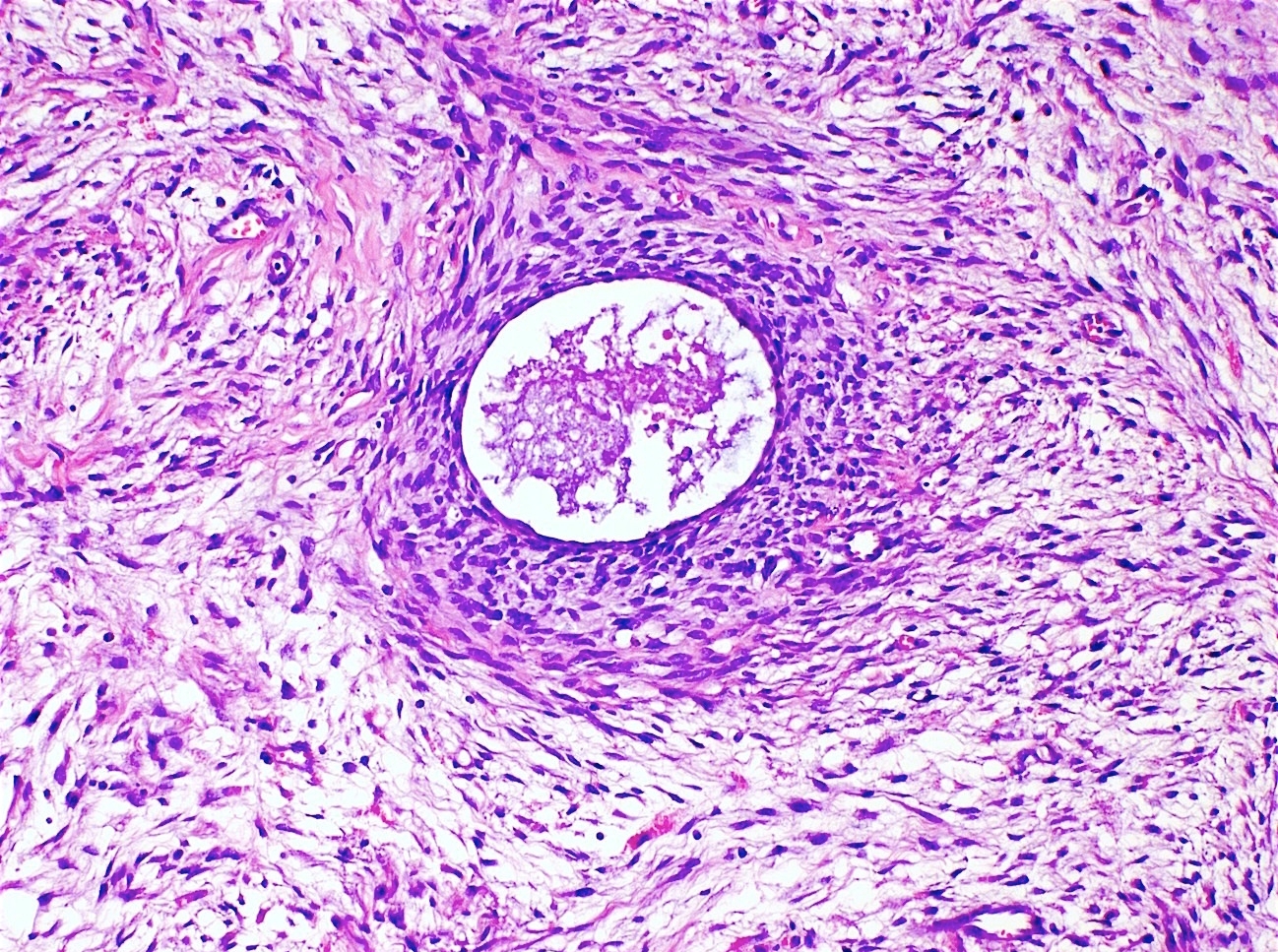
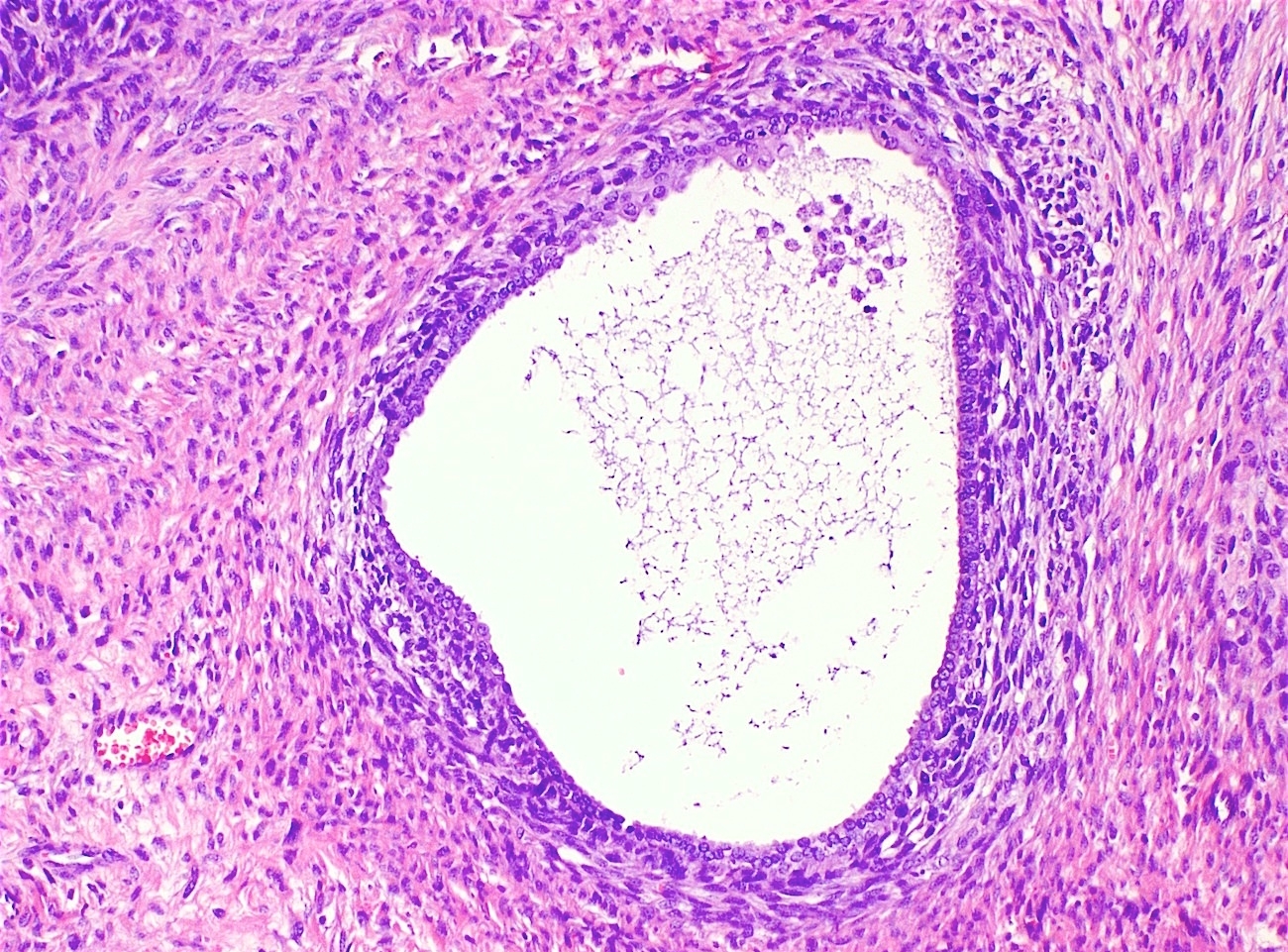
- Biphasic (malignant stromal and benign glandular components)
- Glandular component is bland and evenly dispersed
- Epithelial metaplasia can be appreciated but atypia or frank malignant features are absent
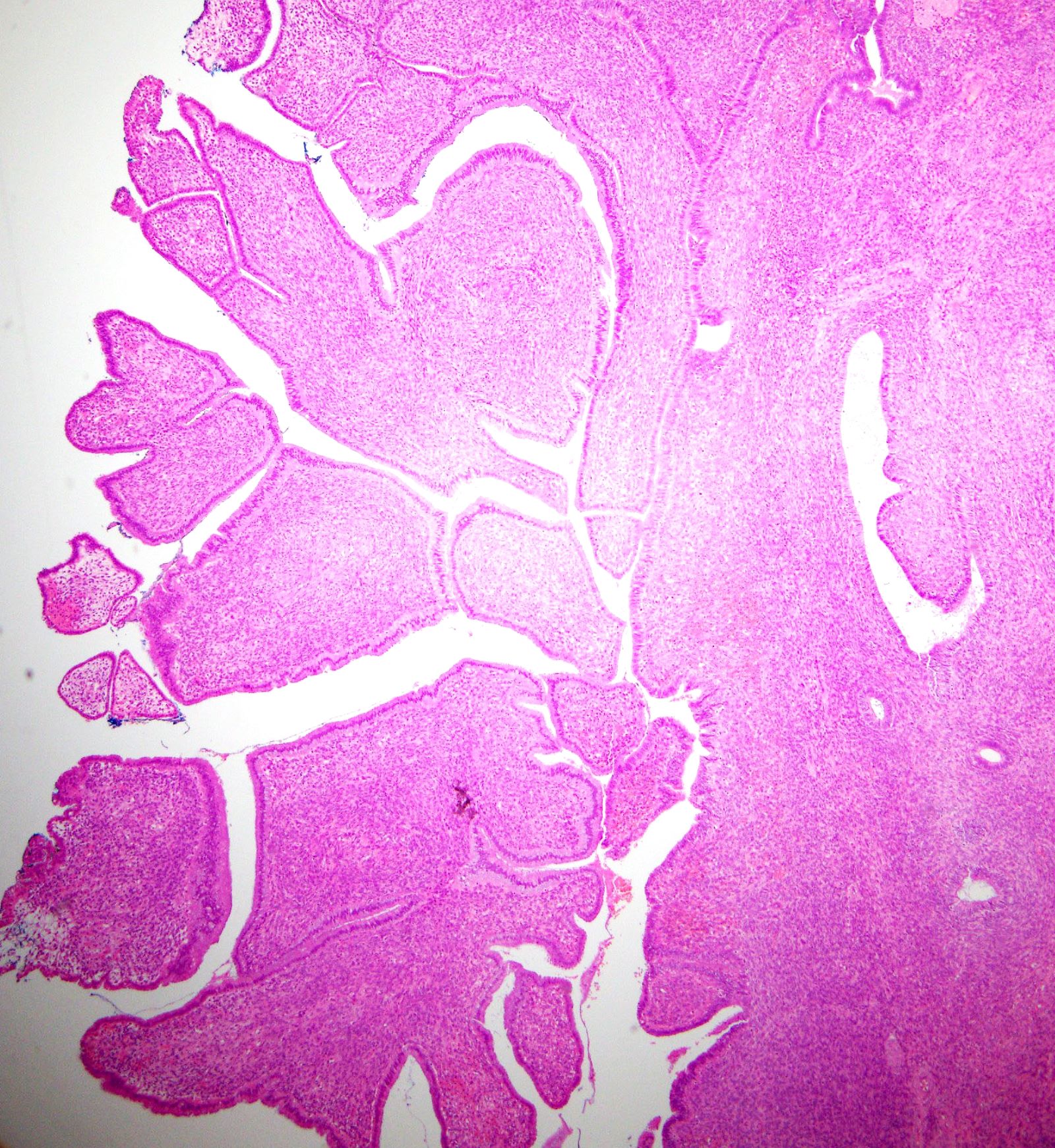
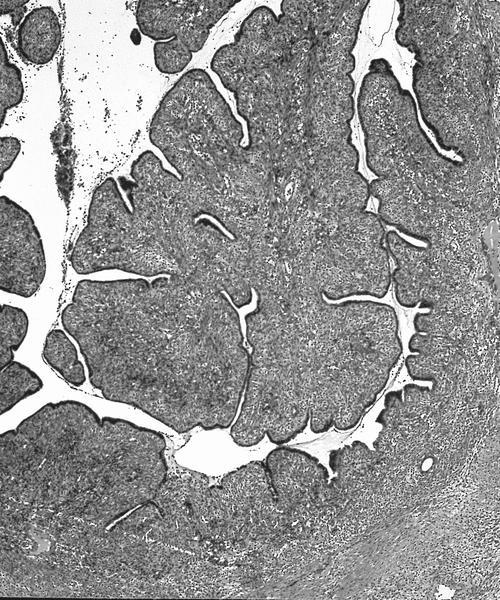
