Table of Contents
Definition / general | Epidemiology | Sites | Etiology | Pathophysiology | Causative organisms | Classification | Clinical features | Diagnosis | Diagrams / tables | Investigation | Case reports | Treatment | Clinical images | Gross description | Gross images | Microscopic (histologic) description | Microscopic (histologic) images | Histochemical stains | Immunohistochemistry & special stains | Molecular / cytogenetics description | Complications and their mechanism | Additional referencesCite this page: Amita R. Infective endocarditis. PathologyOutlines.com website. https://www.pathologyoutlines.com/topic/heartnontumorinfecendocarditis.html. Accessed September 9th, 2025.
Definition / general
- First described by William Osler in 1885
- Defined as an infection on endothelial surfaces of heart which includes:
- Mural endocardium
- Valves and their apparatuses
- Prosthetic or implanted components, including valves, homografts, pacemakers, intracardiac cardioverter defibrillators and their leads
- Ventricular assist devices
- Surgical shunts and septal defect occluders
- Endovascular devices or clips
Epidemiology
- Population based incidence is 4 -10 per 100,000 per year, slightly higher rate in men
- Mortality estimated at 1 per 100,000 per year, greater in intravenous drug abuse (IVDA)
- Endocarditis accounts for ~0.75 admissions per 1000 per year in large community hospitals (eMedicine: Pathology of Infectious Endocarditis [Accessed February 28, 2018])
Sites
- Endocardial surface of valve, endocardium of cardiac chamber
- In the presence of a ventricular septal defect (VSD), vegetations can be found on the right ventricular side of the VSD, on the tricuspid valve or where the jet impinges on the right ventricular wall
- Vegetations found in coarctation usually occur distal to the obstruction
Etiology
- Risk factors:
- Pre existing structural heart disease: rheumatic heart disease (RHD), hypertrophic cardiomyopathy (HCM), subaortic stenosis, ventricular aneurysm
- Degenerative valve disease: mitral valve prolapse, bicuspid aotic valve, calcific aortic valve disease
- Cardiac interventions: prosthetic valve, pacemaker, defibrillator, IVDA (right sided endocarditis), indwelling catheter
- Congenital heart disease: small ventricular septal defect, bicuspid aortic valve, coarctation of aorta, patent ductus arteriosus
- Uncommon with large VSD, mitral stenosis, atrial septal defect
Pathophysiology
- Experimental evidence indicates that 2 factors are needed to establish endocardial infection:
- Endothelial denudation over valve cusp with platelet deposition (thrombus)
- Episode of bacteremia
- Endothelial damage can be due to aberrant jet streams and turbulent flow in the setting of diseased cardiac valves and septal defects or direct trauma from intravascular devices
- A platelet / fibrin clot is formed over the exposed underlying extracellular matrix as the reparative process ensues
- If transient bacteremia occurs before a protective layer of endothelium forms, colonization of the fibrin may develop and progress to an infected vegetation
- Organism proliferates within the thrombus, forming infective vegetations, which elicits inflammatory response resulting in erosion or perforation of the valve cusps, leading to valvular aneurysms, perforations and incompetence, damage to the conduction pathway (if in the septal area), or rupture of a sinus of Valsalva (if in the aortic area)
- Prolonged antibiotic treatment is needed to sterilize the vegetations
- Endocarditis on a prosthetic valve usually begins in the valve ring and may protrude into the valve orifice causing obstruction and regurgitation
