Table of Contents
Definition / general | Essential features | Terminology | ICD coding | Epidemiology | Sites | Pathophysiology | Diagrams / tables | Clinical features | Diagnosis | Laboratory | Radiology description | Prognostic factors | Case reports | Treatment | Gross description | Gross images | Microscopic (histologic) description | Banff scoring of histological lesions | Microscopic (histologic) images | Immunofluorescence description | Positive stains (IHC and special stains) | Electron microscopy description | Molecular / cytogenetics description | Sample pathology report | Differential diagnosis | Additional references | Board review style question #1 | Board review style answer #1 | Board review style question #2 | Board review style answer #2Cite this page: Sağlam A. Active / chronic active antibody mediated rejection. PathologyOutlines.com website. https://www.pathologyoutlines.com/topic/kidneyacutechronichumoralrejection.html. Accessed April 19th, 2024.
Definition / general
- Alloimmune reaction to donor specific antigens resulting in damage to the kidney allograft
- Mediated by antibodies produced by B cells and hence referred to as antibody mediated rejection (ABMR) in the Banff 2019 classification (Am J Transplant 2020;20:2318)
- Has 3 major subcategories:
- Active antibody mediated rejection: characterized by an acute immunologic reaction
- Chronic active antibody mediated rejection: chronic renal injury due to persistent / recurrent ABMR
- Chronic (inactive) ABMR: chronic renal injury due to prior active / chronic active ABMR
Essential features
- Evidence of microvascular / endothelial damage
- Peritubular capillaritis, glomerulitis, microthrombosis and acute tubular injury (active component)
- Transplant glomerulopathy and multilayering of the tubular basement membrane (signs of chronicity)
- C4d positivity (a complement degradation product) along peritubular capillaries
- Intimal arteritis or vasculitis; this lesion is not limited to antibody mediated rejection (ABMR) but may also be indicative of acute T cell mediated rejection as well as mixed T cell mediated rejection and ABMR
- Expression of endothelium associated transcripts (ENDAT)
- Coded by the g, cg, ptc, v, cv and C4d Banff scores
Terminology
- Active antibody mediated rejection: also referred to as acute humoral rejection
- Chronic active antibody mediated rejection: also referred to as chronic humoral rejection
- Chronic antibody mediated rejection: also referred to as chronic humoral rejection
ICD coding
- ICD-10: T86.11 - kidney transplant rejection
Epidemiology
- Antibody mediated rejection has been reported to occur in about 5 - 10% of transplant patients (J Transplant 2012;2012:193724)
- Can be as high as 50% in patients with human leukocyte antigen (HLA) incompatible transplant
Sites
- Renal disease
Pathophysiology
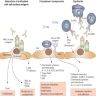
- Due to circulating antibodies against donor HLA, non-HLA or ABO antigens, i.e. donor specific antibodies (DSA) (Clin Biochem 2016;49:320)