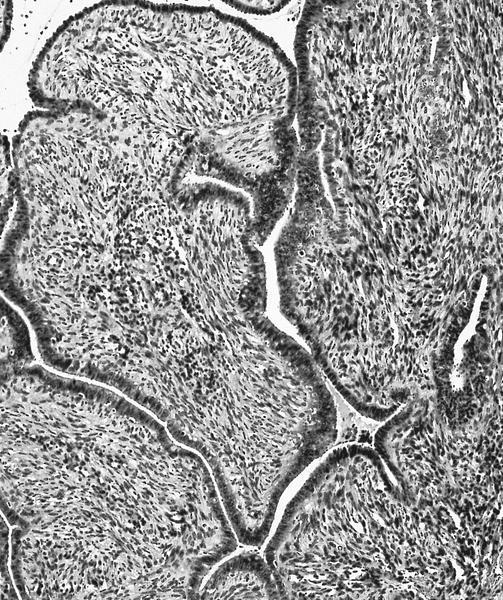
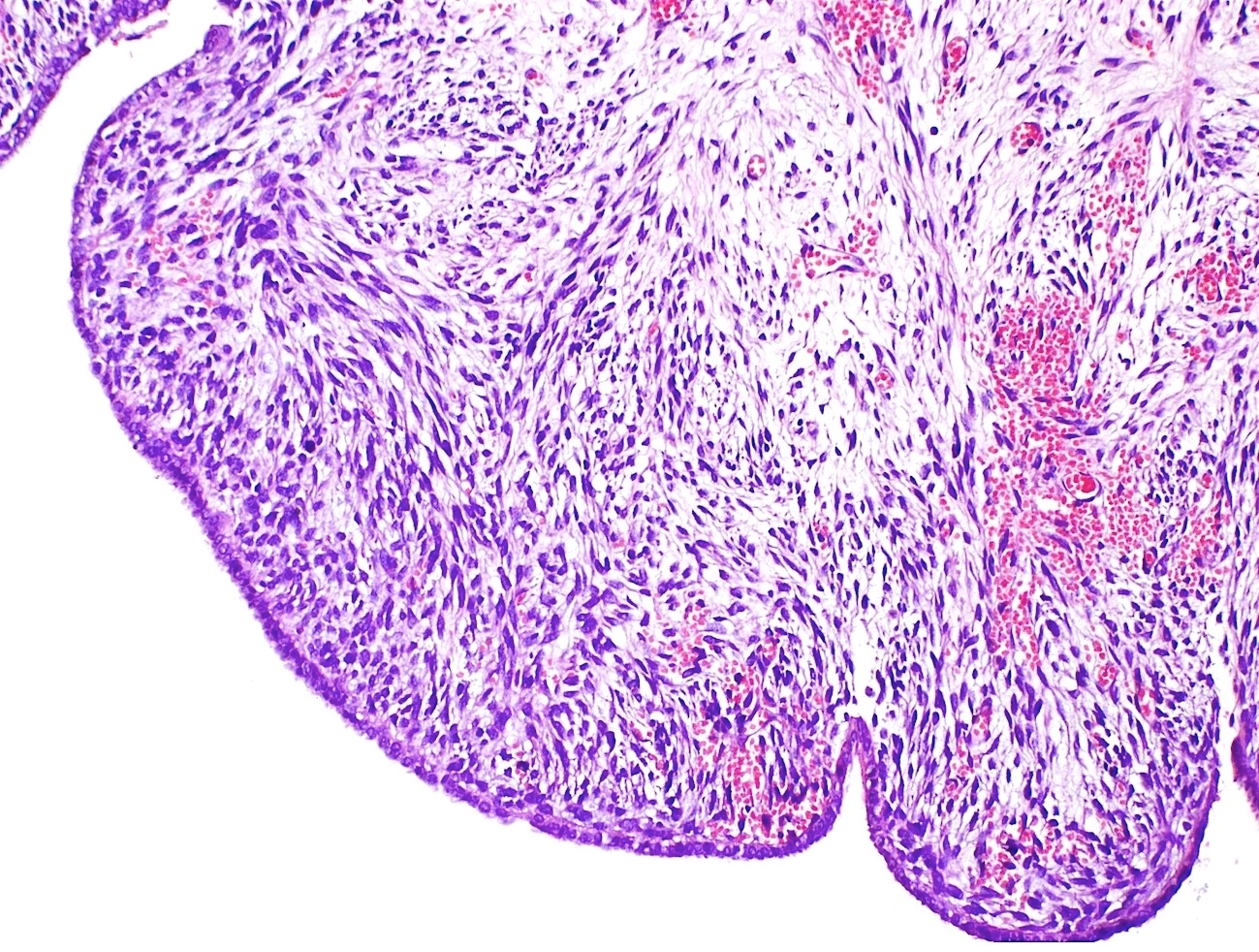
- Most glands have narrow lumens, usually compressed by the underlying mesenchymal growth giving a leaf-like appearance
- Cystic dilation with rigid contours is common
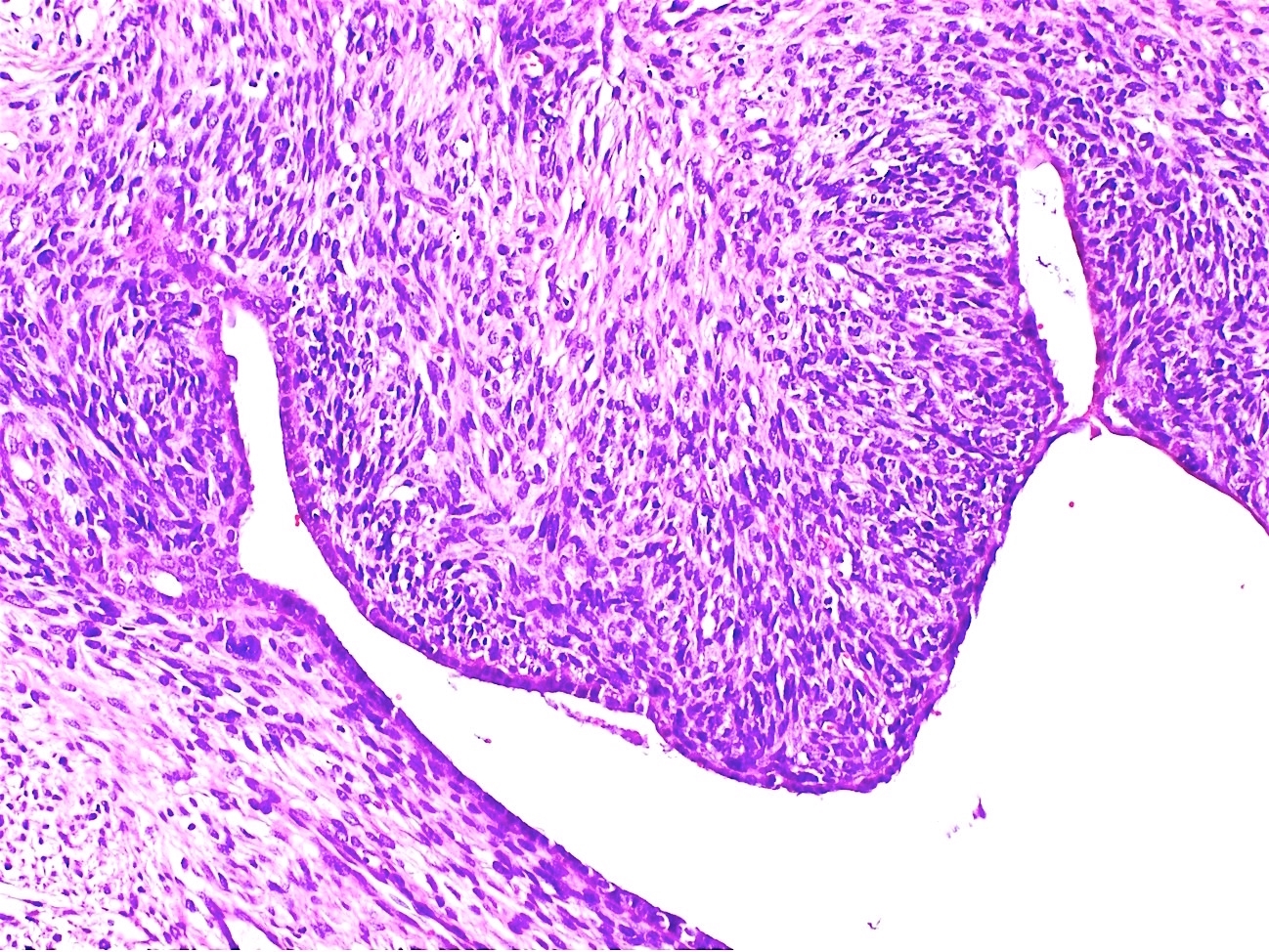
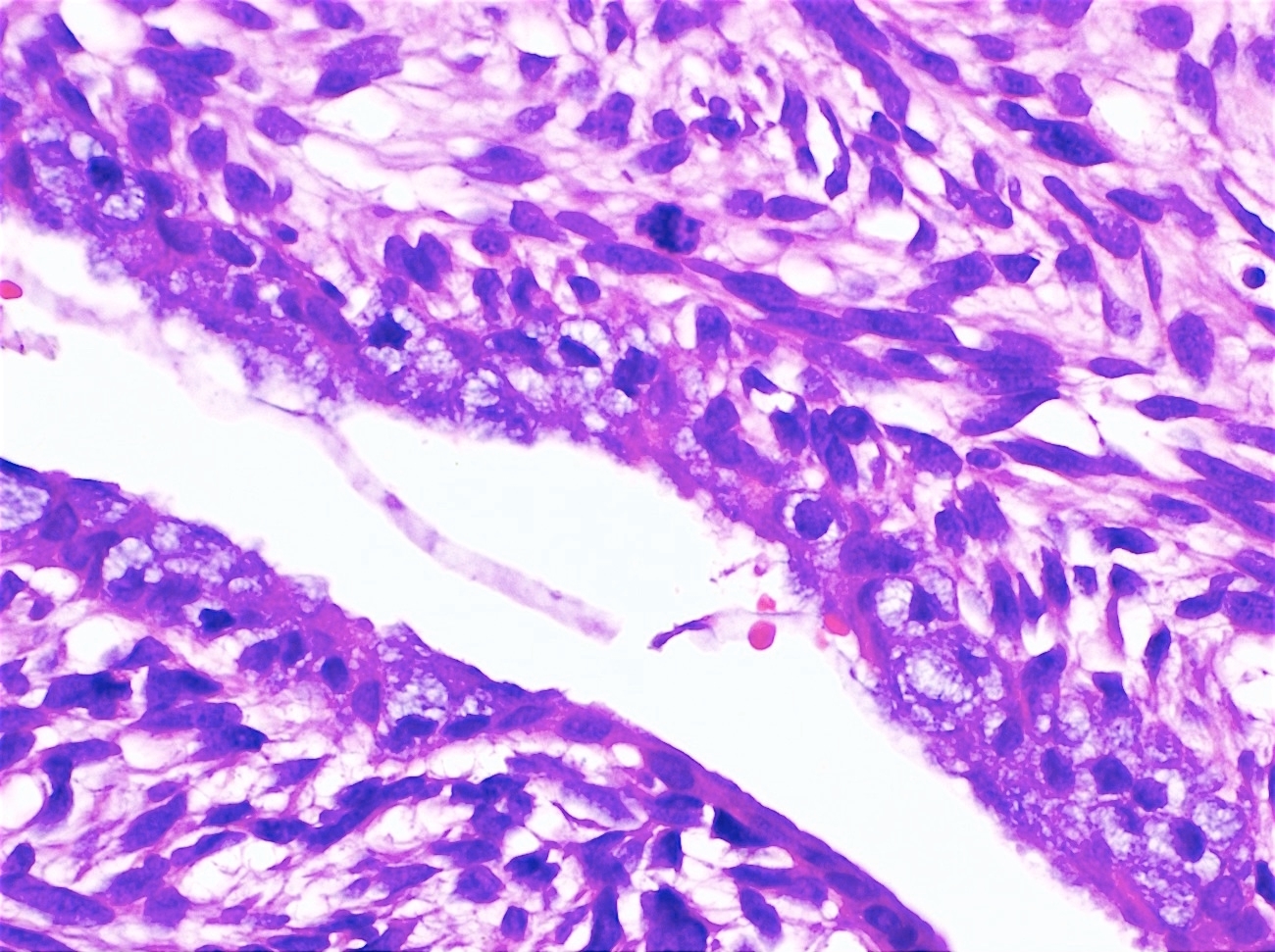
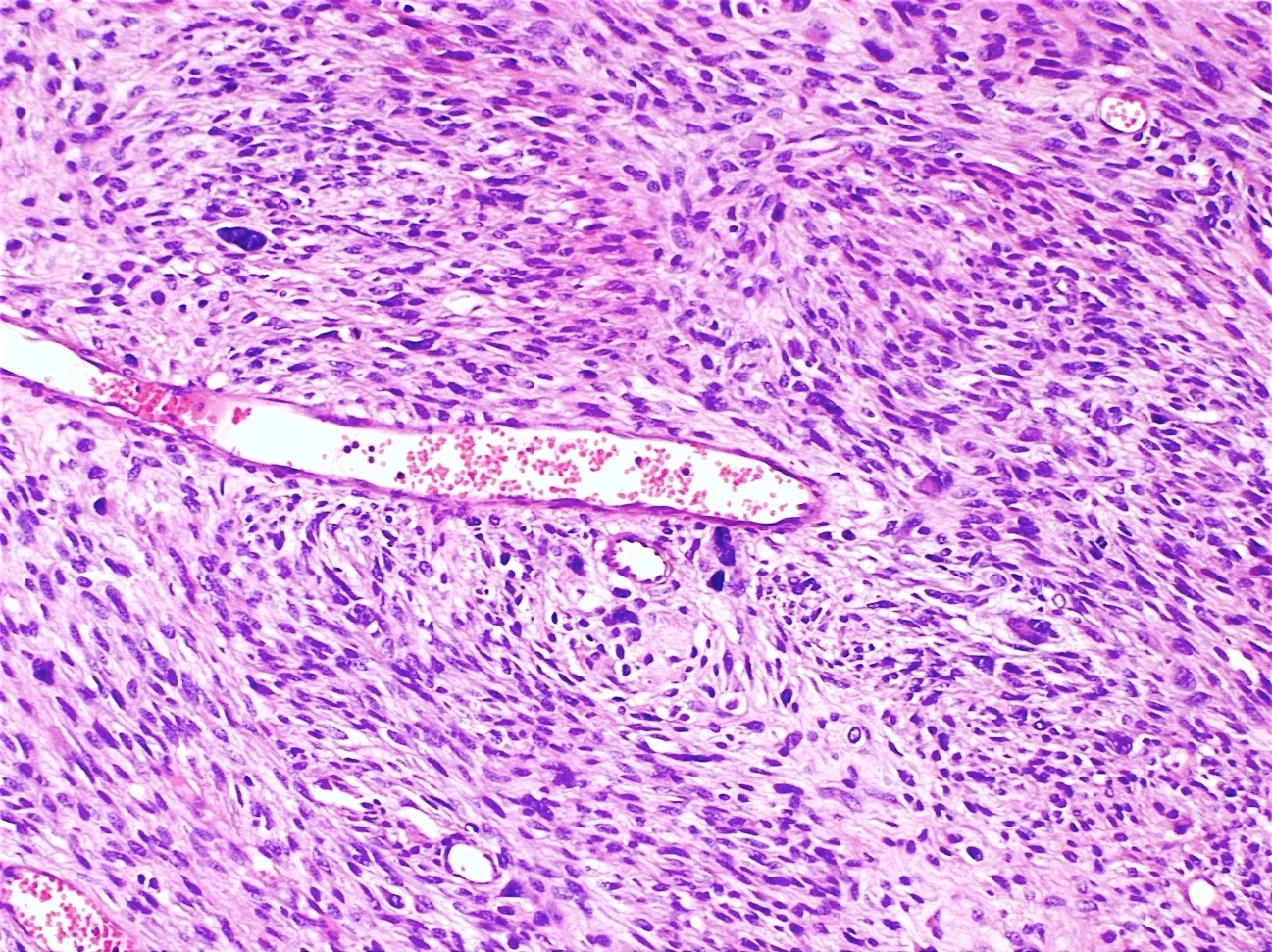
- Periglandular cuffing:
- Stroma around the glands is usually more cellular and atypical; in these cellular areas, mitotic activity is increased, usually ≥ 4 mitoses/10 high power fields
- Stroma in this region is sometimes referred to as the cambium layer
- Diagnosis of adenosarcoma relies on the identification of the following features:
- Intraglandular projections and leaf-like (phyllodes-like) architecture
- Marked stromal cytologic atypia
- Periglandular stromal condensation (cuffing)
- Rigid cystic dilation
- Mitotic activity ≥ 2 mitoses/10 high power fields
- Diagnosis of adenosarcoma is favored if ≥ 2 of the above features are diffusely present
- Uterine polyps that are morphologically worrisome for (but not diagnostic of) Müllerian adenosarcoma have recently been shown to follow a benign clinical course, requiring only conservative management (Mod Pathol 2022;35:106)
- Tumors with up to 3 of the above changes, when focal, fall under this category
- The term "atypical uterine polyp" has been proposed for such cases
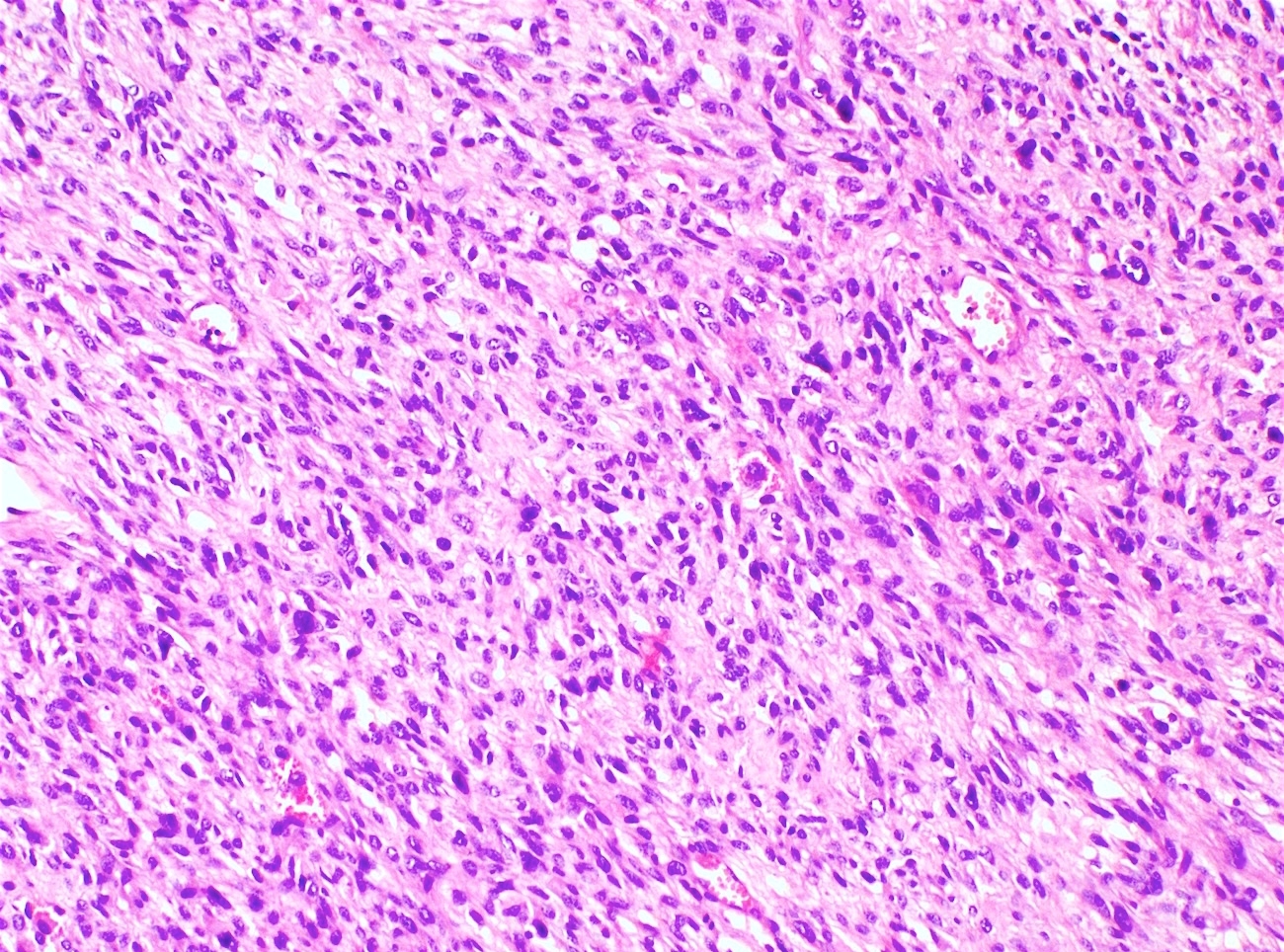
- High grade sarcoma is defined as pleomorphic sarcoma cells that are identifiable