- Annular abscesses around the sewing ring of the valve are common, leading to dehiscence of sutures and paraprosthetic leaks
- Organisms which can bind via receptors to fibronectin, other subendothelial components or to platelets are the most virulent
- Healing process depends on the degree of valve damage in the initial acute phase
- Over time, vegetation decreases in size and organizes as a fibrous nodule which may calcify
- Cusp fibrosis leads to cusp thickening and retraction
Causative organisms
- Streptococci:
- 50% - 80% of infective endocarditis (IE) cases
- S. viridans (e.g. S. anguis, S. milleri, S. mutans, S. mitior), normally found in the upper aerodigestive tract, may disseminate during tonsillectomy, dental extraction, dental cleaning etc. resulting in bacteremia
- Staphylococci:
- 20% - 30% of subacute cases of IE and 50% of acute cases
- S. aureus and S. epidermidis
- Common in patients with indwelling central venous catheters, IVDA, diabetes, chronic hemodialysis and prosthetic valve endocarditis
- Staphylococcal endocarditis can affect functionally normal native valves and cause extensive tissue damage and septic emboli
- Prosthetic valve endocardits is mainly due to coagulase negative staphylococci
- Enterococci: 5% - 15% of IE cases; e.g.: S. faecalis, S. bovis, S. faecium
- HACEK organisms: Haemophilus parainfluenza, Haemophilus aphrophilus, Actinobacillus (Haemophilus) actinomycetemcomitans, Cardiobacterium hominis, Eikenella species, and Kingella species
- Less common organisms: Candida, Aspergillus, Histoplasma, and Brucella (common in IVDA, alcoholics, and patients with prosthetic heart valves)
Classification
- Older classification was based on course of untreated disease: acute, subacute, chronic
- Newer classification system is based on causative organism
- Also classify based on native valve or prosthetic valve endocarditis
Clinical features
- Vague constitutional symptoms such as fever, rigors, night sweats, anorexia, weight loss, arthralgia
- Development of new murmur or change in nature of an existing murmur (a regurgitant murmur may disappear or worsen)
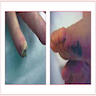
- Skin lesions
- Osler nodes: tender lesions found on finger pulps and thenar / hypothenar eminences
- Janeway lesions: transient, nontender macular papules on palms or soles
- Splinter hemorrhages
- Petechiae (embolic or vasculitic)
- Clubbing: in long standing disease
- Eyes: Roth spots (boat shaped hemorrhages with pale centers, in retina) and conjunctival splinter hemorrhages
- Neurological:
- Confusion, hemiplegia and sensory dysfunction due to emboli
- Subarachnoid hematoma due to rupture of mycotic aneurysm
- Renal:
- Infarction causes loin pain and hematuria
- Immune complex deposition may cause glomerulonephritis
- Splenomegaly and splenic infarction due to emboli
Diagnosis
- Requires (a) 2 major criteria or (b) 1 major and 2 minor criteria or (c) 5 minor criteria (see Table below)
- These are associated with a 99% specificity for diagnosis in follow up studies
Investigation
- Blood count: normochromic normocytic anemia is usual, while neutrophil leucocytosis is common
- ESR: may be raised
- Renal and liver function test: levels of creatinine may be raised; levels of liver enzymes may be raised in a hepatocellular (nonobstructive) pattern
- CRP: increases acutely in bacterial infection
- Urine microscopy: microscopic hematuria is common in early disease
- Culture: all skin lesions, drip sites or other foci of infection should be cultured
- Electrocardiography (ECG): ECG regularly (daily if aortic or septal root abscess is suspected)
- Culture negative endocarditis:
- In up to 10% of cases of IE diagnosed by clinical criteria, blood cultures may be negative