- DSAs can be preformed (in which case antibody mediated rejection occurs during the very early posttransplant period - hyperacute / accelerated rejection) or may develop de novo after transplantation (usually due to inadequate immunosuppression or nonadherence)
- These antibodies bind to donor specific antigens on the vascular endothelium of the graft and result in complement activation
- This leads to activation of polymorphonuclear inflammatory cells, NK cell and monocyte recruitment and inflammation, as well as activation of the coagulation cascade
- This in turn leads to widespread microvascular injury evident as peritubular capillaritis, glomerulitis and microvascular thrombosis
- Eventually, transplant glomerulopathy develops (chronic phase) due to recurrent injury and repair (manifested as proteinuria) - glomerular basement membrane remodeling, mesangial matrix expansion, capillary obliteration, foot process effacement
- References: Transplant Rev (Orlando) 2017;31:257, Transplant Rev (Orlando) 2017;31:47
Clinical features
- Acute antibody mediated rejection (ABMR):
- Usually seen during the first few weeks after transplantation but can occur later, usually associated with decreased immunosuppression or noncompliance
- Presents with acute renal failure or oliguria, sometimes severe enough to require dialysis
- Chronic / chronic active ABMR: chronic renal failure with proteinuria
- Subclinical ABMR: stable creatinine but histological evidence of ABMR
- Reference: J Transplant 2012;2012:193724
Diagnosis
- Established via indication or protocol biopsies (Transplant Rev (Orlando) 2017;31:257, Transplant Rev (Orlando) 2017;31:47)
Diagnostic criteria and Banff classification / grading (modified from the Banff 2019 revision, Am J Transplant 2020;20:2318):
- Active antibody mediated rejection (ABMR): all 3 criteria must be met for diagnosis
- Histologic evidence of acute tissue injury; 1+ of the following should be present:
- Microvascular inflammation (g > 0 or ptc > 0), in the absence of recurrent or de novo glomerulonephritis; ptc ≥ 1 alone is not sufficient and g must be ≥ 1 if:
- Borderline infiltrate is present
- Acute T cell mediated rejection is also present
- Infection is present
- Intimal or transmural arteritis (v > 0)
- Acute thrombotic microangiopathy (in the absence of any other cause)
- Acute tubular injury (in the absence of any other cause)
- Microvascular inflammation (g > 0 or ptc > 0), in the absence of recurrent or de novo glomerulonephritis; ptc ≥ 1 alone is not sufficient and g must be ≥ 1 if:
- Evidence of current / recent antibody interaction with vascular endothelium; 1+ of the following should be present:
- Linear C4d staining in peritubular capillaries (C4d2 or C4d3 by immunoflourescence on frozen sections or C4d > 0 by immunohistochemistry on paraffin sections)
- At least moderate microvascular inflammation ([g + ptc] ≥ 2) in the absence of recurrent or de novo glomerulonephritis; ptc ≥ 2 alone is not sufficient and g must be ≥ 1 if:
- Borderline infiltrate is present