at low power magnification; nuclei are enlarged, hyperchromatic and contain prominent nucleoli
- Rhabdomyosarcomatous differentiation is frequent
- Sarcomatous component may have sex cord differentiation (Int J Gynecol Pathol 2016;35:153)
- Adenosarcoma with sarcomatous overgrowth:
- Stromal overgrowth is defined as pure sarcoma representing ≥ 25% of the tumor
- Sarcoma can be homologous or heterologous and frequently displays high grade cytologic features
- Aggressive variant (Am J Surg Pathol 1989;13:28)
- Seen in approximately 10% of cases
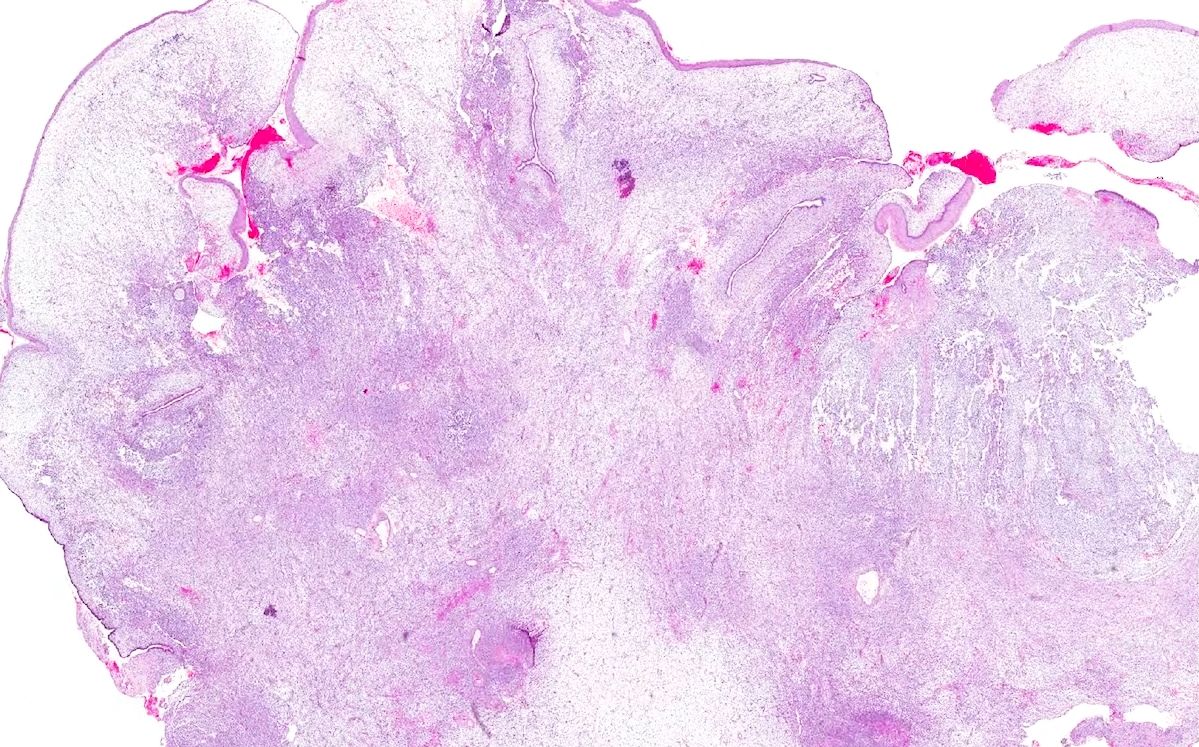
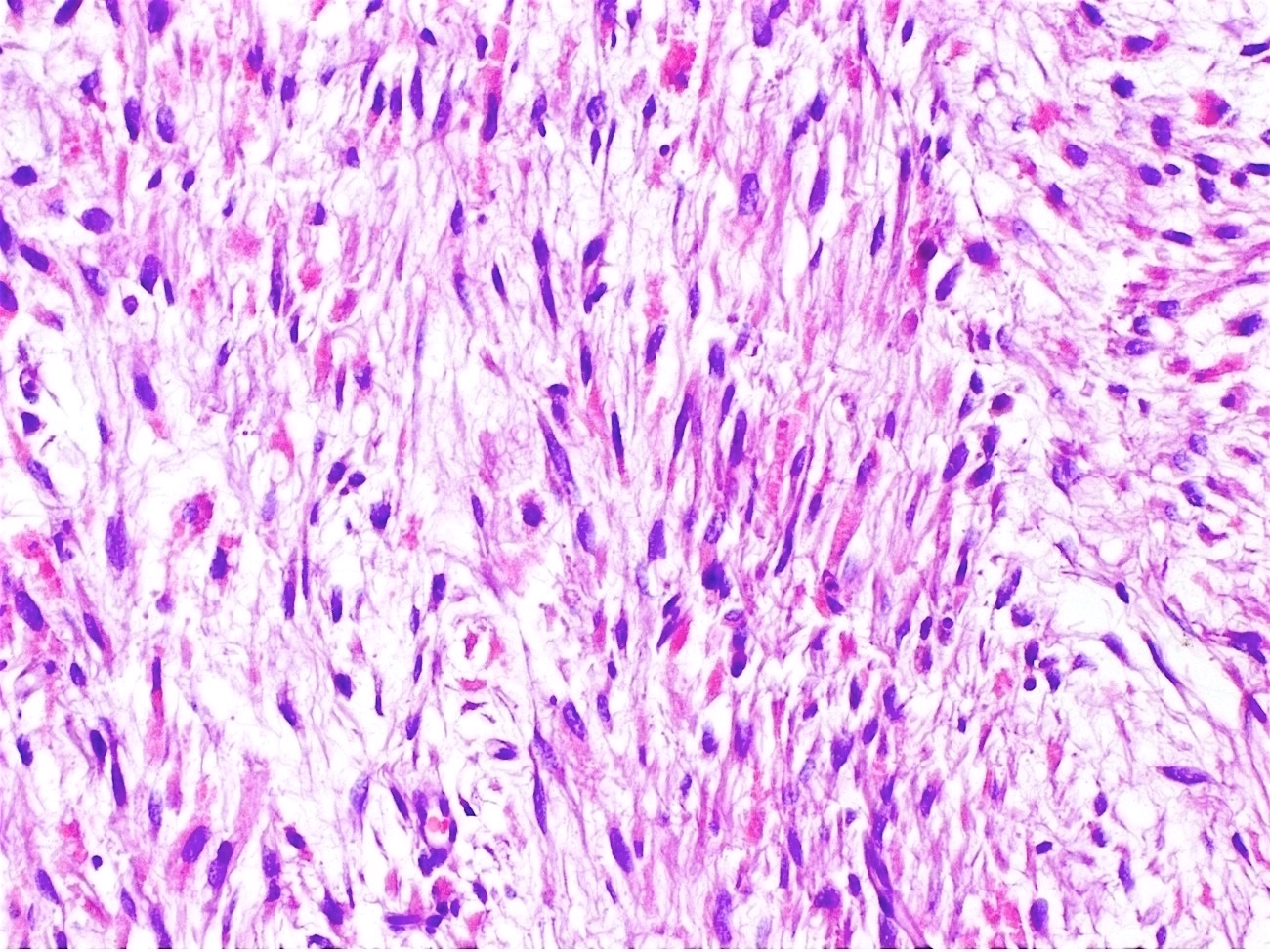
Microscopic (histologic) images
Contributed by Carlos Parra-Herran, M.D. and AFIP images
Contributed by Ayse Ayhan, M.D., Ph.D.