- This is due to either subtherapeutic antibiotic therapy or an organism whose growth requirements make in vitro growth difficult
- Gram negative organisms of the HAECK group, Brucella, Fungi, Coxiella, Chlamydia are more likely in these cases
- In these cases, diagnosis is best obtained by serology or broad range PCR
Case reports
- 67 year old man with fungal endocarditis of the mitral valve (Clin Cardiol 2000;23:55)
- Fungal endocarditis: an autopsy study (Asian Cardiovasc Thorac Ann 2004;12:95)
- Cardiac valves in patients with Q fever endocarditis (J Infect Dis 2003;187:1097)
Treatment
- Blind treatment with antibiotics should not be undertaken since it will delay diagnosis and identification of the causal organism
- Antibiotics should not be initiated before three sets of blood cultures have been taken
- Current guidelines on antibiotic prophylaxis:
- American Heart Association (2007): changes past recommendation; now, no antibiotic prophylaxis after GI or GU procedures in group A patients (prosthesis, congenital heart disease, previous history of IE) (AHA: Prevention of Infective Endocarditis [Accessed 28 February 2018], Prophylaxis Against Infective Endocarditis, London: National Institute for Health and Clinical Excellence (UK) 2008 )
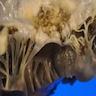
Gross description
- Vegetations are seen attached to atrial aspect of AV valves and ventricular aspect of semilunar valves in relation to the line of apposition
- Large vegetations may extend into adjacent parts of cusp or leaflets, chordae tendinae or sinus of valsalva
- Largest vegetation is seen in fungal infections
- Colour varies from red to pink to yellow and may be soft or friable or firm
- Surface is smooth, often irregular granular
- Lesions may calcify with time after antibiotic treatment
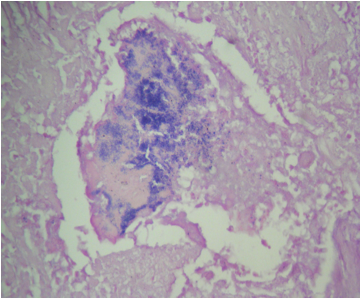
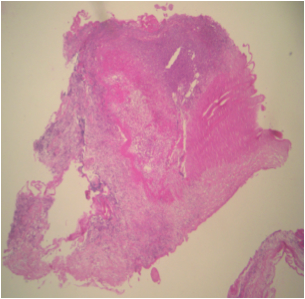
Microscopic (histologic) description
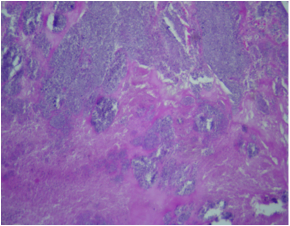
- Two features always present are inflammation and surface thrombus
- Depending on the duration of the infection and treatment, the inflammation may be primarily neutrophilic or mostly lymphohistiocytic
- Fresh vegetations contain platelets and fibrin with a rich infiltrate of neutrophils
- Colonies of embedded organisms may be seen
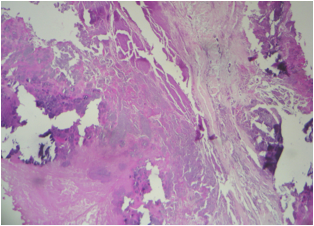
- Organizing and organized thrombi with fibrosis are common in lesions that have been present for weeks or more
- In chronic lesions, vegetations may show varying degrees of organization, vascularization and calcification
- Care must be taken to distinguish between irregularly sized granules of calcium and viable bacteria
- Numerous giant cells with lymphocytes are a feature of Coxiella burnetti (Q fever) endocarditis
Microscopic (histologic) images
Histochemical stains
- Gram stain, Methanemine silver stain (GMS), PAS
- Combination of these identifies fungal spores and hyphae and may show cocci with altered staining characteristics
- Giemsa and Machiavello stains: for suspected Rickettsial infections
Immunohistochemistry & special stains
- Useful in culture negative endocarditis, due to fastidious microbes like Coxiella and Legionella
Molecular / cytogenetics description
- Polymerase chain reaction (PCR) to detect fastidious bacteria in culture negative cases; specific organisms that have been tested in this way include Rickettsia (Coxiella, Bartonella, T whipplei)
Complications and their mechanism
- Cardiac failure: volume overload due to regurgitant lesions, infarcts
- Systemic infarcts: emboli
- Systemic abscess: infected emboli
- Aneurysms: infected emboli
- Renal failure: emboli, immune mediated glomerulonephritis