- Acute T cell mediated rejection is also present
- Infection is present
- Increased expression of gene transcripts / classifiers in the biopsy tissue strongly associated with ABMR (endothelial associated transcripts [ENDAT])
- Serologic evidence of donor specific antibodies (DSA) to HLA or other antigens
- C4d staining or expression of validated transcripts / classifiers (criterion 2 above) may substitute for DSA
- However, thorough DSA testing (including testing for non-HLA antibodies if HLA antibody testing is negative) is strongly advised whenever criteria 1 and 2 are met
- Histologic evidence of acute tissue injury; 1+ of the following should be present:
- Chronic active ABMR: all 3 criteria must be met for diagnosis
- Morphologic evidence of chronic tissue injury; 1+ of the following should be present:
- Transplant glomerulopathy (cg > 0) if no evidence of chronic thrombotic microangiopathy or chronic recurrent / de novo glomerulonephritis; includes changes evident by electron microscopy alone (cg1a)
- Severe peritubular capillary basement membrane multilayering (requires electron microscopy)
- Arterial intimal fibrosis of new onset, excluding other causes (leukocytes within the sclerotic intima favor chronic ABMR if there is no prior history of T cell mediated rejection but are not required)
- Same as criterion 2 for active ABMR
- Same as criterion 3 for active ABMR
- Morphologic evidence of chronic tissue injury; 1+ of the following should be present:
- Chronic (inactive) ABMR
- Transplant glomerulopathy (cg > 0) or severe peritubular capillary basement membrane multilayering
- Absence of criterion 2 for active ABMR
- Prior documented active or chronic active ABMR
- C4d staining without evidence of rejection: all 4 features must be present for diagnosis
- Linear C4d staining in peritubular capillaries (C4d2 or C4d3 by immunoflorescence on frozen sections or C4d > 0 by immunohistochemistry on paraffin sections)
- Criterion 1 for active or chronic active ABMR not met
- No molecular evidence for ABMR (criterion 2 for active and chronic active ABMR)
- No acute or chronic active T cell mediated rejection or borderline changes
Laboratory
- Acute antibody mediated rejection (ABMR): acute increase in serum creatinine levels
- Chronic active ABMR: chronic increase in serum creatinine levels along with proteinuria, usually nephrotic range
- Serum donor specific antibodies: anti-HLA or non-HLA antibodies (J Transplant 2012;2012:193724)
Radiology description
- Contrast enhanced ultrasound has been shown to be of diagnostic value in identifying cases of vascular rejection (Clin Hemorheol Microcirc 2018;69:77)
- New technologies for identification of acute rejection are at the experimental stage (Am J Nucl Med Mol Imaging 2019;9:110)
- Imaging seems to rely on analysis of changes in blood flow, which decreases with acute rejection induced inflammation and detection of recruitment of activated leucocytes with 18F-fluoro-deoxy-glucose positron emission tomography (Clin Kidney J 2017;10:97)