Positive stains
- CD10, WT1, muscle specific actin, desmin, ER, PR (Gynecol Oncol 2004;93:680, Int J Gynecol Cancer 2004;14:1118)
- High grade adenosarcoma and adenosarcoma with sarcomatous overgrowth frequently show abnormal p53 staining (overexpressed or null)
Negative stains
- Low grade adenosarcoma has wild-type p53 expression (Am J Surg Pathol 2009;33:278)
- Adenosarcoma with sarcomatous overgrowth may show loss of CD10 and PR
Electron microscopy description
- Stromal cells resemble endometrial stromal cells
Molecular / cytogenetics description
- Müllerian adenosarcoma harbors a number of somatic gene alterations that are exclusive to the mesenchymal component; this supports the hypothesis that this lesion is primarily a mesenchymal neoplasm (J Pathol 2016;238:381)
- Amplification of MDM2 and CDK4 is seen in approximately 25% of cases
- Adenosarcomas with sarcomatous overgrowth have a higher number of copy number variations, MYBL1 amplification, ATRX mutations, global chromosomal instability and chromothripsis (up to thousands of clustered chromosomal rearrangements occur in a single event in localized and confined genomic regions in one or a few chromosomes) (Mod Pathol 2016;29:1070, J Pathol 2015;235:37, Am J Surg Pathol 2017;41:1513)
Sample pathology report
- Uterus, total hysterectomy:
- Müllerian adenosarcoma, high grade, with sarcomatous overgrowth and heterologous rhabdomyoblastic differentiation (3.1 cm); lesion involves cervix and lower uterine segment
- Myometrial / cervical stromal invasion is present (> 50% of the wall)
- Lymphovascular invasion is not identified
- Margins are negative
- AJCC stage pT1c Nx Mx (FIGO stage Ic)
- Cervix, polyp, polypectomy:
- Müllerian adenosarcoma, low grade (2.5 cm) (see comment)
- Comment: Tumor cells are positive for ER and PR (strong staining in > 90% of cells) as well as CD10.
Differential diagnosis
- Adenofibroma:
- Benign glands within fibrotic stroma
- < 2 mitoses/10 high power fields
- Less stromal cellularity without periglandular cuffing or atypia
- This entity is no longer recognized by the WHO; there is growing consensus that this lesion does not exist in the uterus
- Carcinosarcoma:
- Epithelial component is malignant (benign in adenosarcoma) (Am J Surg Pathol 2021;45:374)
- Endocervical / endometrial polyp:
- Glands lack leaf-like architecture or rigid cystic dilation
- Lack of periglandular stromal condensation
- Lack of stromal atypia
- Endometrial stromal sarcoma:
- Absence of epithelial elements
- Normal epithelial elements can be entrapped by the mesenchymal proliferation, mimicking adenosarcoma; however, this usually happens only at the periphery of the lesion and on the endometrial surface; moreover, leaf-like growth and periglandular condensation are absent
- Rhabdomyosarcoma:
- Differential in cases of high grade adenosarcoma with heterologous differentiation
- Pure rhabdomyosarcoma lacks benign epithelial elements admixed within the tumor
Additional references
Practice question #1
A 32 year old woman presents with abnormal vaginal bleeding and is found to have a 4.5 cm polypoid lesion protruding from the cervical os. Histologic evaluation shows that the lesion arises from the cervix and has bland epithelium with leaf-like architecture. There is periglandular cuffing by markedly atypical stromal cells with a mitotic index of 8 per 10 high power fields. The stromal component comprises 75% of the lesion. Which of the following features defines this as a high grade sarcoma?
- ≥ 25% of the lesion is the stromal component
- Mitotic index > 2 per 10 high power fields
- Periglandular cuffing by stromal cells
- Pleomorphic tumor cells visible at low power
Practice answer #1
D. Pleomorphic tumor cells visible at low power categorize this lesion as a high grade adenosarcoma, which is associated with metastasis, recurrence and overall poor prognosis. The presence of sarcomatous overgrowth (≤ 25% stromal component) is frequently associated with high grade cytologic features.
Comment Here
Reference: Cervix - Adenosarcoma
Comment Here
Reference: Cervix - Adenosarcoma