Prognostic factors
- Chronic active antibody mediated rejection is associated with poor graft survival and is today the leading cause of graft scarring and loss (Am J Transplant 2009;9:2520)
Case reports
- 8 year old boy with development of ABMR shortly after an episode of acute T cell mediated rejection (CEN Case Rep 2018;7:288)
- 22 year old man with severe eosinophilic antibody mediated rejection (Indian J Nephrol 2018;28:389)
- 32 year old man with acute T cell mediated rejection accompanied by C4d- acute antibody mediated rejection (Nephrology (Carlton) 2015;20:70)
- 44 year old man with probable C4d- accelerated acute antibody mediated rejection due to non-HLA antibodies (Nephrology (Carlton) 2015;20:75)
- 49 year old woman with acute ABMR managed with bortezomid based treatment (Genet Mol Res 2015;14:17951)
Treatment
- Plasmapheresis (J Transplant 2012;2012:193724)
- Intravenous immunoglobulin
- Rituximab - efficacy unclear
- Bortezomid - efficacy unclear
- Proteosome inhibition - efficacy unclear
- Complement inhibition
- References: Am J Transplant 2018;18 Suppl 3:3, Transplantation 2018;102:557
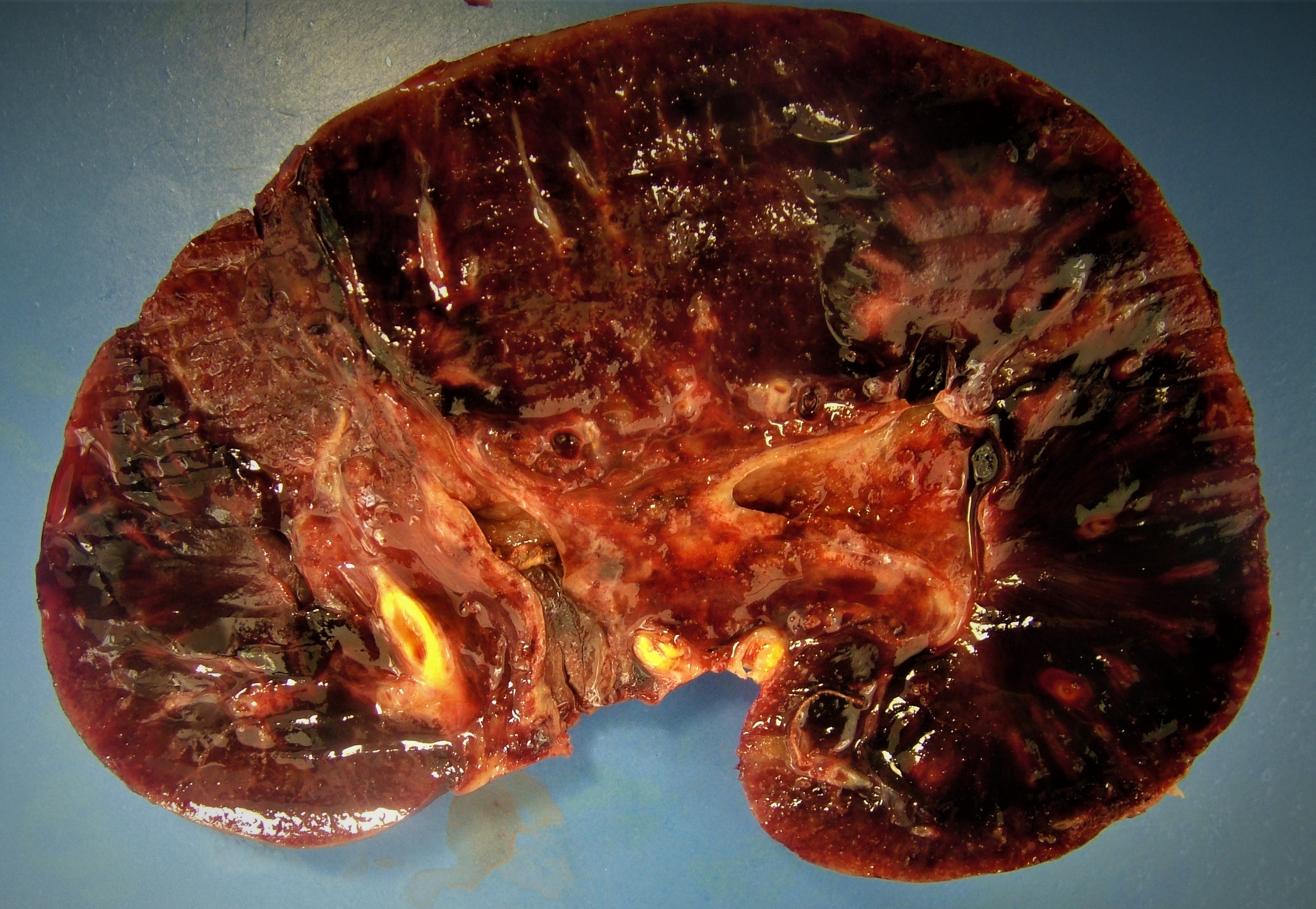
Gross description
- Active / chronic active ABMR: kidneys are swollen and congested, may have large areas of hemorrhage or infarction
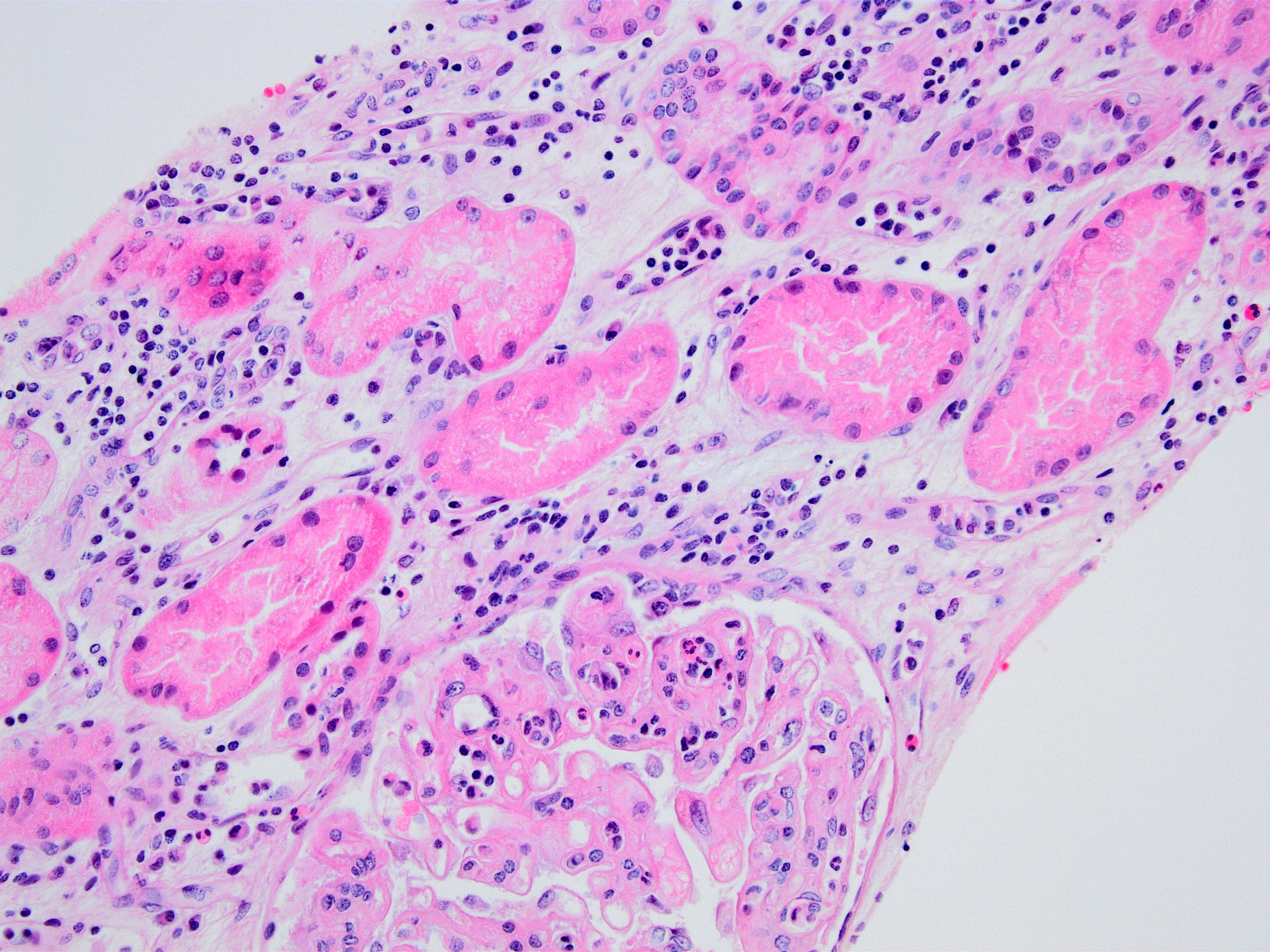
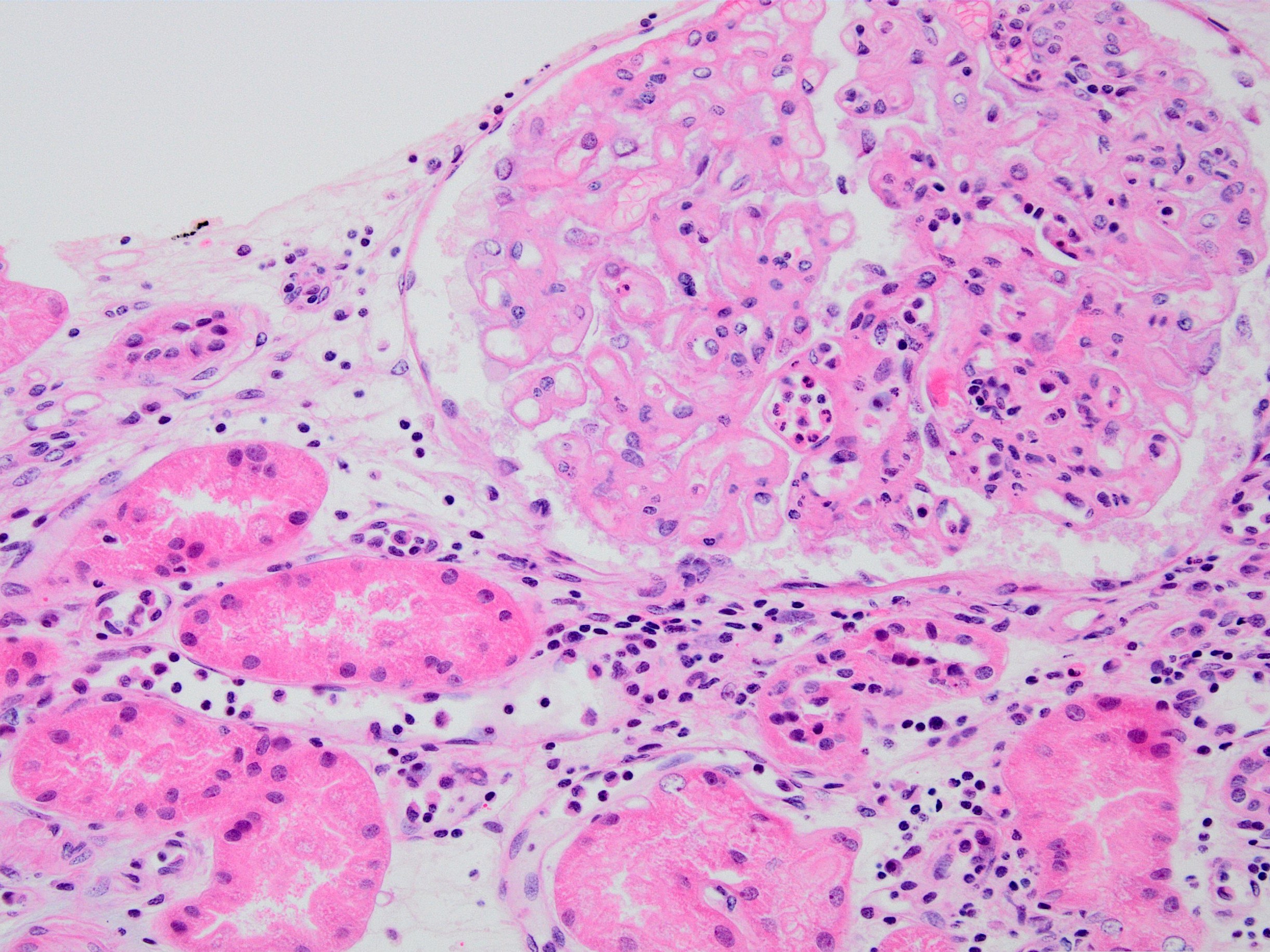
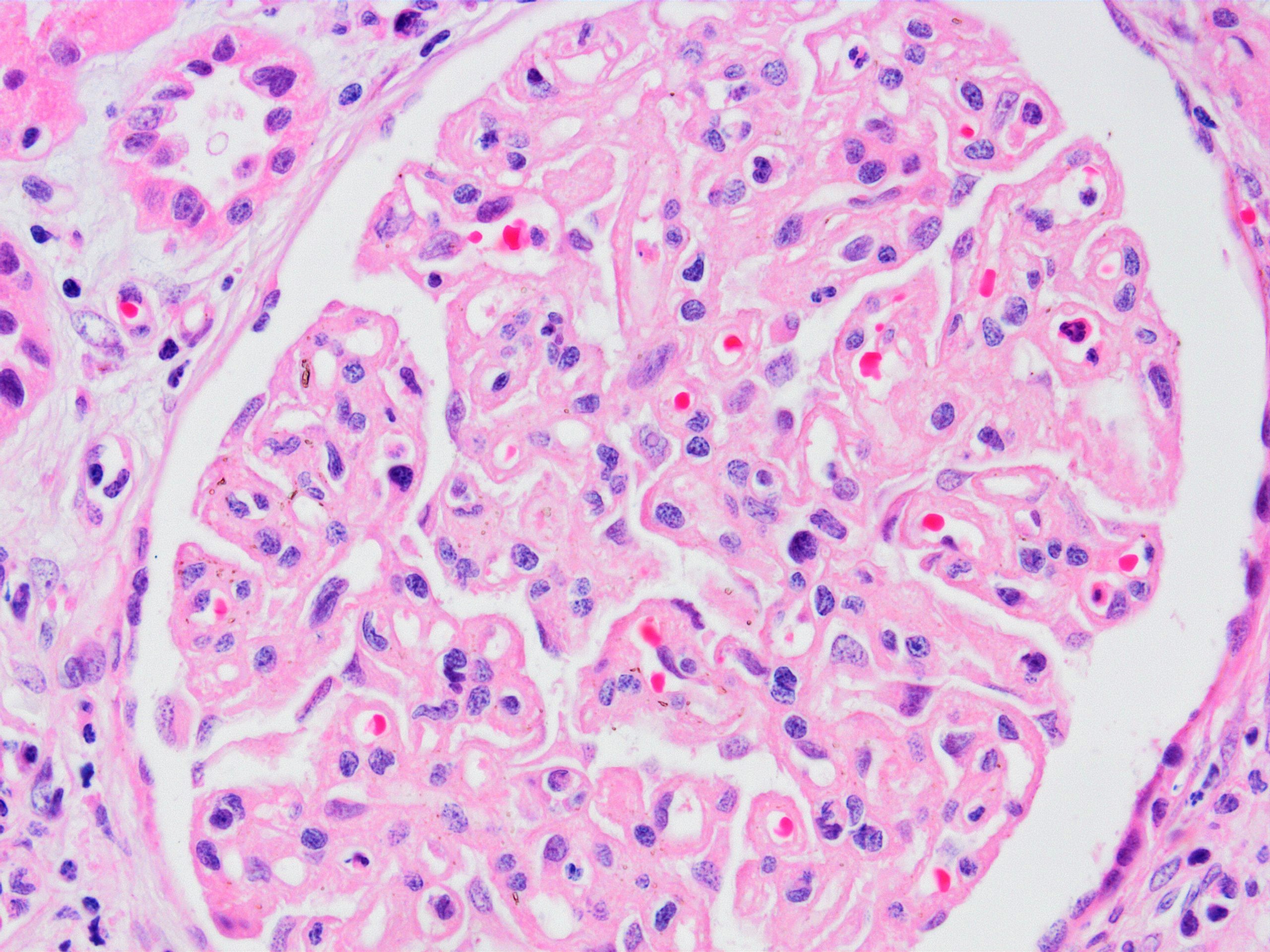
Microscopic (histologic) description
- Hyperacute antibody mediated rejection (ABMR):
- Transmural vasculitis
- Severe cortical necrosis
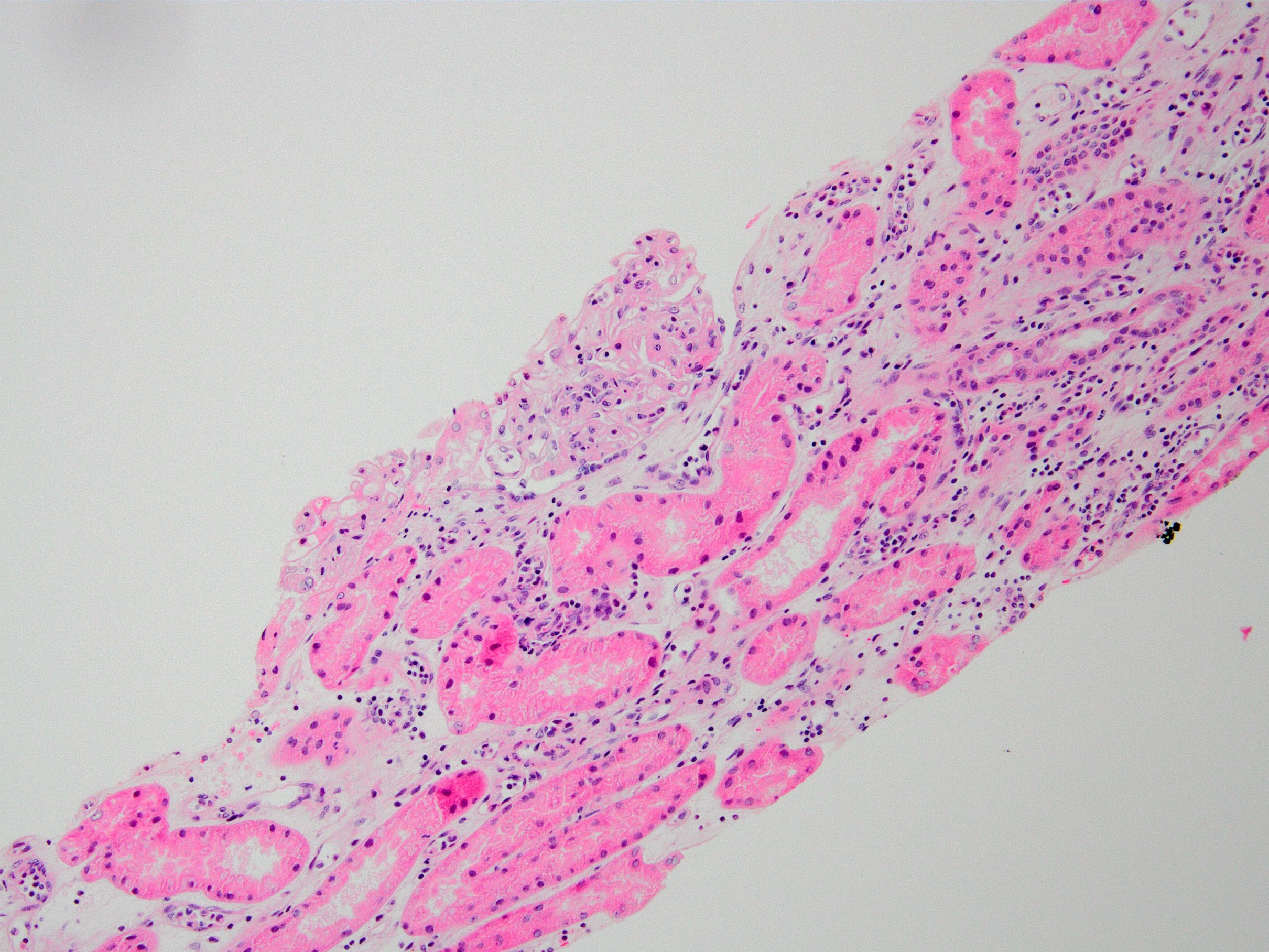
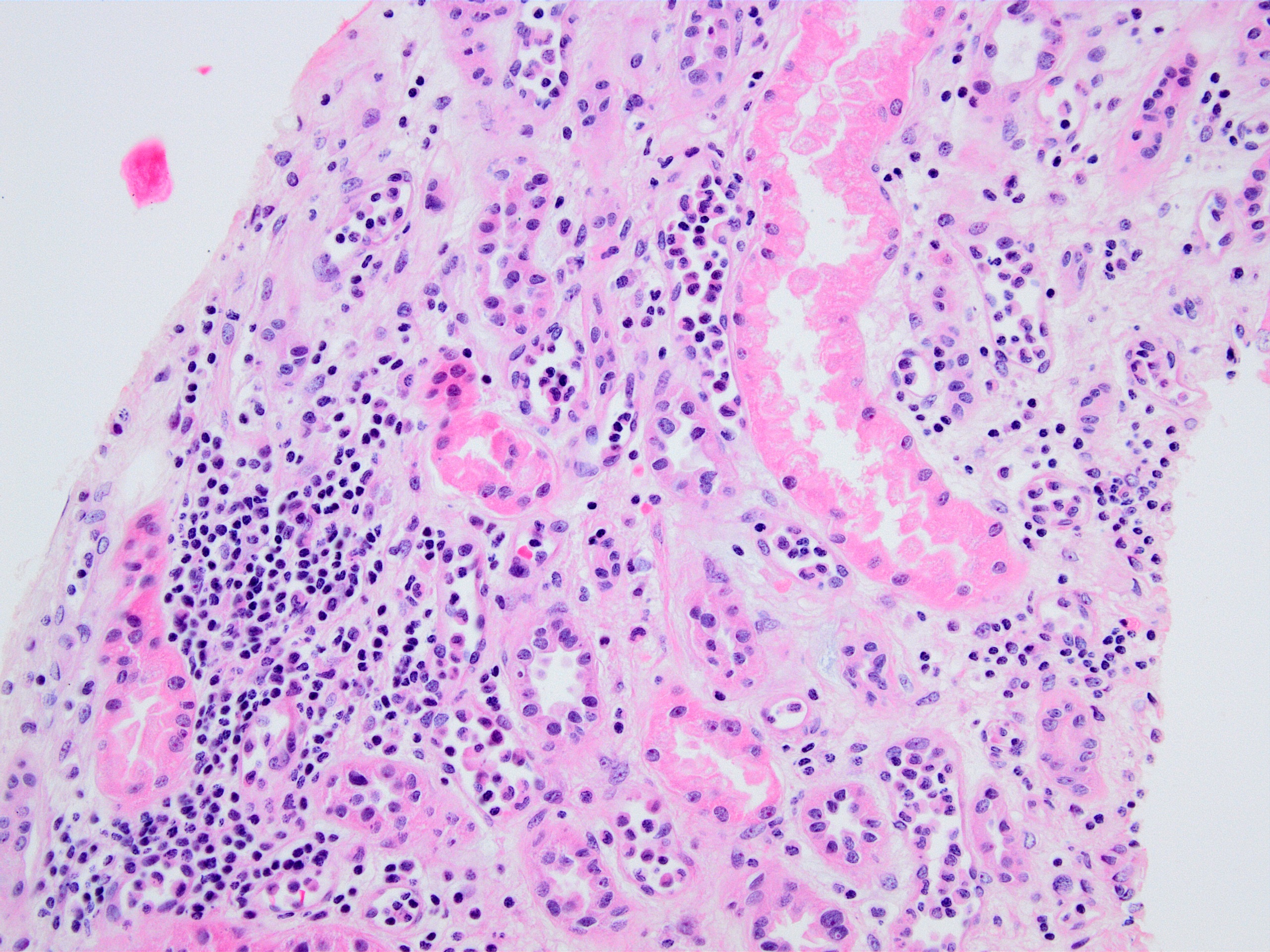
- Active ABMR:
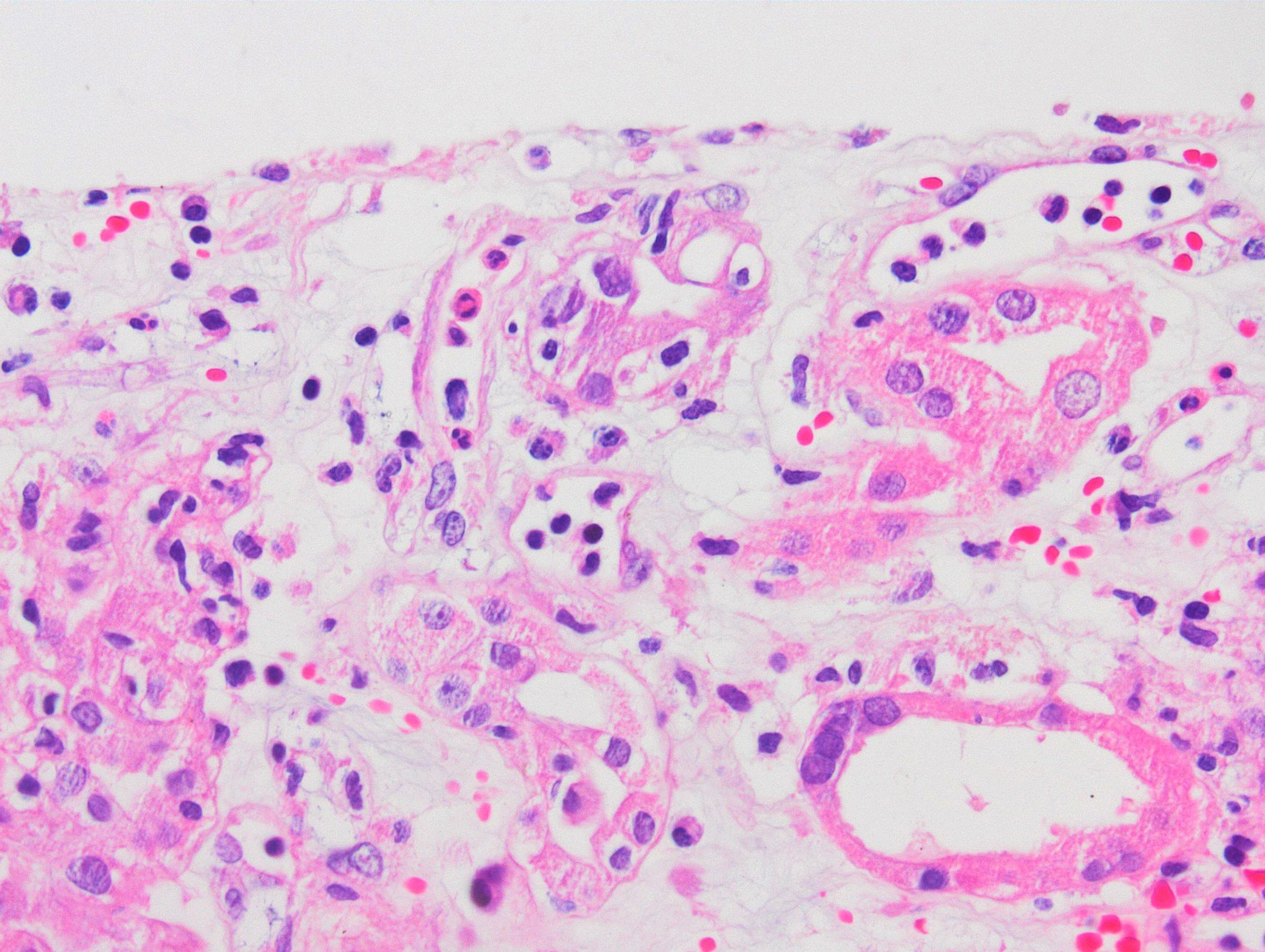
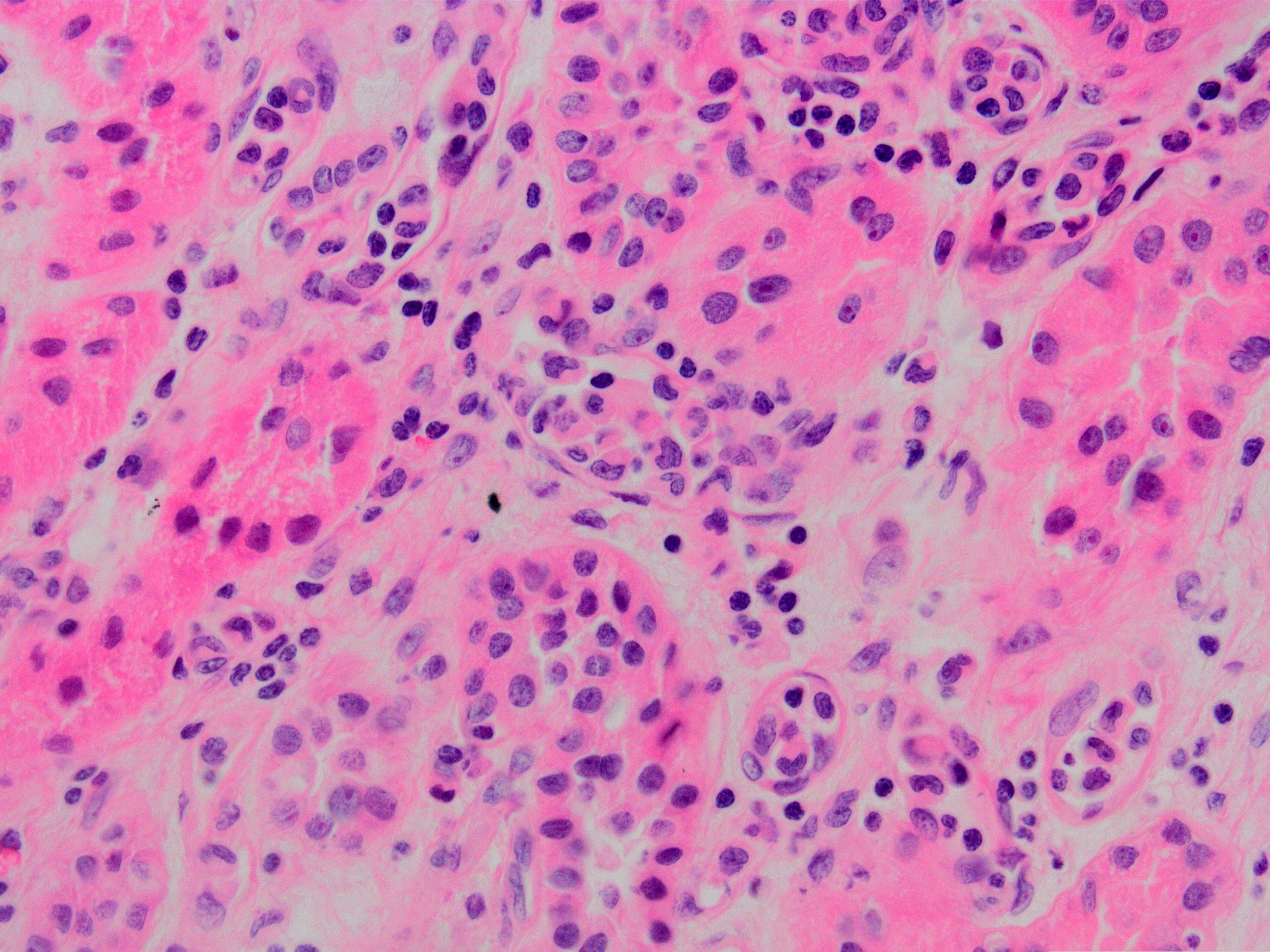
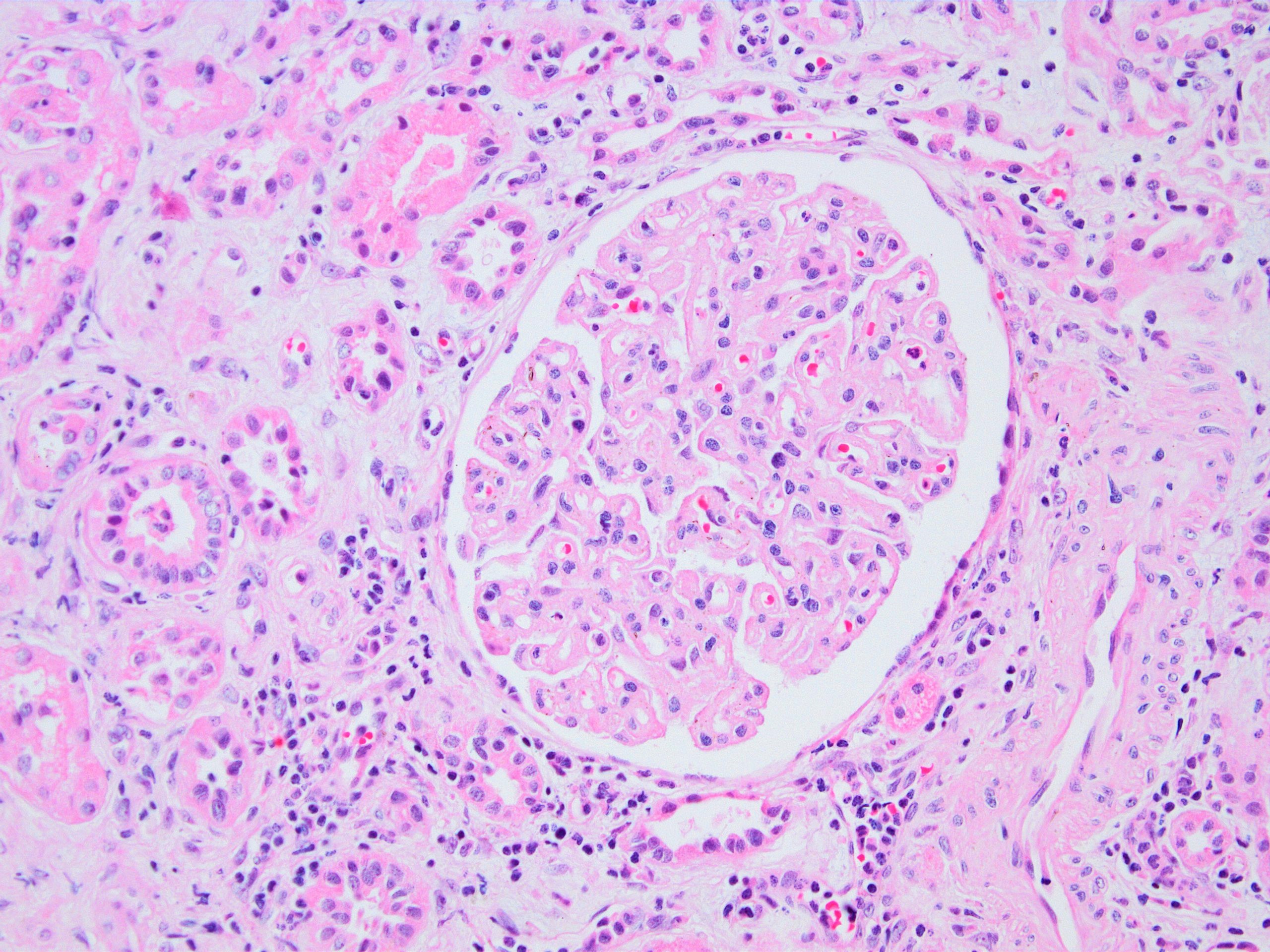
- Peritubular capillaritis: presence of inflammatory cells within the lumens of the capillaries - most prominently neutrophils and monocytes
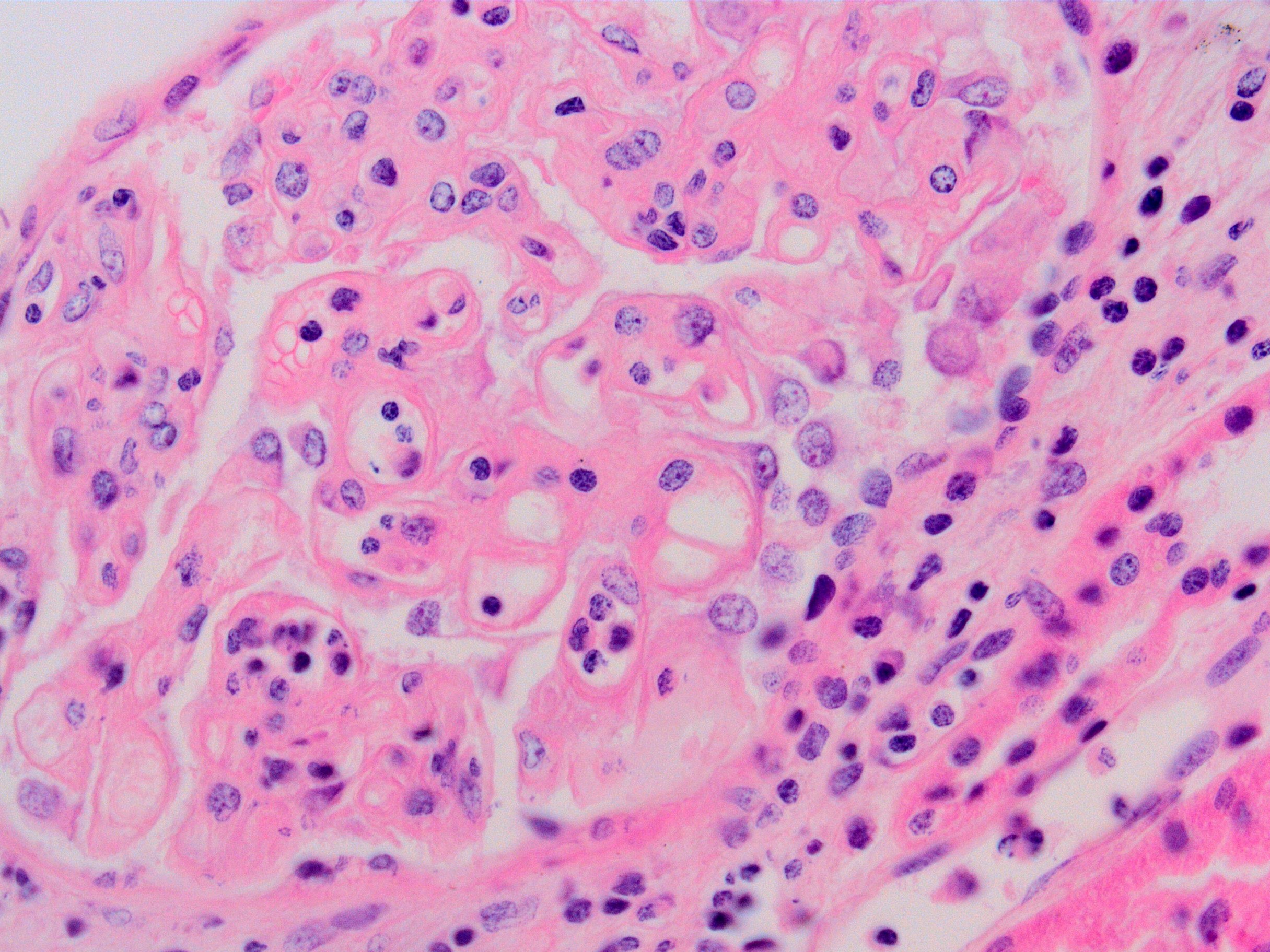
- Glomerulitis: inflammatory cells within glomerular capillary lumens
- Intimal or transmural arteritis: inflammatory cells within the intima or walls of vessels
- Thrombotic microangiopathy and fibrinoid necrosis of vessel walls
- Acute tubular injury: dilatation of the tubular lumen, flattening of tubular epithelial cells, loss of nuclear staining of tubular epithelial cells, shedding of tubular epithelial cells into the lumen and denudation, regenerative changes in tubular epithelial cells such as nucleolar enlargement and hyperchromasia
- Linear C4d staining along peritubular capillaries
- Chronic active ABMR: in addition to above mentioned changes for active ABMR, changes associated with chronic injury are present
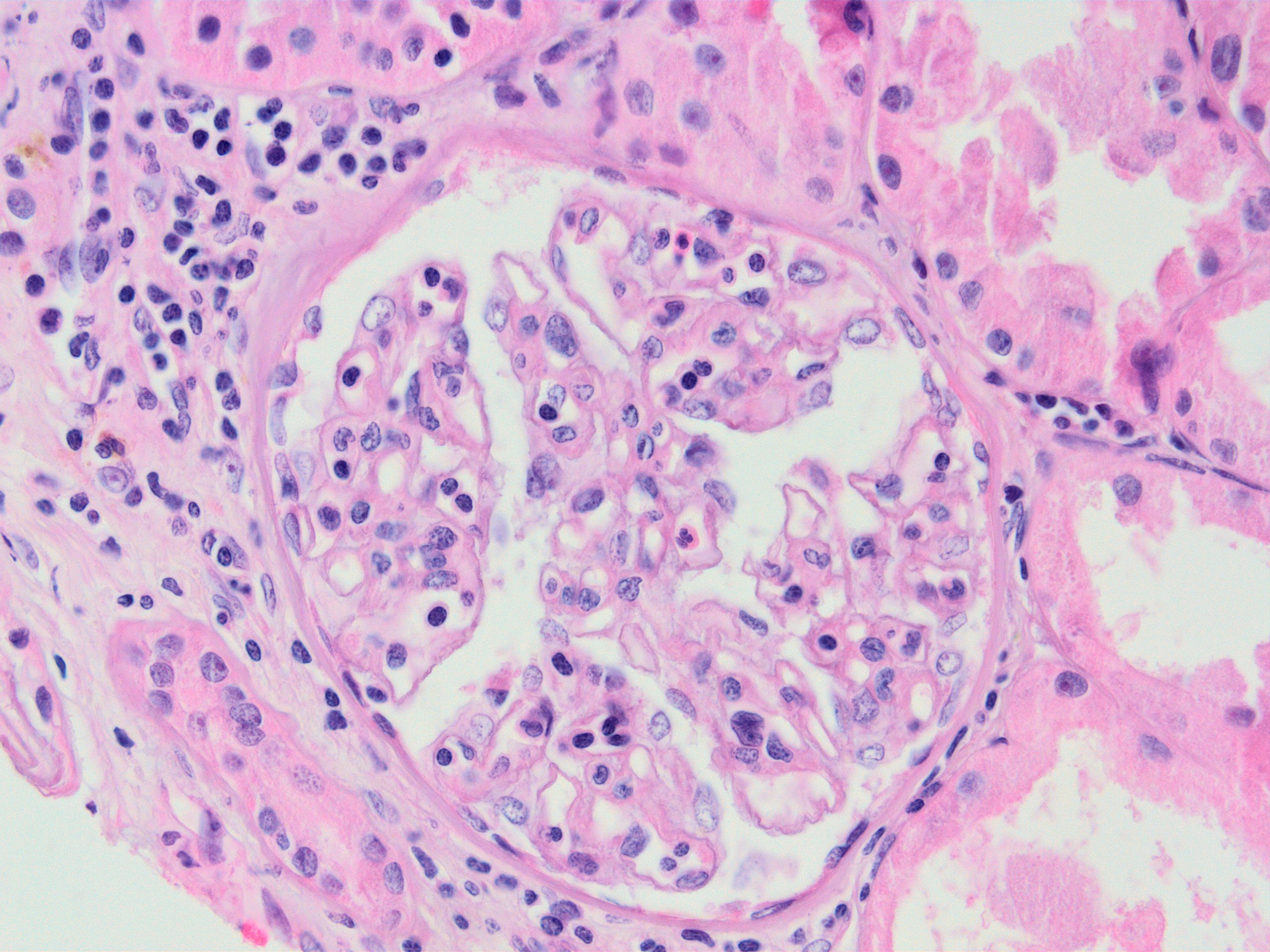
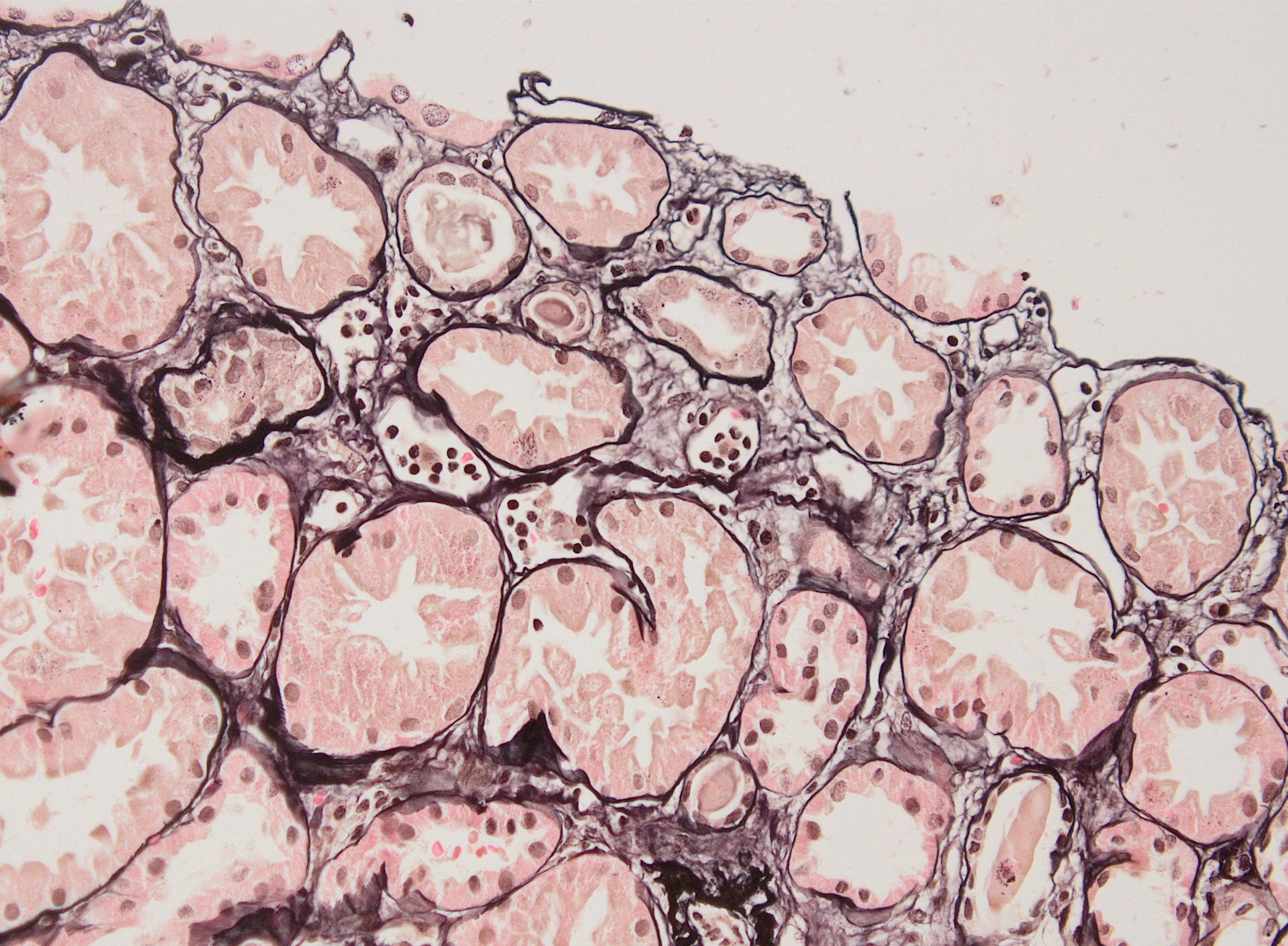
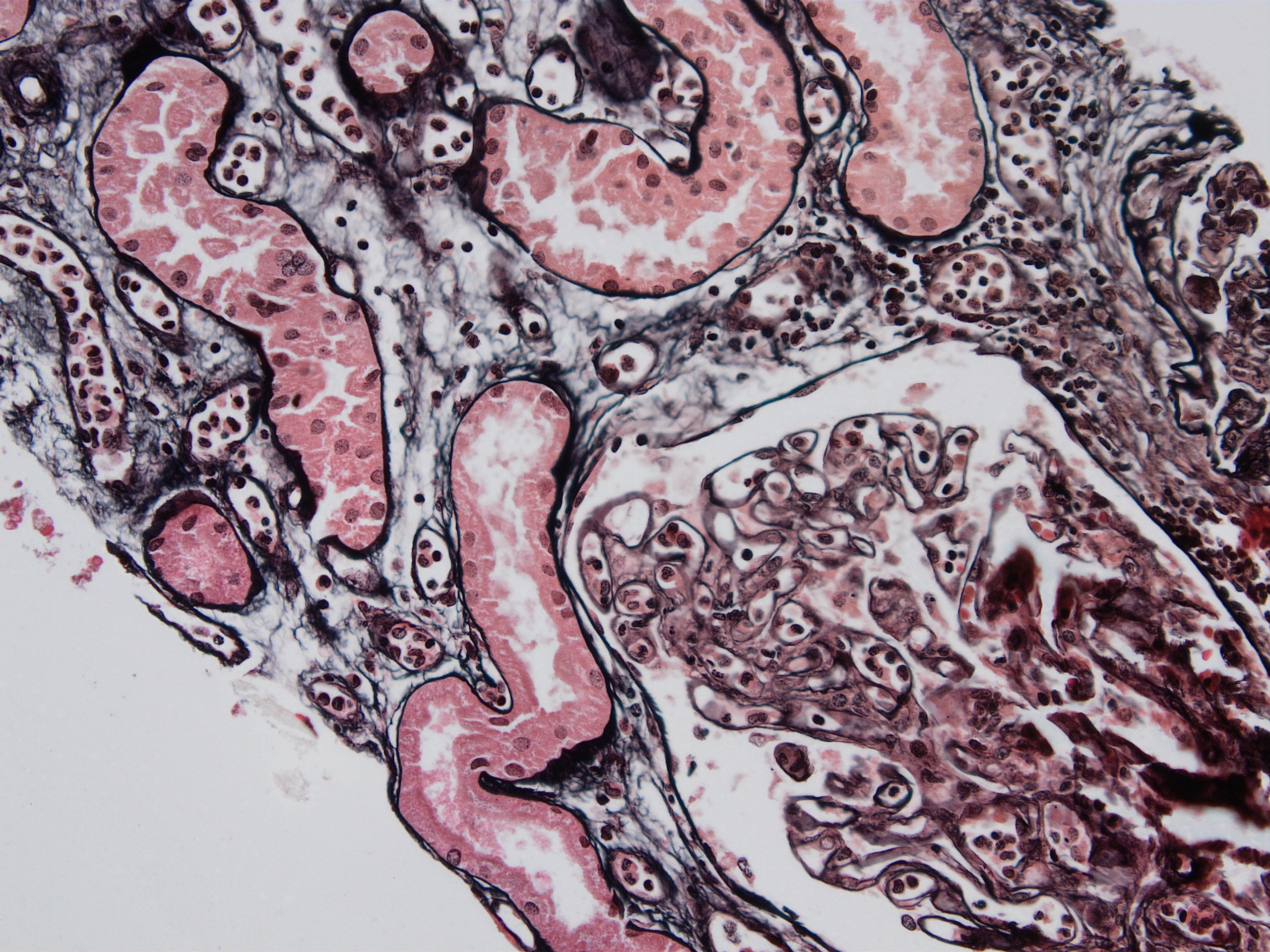
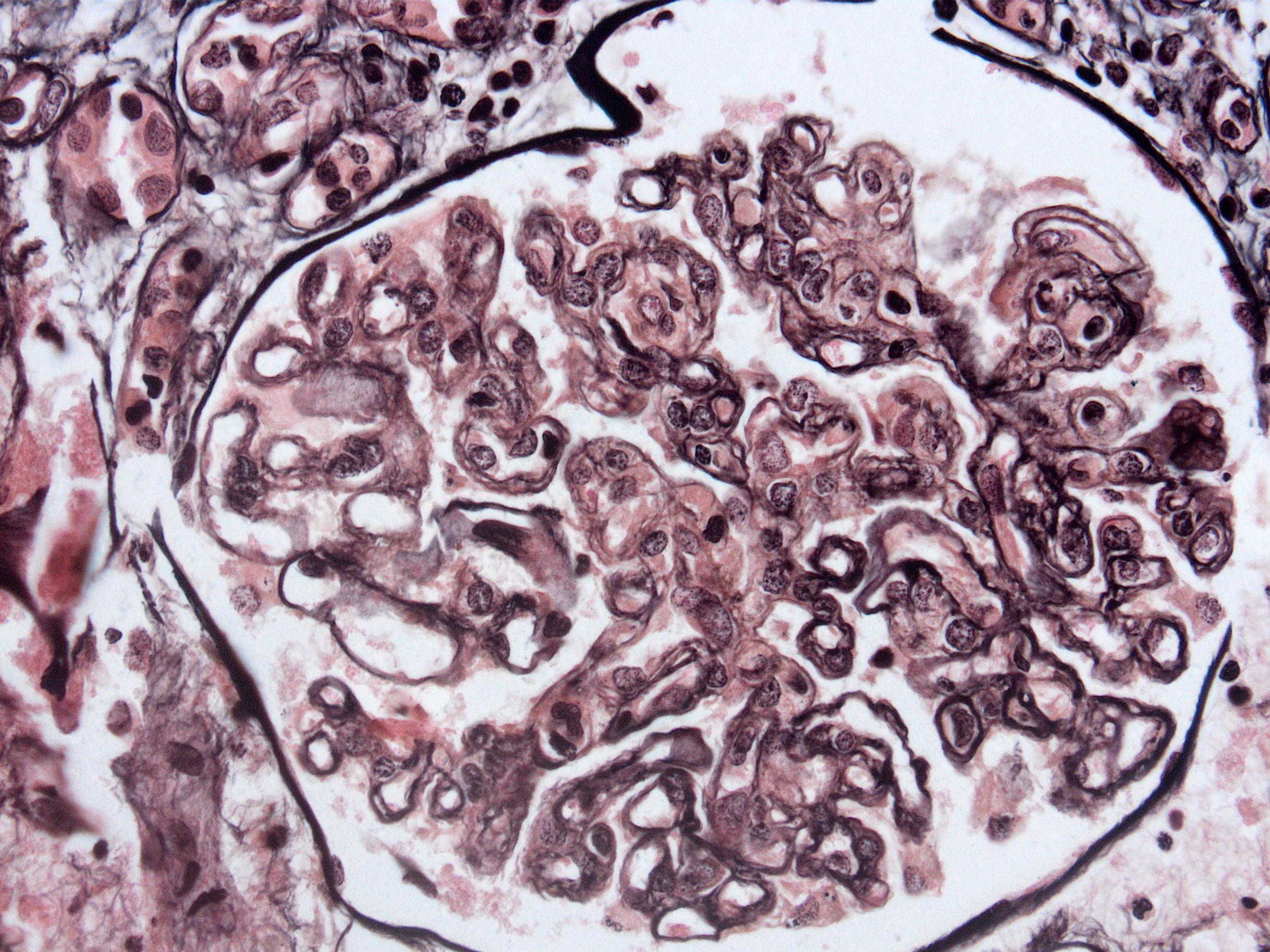
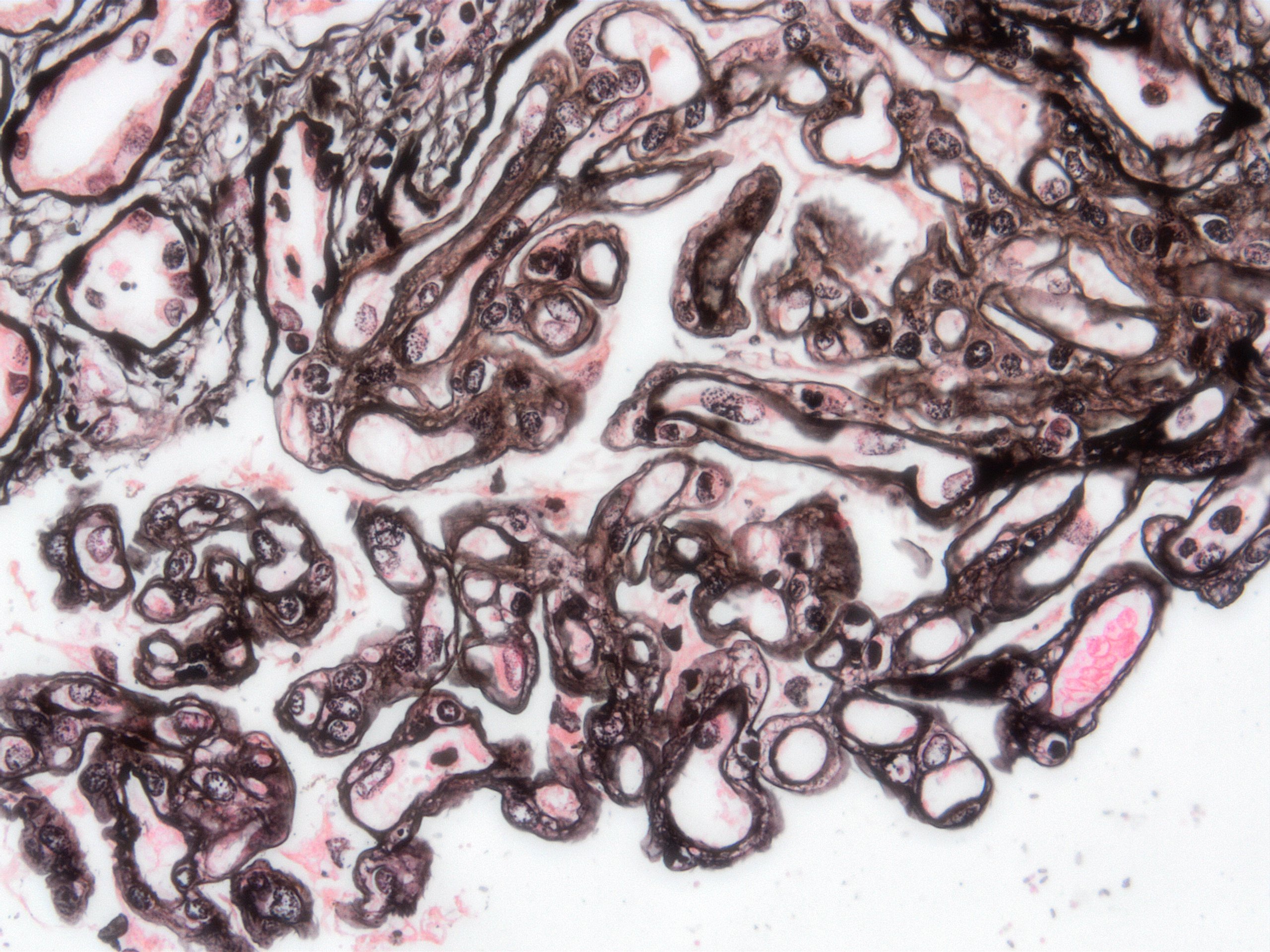
- Transplant glomerulopathy: double contours along the glomerular basement membrane, expansion of mesangium and obliteration of capillary lumina; usually accompanied by linear C4d staining along the glomerular basement membrane
- Peritubular basement membrane multilayering (with electron microscopy)
- Arterial intimal fibrosis with presence of inflammatory cells (transplant arteriopathy)
- Interstitial fibrosis and tubular atrophy
- Chronic ABMR:
- Transplant glomerulopathy: double contours along the glomerular basement membrane, expansion of mesangium and obliteration of capillary lumina; usually accompanied by linear C4d staining along the glomerular basement membrane
- Peritubular basement membrane multilayering (with electron microscopy)
- Arterial intimal fibrosis with presence of inflammatory cells (transplant arteriopathy)
- Interstitial fibrosis and tubular atrophy
- References: Transplant Rev (Orlando) 2017;31:47, Mod Pathol 2018;31:235, Transplantation 2018;102:1795
Banff scoring of histological lesions
- v - vascular inflammation: the most severely affected artery dictates the score; an asterisk is added to the v score if interstitial hemorrhage or infarct present
- v0: no arteritis
- v1: intimal arteritis with < 25% luminal area lost (minimum = 1 cell, 1 artery)
- v2: intimal arteritis with ≥ 25% of luminal area lost in 1+ arteries
- v3: transmural arteritis or fibrinoid necrosis (medial smooth muscle necrosis) with lymphocyte infiltrate in vessels
- g - glomerulitis: percentage of glomerular capillaries partially or completely occluded by inflammatory cells (polymorphonuclear leucocytes and mononuclear cells) and endothelial cell enlargement
- g0: no glomerulitis
- g1: < 25% of glomeruli involved (mostly segmental)
- g2: 25 - 75% of glomeruli involved (segmental to global)
- g3: > 75% of glomeruli involved (mostly global)
- ptc - peritubular capillaritis: the most severely affected peritubular capillary (PTC) dictates the score; an asterisk is added to the ptc score if neutrophils are lacking / only mononuclear cells are present
- ptc0: < 3 cells/PTC
- ptc1: 1+ inflammatory cells in > 10% of cortical PTCs with 3 - 4 cells in most severely involved PTC
- ptc2: 1+ inflammatory cells in > 10% of cortical PTCs with 5 - 10 cells in most severely involved PTC
- ptc3: 1+ inflammatory cells in > 10% of cortical PTCs with > 10 cells in most severely involved PTC
- C4d: percentage of PTC (or vasa recta in the medulla) that has linear circumferential staining, scored in at least 5 high powered fields of cortex or medulla without scarring or infarct
- C4d0: no staining of PTC and medullary vasa recta
- C4d1: < 10% of PTC and medullary vasa recta
- C4d2: 10 - 50% of PTC and medullary vasa recta
- C4d3: > 50% of PTC and medullary vasa recta
- cg - transplant glomerulopathy: percentage of glomerular capillary loops with duplication of glomerular basement membrane in most affected nonsclerotic glomerulus
- cg0: none by light microscopy (LM) and electron microscopy
- cg1a: only by electron microscopy in 3+ glomerular capillaries
- cg1b: ≤ 25% by LM (1+ glomerular capillaries with glomerular basement membrane double contours by LM)
- cg2: 26 - 50% by LM
- cg3: > 50% by LM
- cv - transplant arteriopathy: arterial fibrointimal thickening; percentage of narrowing of lumen of most severely affected artery
- cv0: none
- cv1: ≤ 25% of the luminal area
- cv2: 26 - 50% of the luminal area
- cv3: > 50% of the luminal area
- Peritubular capillary basement membrane multilayering (ptclm) - electron microscopic evaluation of the most affected PTC
- ptclm 1: 1 PTC with ≥ 7 layers +2 PTC with ≥ 5 layers
- Reference: Transplantation 2018;102:1795
Images hosted on other servers:
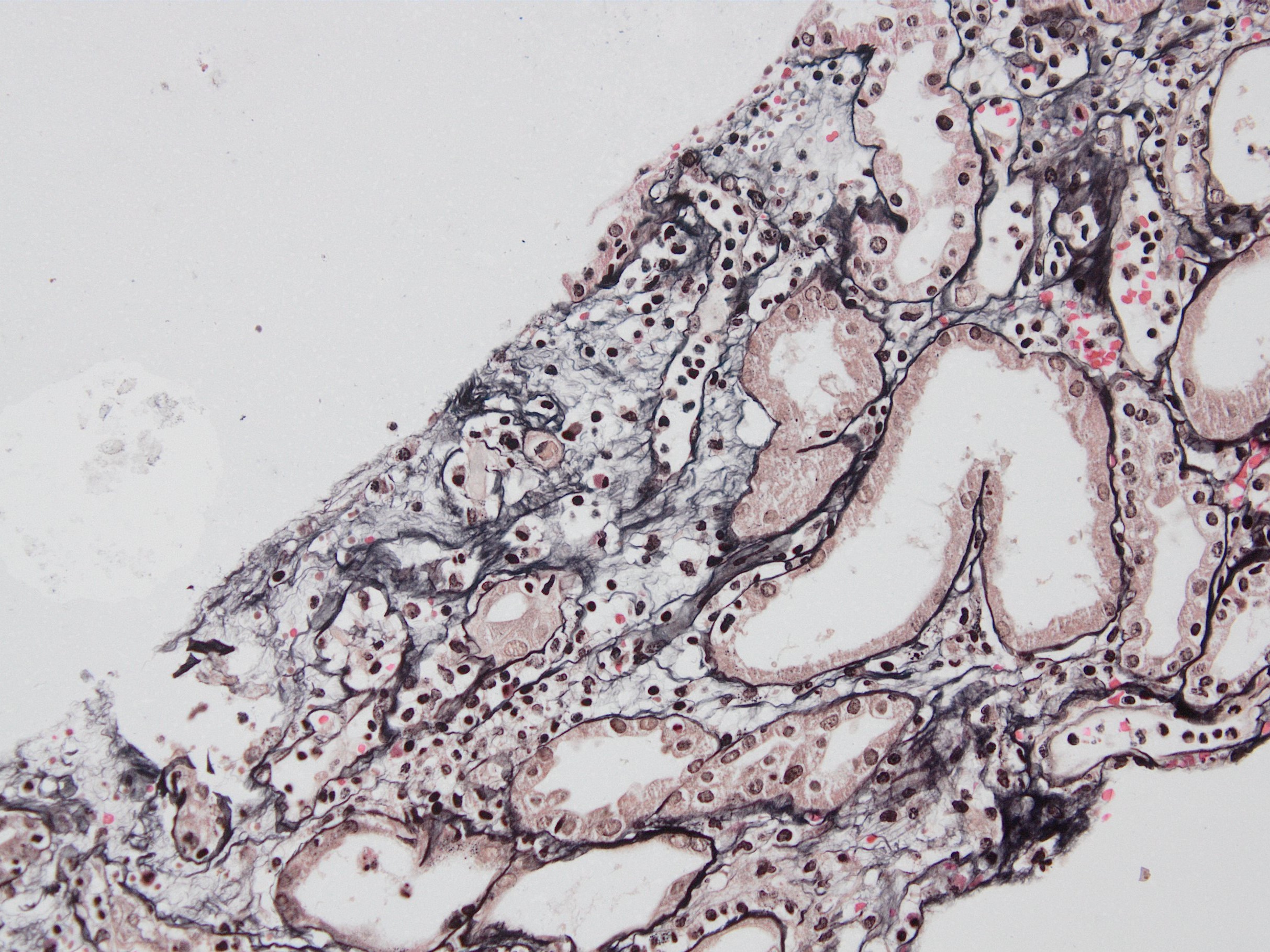
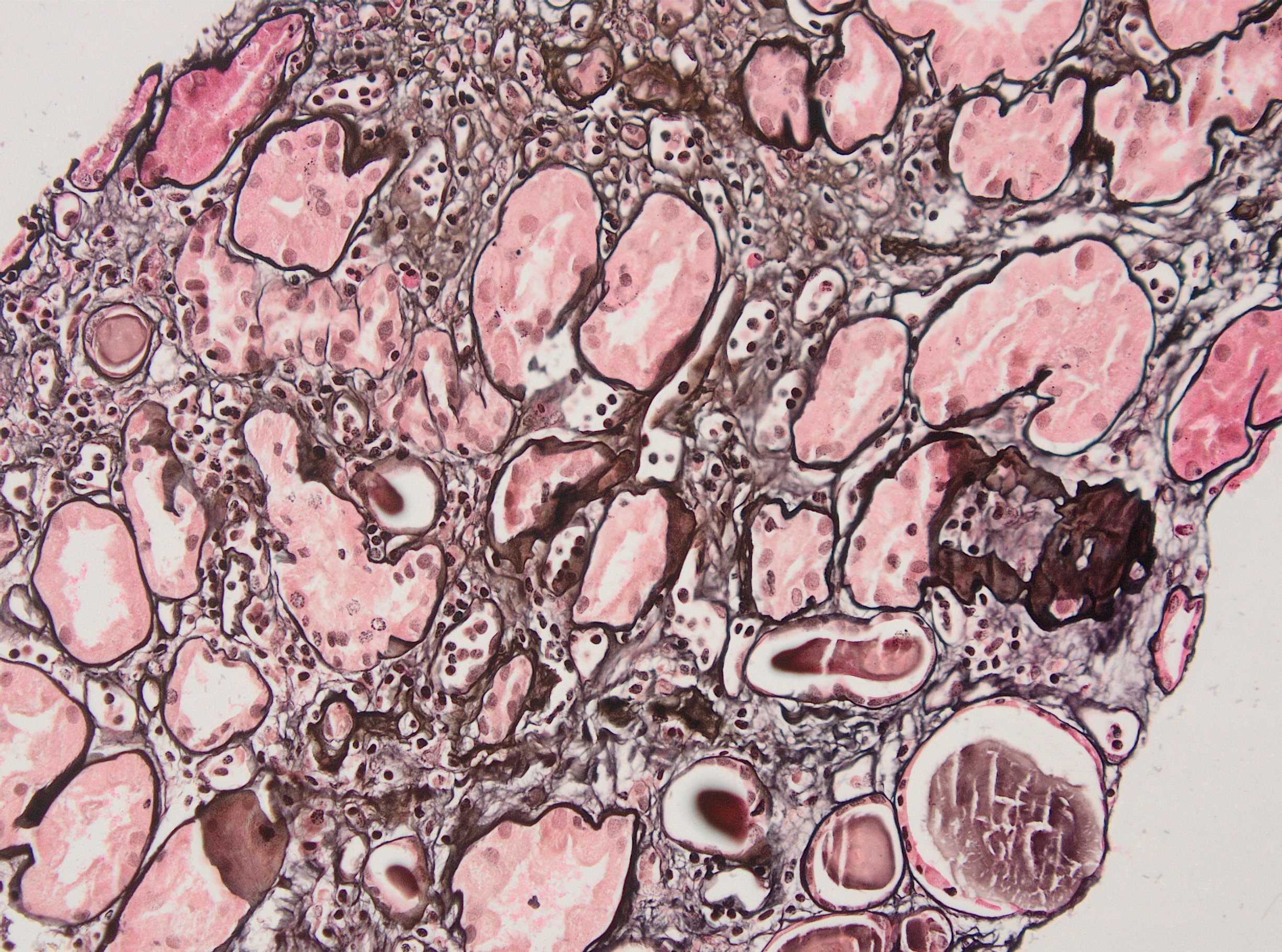
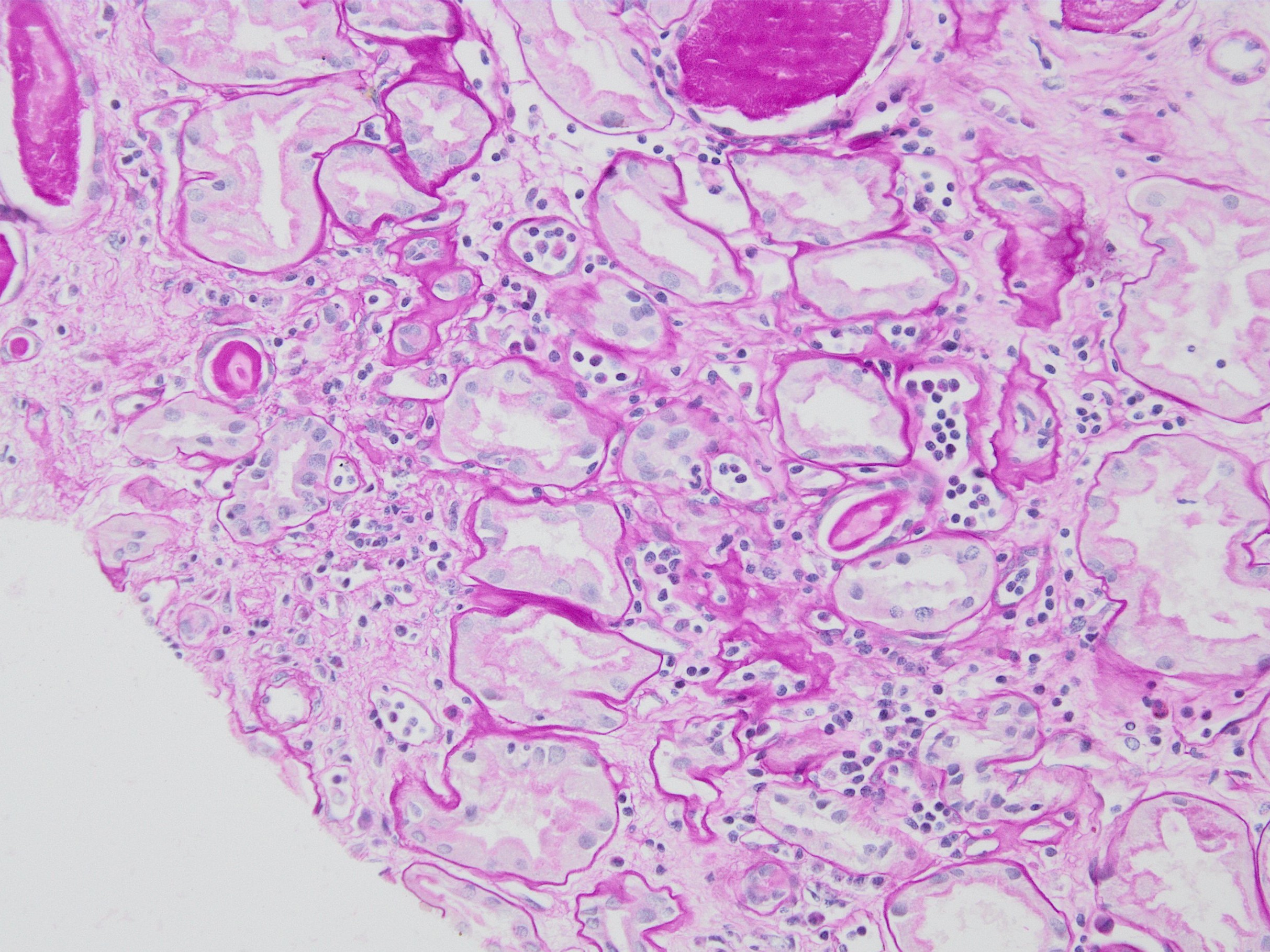
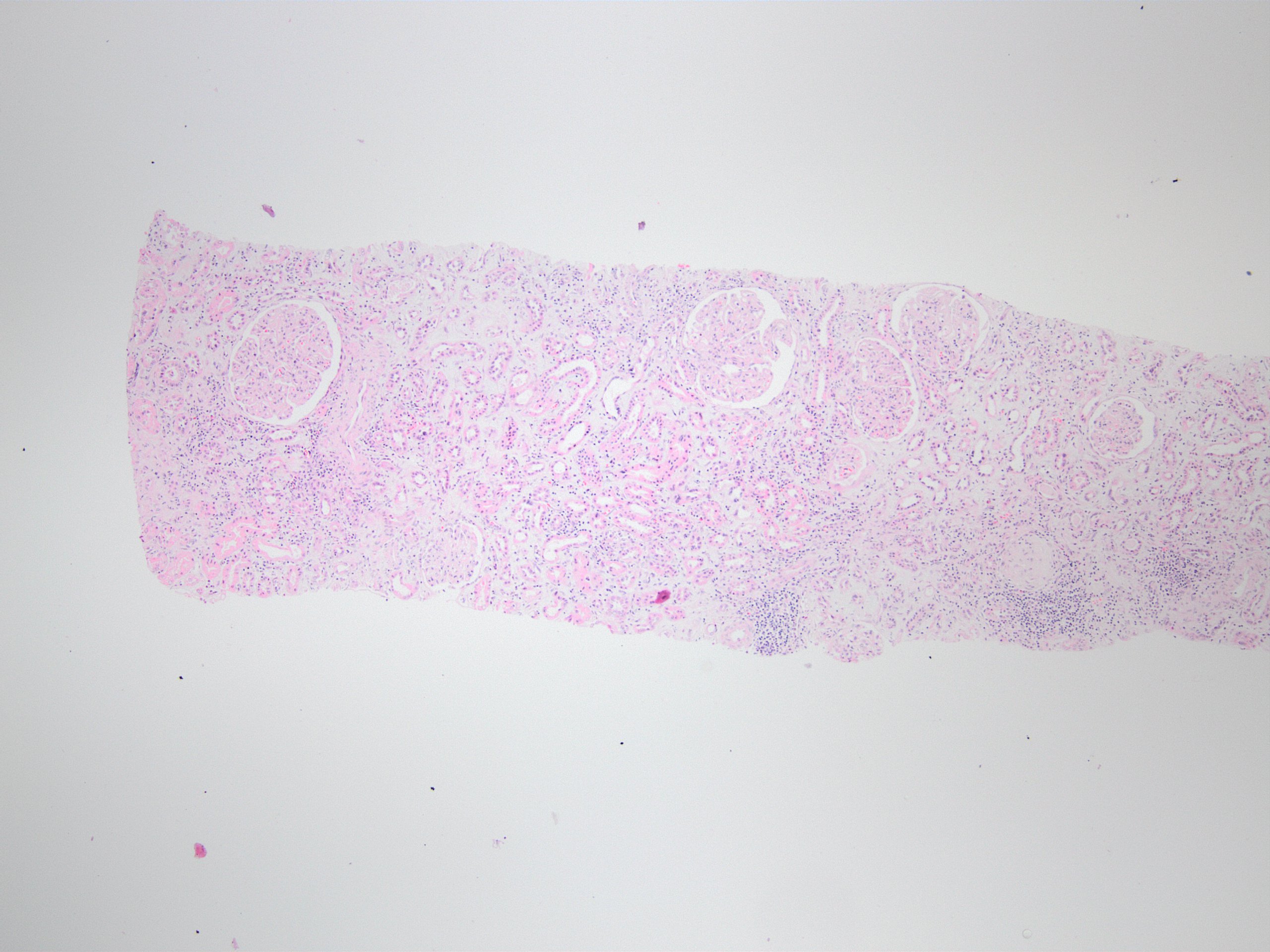
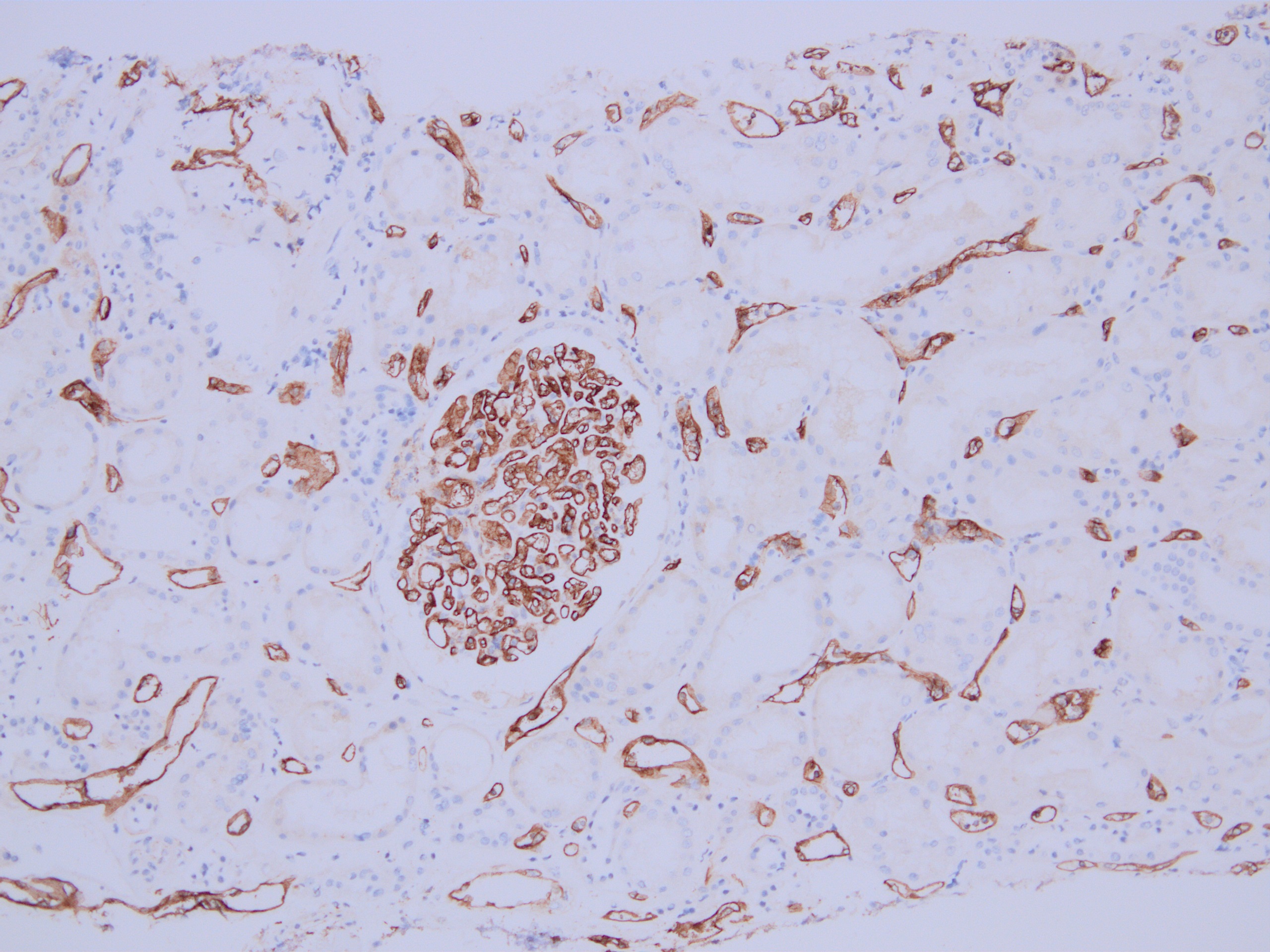
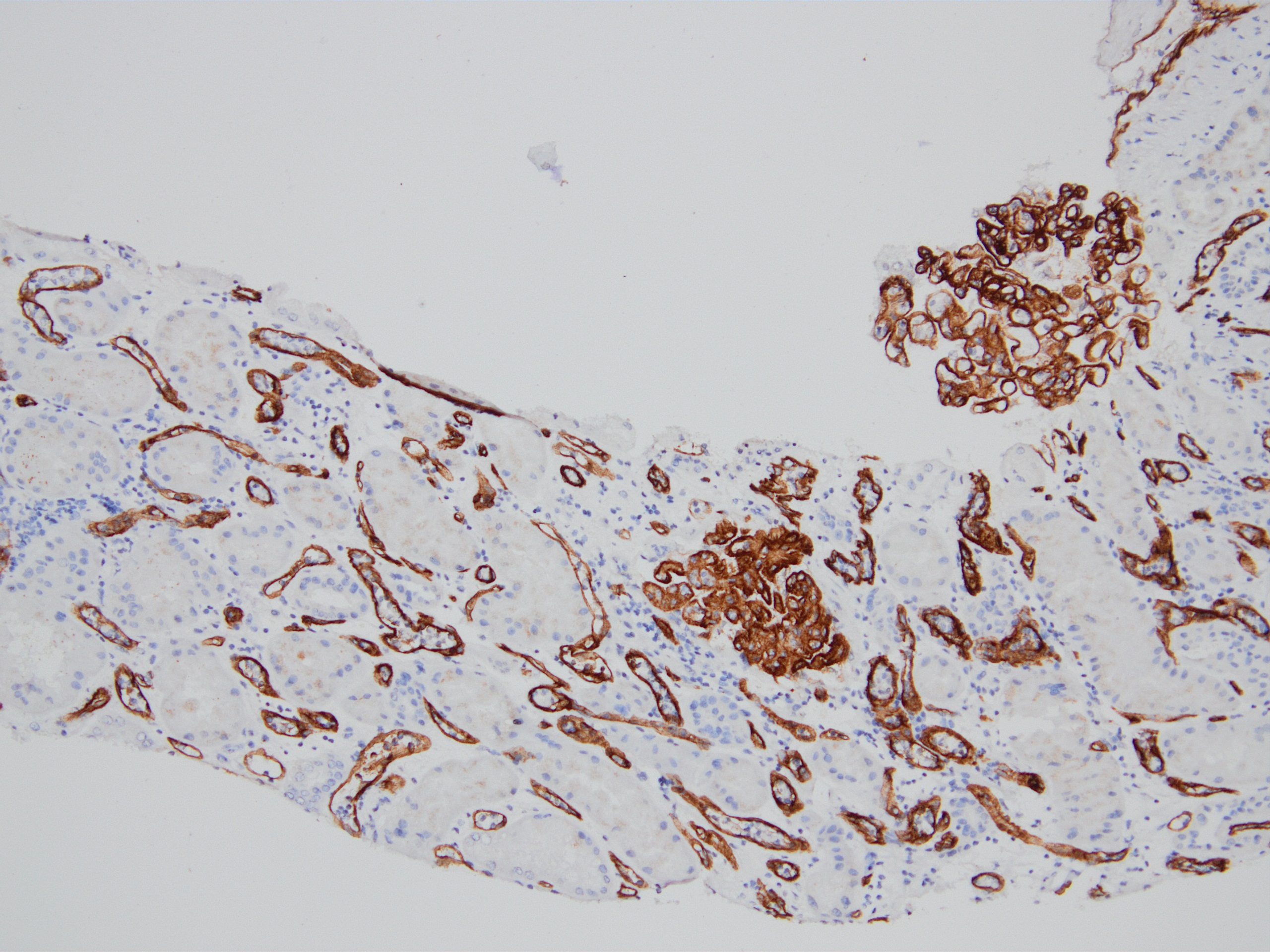
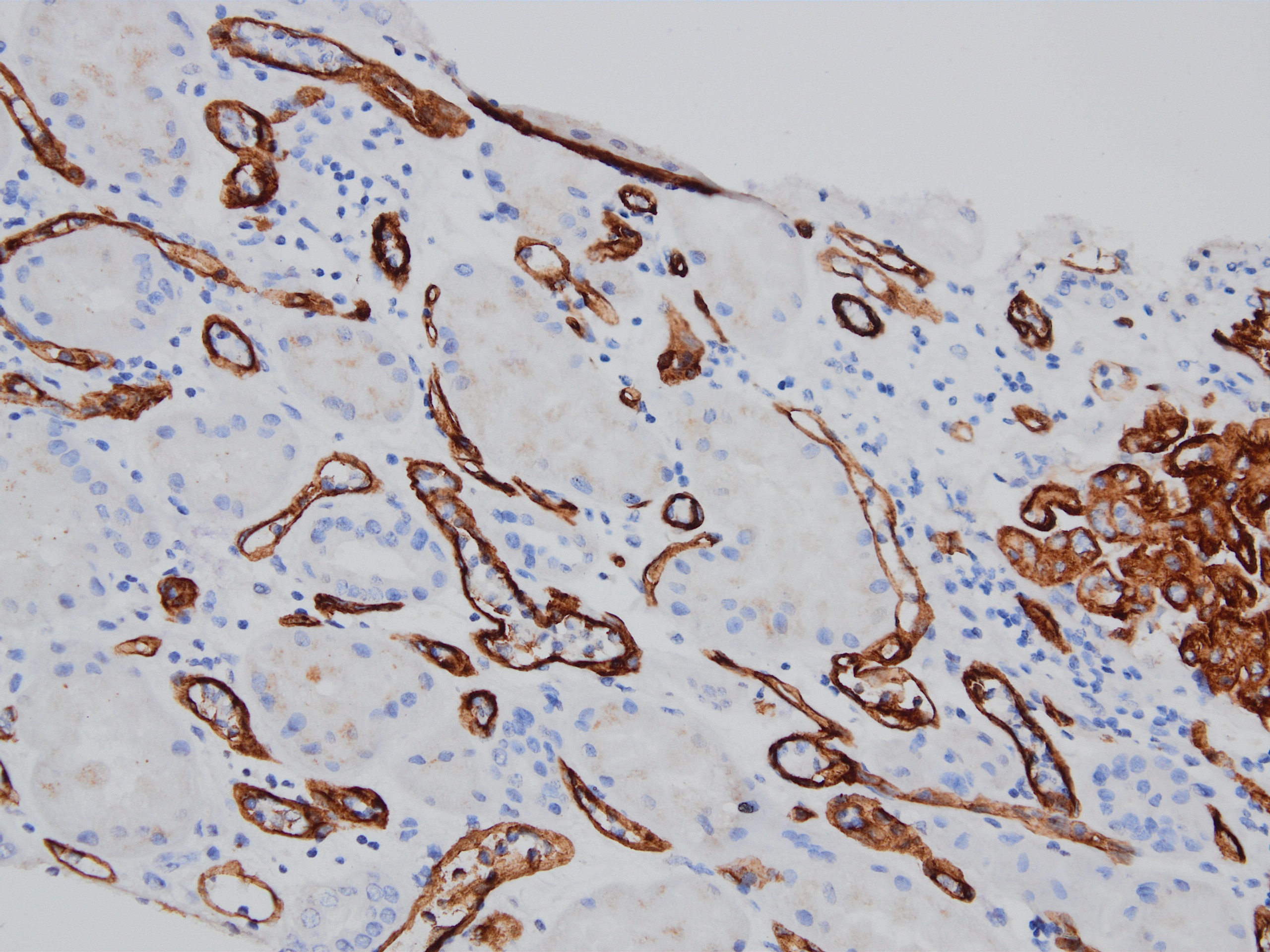
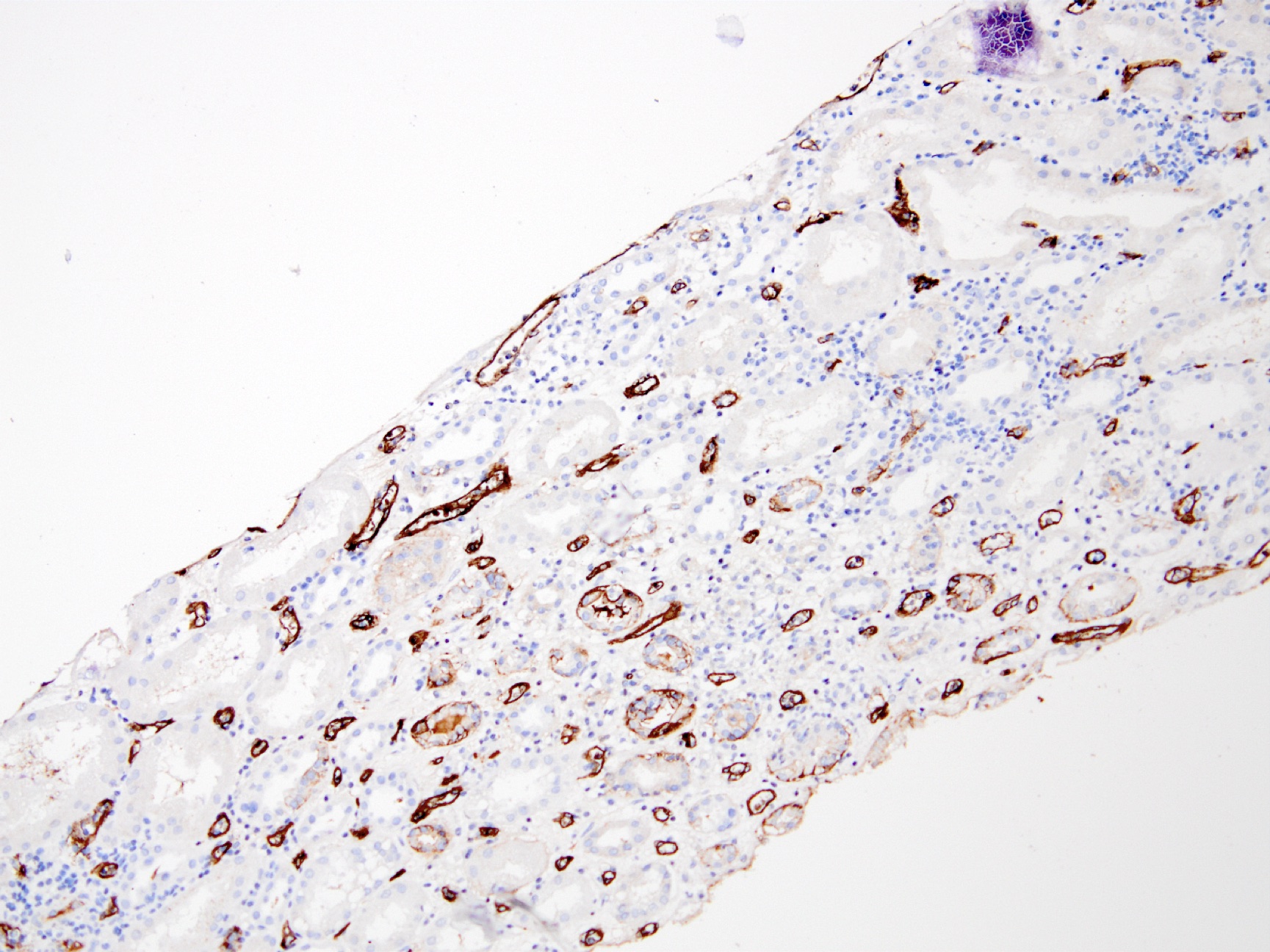
Microscopic (histologic) images
Contributed by Arzu Sağlam, M.D.
Immunofluorescence description
- No immunoglobulin or only nonspecific scarce staining
- C3 may be present along PTC
- C4d linear staining in peritubular capillaries (C4d2 or C4d3)
- References: Mod Pathol 2018;31:235, Transplantation 2018;102:1795
Positive stains (IHC and special stains)
- C4d linear staining in peritubular capillaries (C4d2 or C4d3 by immunofluorescence on frozen sections or C4d > 0 by immunohistochemistry on paraffin sections)
- This is an indirect sign of complement activation - deposition of the complement split product C4d
- May be negative in chronic active antibody mediated rejection (ABMR)
- Can also be present along glomerular capillary basement membrane in addition to peritubular capillaries or sometimes solely (in which case it is supportive of transplant glomerulopathy and chronic active ABMR)
- References: Mod Pathol 2018;31:235, Transplantation 2018;102:1795
Electron microscopy description
- Active antibody mediated rejection (ABMR):
- Glomerular changes resemble those of thrombotic microangiopathy
- Neutrophils, platelets and fibrin within glomerular capillary lumina
- Swelling of endothelial cells and widening of the subendothelial space
- Peritubular capillaries
- Neutrophils, platelets and fibrin within capillary lumina
- Swelling of endothelial cells and widening of the subendothelial space
- Apoptosis and fragmentation of endothelial cells
- Glomerular changes resemble those of thrombotic microangiopathy
- Most commonly used to diagnose chronic active ABMR:
- Early detection of transplant glomerulopathy - formation of new layer of glomerular basement membrane
- Multilayering of tubular basement membrane
- References: Mod Pathol 2018;31:235, Transplantation 2018;102:1795
Molecular / cytogenetics description
- Presence of antibody mediated rejection specific gene transcripts - endothelial damage associated transcripts (ENDAT) (Am J Transplant 2018;18:293, BMC Nephrol 2015;16:132)
- Of use in situations in which combination of histologic, immunohistochemical and serologic data remain equivocal for diagnosis of antibody mediated rejection
- Incorporated into the updated Banff classification; however, no specific recommendations are given regarding which molecular classifiers / transcript sets should be tested for or the platform(s) used to assess gene expression
- A 770 gene panel encompassing genes involved in rejection, tolerance, viral infections, innate and adaptive immune responses, the Banff Human Organ Transplant Panel (B-HOT), has been made commercially available and is going to be used on the NanoString platform for research purposes (Am J Transplant 2020;20:2305)
- A simplified molecular panel will hopefully be standardized and integrated into the Banff classification criteria in the near future
Sample pathology report
- Kidney, allograft biopsy:
- Microscopic description: Serial sectioning shows 26 glomeruli, 2 of which are globally sclerotic. Most noticeable change in the glomeruli is the presence of intracapillary inflammatory cells (g2) in most, among which neutrophils and monocytes / macrophages can be identified in addition to lymphocytes. Endocapillary or extracapillary proliferation, necrosis, intracapillary thrombi or double contouring of the capillary walls are absent (cg0). İnterstitium shows multifocal edema and inflammatory infiltrate, mostly confined to the lumen of the peritubular capillaries (ptc2, i0). The inflammatory cells are composed predominantly of mononuclear cells with fewer neutrophils. Tubulitis is scarce and when present only mild (t1). Tubular epithelial cells show flattening and reactive nuclear changes suggestive of mild acute tubular injury. Patchy narrow foci of IFTA can be identified, comprising about 5 - 10% of the cortical parenchyma (i-IFTA0, ci0, ct0). Immunohistochemical staining shows presence of diffuse linear C4d along peritubular capillaries and glomerular capillaries (C4d 3). SV40 is negative. Arteries display mild intimal fibrosis (cv1), arterioli show multifocal mild to moderate hyalinosis (ah2). Intimal arteritis not identified (v0).
- Immunofluorescence microscopy: 2 glomeruli observed.
- Anti IgG Ab: no deposits
- Anti IgA Ab: no deposits
- Anti IgM Ab: no glomerular deposits, blush segmental reactivity on walls of arterioli
- Anti C3 Ab: no glomerular deposits, segmental 1+ granular reactivity along ptc, blush segmental reactivity on walls of arterioli
- Anti c1q Ab: no deposits
- Anti kappa Ab: no glomerular deposits, 2+ staining of tubular casts
- Anti lambda Ab: no glomerular deposits, 2+ staining of tubular casts
- Diagnosis
- Kidney, allograft biopsy:
- Acute antibody mediated rejection, moderate peritubullary capillaritis and glomerulitis (ptc2, g2). Arterial intimal fibrosis (mild), arteriolar hyalinosis (ah2) (see comment)
- Comment: Biopsy lacks light microscopic changes suggestive of chronic damage, vascular changes are most probably donor related.
- Kidney, allograft biopsy:
Differential diagnosis
- Peritubular capillaritis (ptc) is also present in the following conditions:
- Acute tubular necrosis / injury (ATN / ATI): morphological distinction is not possible but absence of C4d staining is helpful; possibility of concurrent ATN / ATI and antibody mediated rejection
- Acute pyelonephritis: neutrophilic tubulitis and presence of neutrophil aggregates within tubuli are helpful but their absence does not rule out acute pyelonephritis, especially considering that the patients in question are immunosuppressed; absence of C4d staining is helpful
- BK nephritis: viral cytopathic changes in tubular epithelial cells and positive polyomavirus immunostain (should be performed on every allograft renal biopsy specimen)
- Thrombotic microangiopathy is also present in other conditions such as drugs (most notably acute cyclosporine toxicity), infections and recurrent thrombotic microangiopathy: absence of C4d staining is helpful
- C4d staining is also present in patients with ABO blood group incompatible (and less frequently in HLA incompatible) donors: biopsy specimen lacks morphological findings of microvascular inflammation and clinical evidence of graft dysfunction
- Transplant glomerulopathy is also present in the following conditions:
- Chronic thrombotic microangiopathy: capillaritis and C4d in ptc is lacking
- Recurrent / de novo immunocomplex glomerulonephritis (those with MPGN pattern): Immunofluorescence positivity helps; may also have C4d staining along the glomeruli but ptc staining is missing
- Transplant arteriopathy:
- Arteriosclerosis: no inflammatory cells in the vessel wall
- May also be a sign of T cell mediated rejection or both T cell mediated rejection and AMR
- Acute T cell mediated rejection: may frequently accompany AMR, presence of tubulitis and interstitial inflammatory infiltrate is seen; vascular lesions can be a feature of both entities
Additional references
Board review style question #1
Based on the photomicrograph of C4d immunohistochemistry, which of the following would be expected on the H&E stained sections?
- Intranuclear inclusions within tubular epithelial cells
- Neutrophil leucocytes within tubular lumens
- Normal glomeruli
- Presence of inflammatory cells within peritubular capillaries
- Severe tubulitis
Board review style answer #1
D. Presence of inflammatory cells within peritubular capillaries
Comment Here
Reference: Active / chronic active antibody mediated rejection
Comment Here
Reference: Active / chronic active antibody mediated rejection
Board review style question #2
Which of the following histological changes is suggestive of chronic ABMR?
- C4d positivity along peritubular capillaries
- Presence of glomerular basement membrane double contours
- Presence of inflammatory cells within glomerular capillaries
- Presence of inflammatory cells within peritubular capillaries
- Presence of inflammatory cells within the interstitium
Board review style answer #2
B. Presence of glomerular basement membrane double contours
Comment Here
Reference: Active / chronic active antibody mediated rejection
Comment Here
Reference: Active / chronic active antibody mediated rejection