Table of Contents
Definition / general | Essential features | Terminology | ICD coding | Epidemiology | Sites | Pathophysiology | Clinical features | Diagnosis | Laboratory | Radiology description | Radiology images | Prognostic factors | Case reports | Treatment | Clinical images | Gross description | Gross images | Microscopic (histologic) description | Microscopic (histologic) images | Cytology description | Positive stains | Negative stains | Electron microscopy description | Sample pathology report | Differential diagnosis | Additional references | Practice question #1 | Practice answer #1 | Practice question #2 | Practice answer #2Cite this page: Chapel DB, Husain AN. Peritoneal inclusion cyst (peritoneum). PathologyOutlines.com website. https://www.pathologyoutlines.com/topic/pleuraperitmulticysticmeso.html. Accessed September 21st, 2025.
Definition / general
- Peritoneal inclusion cyst(s) are comprised of one or more cystic spaces lined by bland mesothelial cells
- This is a broad diagnostic category of uncertain pathogenesis, ranging from small (0.1 cm) incidental unilocular cysts to florid (> 30 cm) symptomatic multilocular tumors
Essential features
- F > M
- Uniloculated or multiloculated cysts lined by bland mesothelial cells
- Complete resection is mainstay of treatment
- Recurrence rates range from 3% (in series enriched for smaller, unilocular lesions) to 50% (in series emphasizing florid, multilocular lesions) but death from disease is exceptionally rare in either scenario
- Pathogenesis (reactive versus neoplastic) is uncertain
- Knowledge is limited by shifting definitions and variable inclusion criteria in studies across time
Terminology
- WHO advocates for (multiloculated) peritoneal inclusion cyst(s) and discourages (benign) multicystic mesothelioma and (benign) cystic mesothelioma
ICD coding
- ICD-10: D19.1 - benign neoplasm of mesothelial tissue of peritoneum
Epidemiology
- Rare; accounts for 3 - 5% of peritoneal mesothelial lesions (Pleura Peritoneum 2019;4:20190024)
- 80 - 90% in women (Cancer 1982;50:1615, Am J Surg Pathol 1988;12:737, Hum Pathol 2003;34:369, Eur J Surg Oncol 2018;44:1100)
- Age at diagnosis is 11 - 77 years
- In women, most common in the third and fourth decades (Cancer 1982;50:1615, Am J Surg Pathol 1988;12:737, Hum Pathol 2003;34:369, J Pediatr Adolesc Gynecol 2009;22:41, Eur J Surg Oncol 2018;44:1100)
- In men, most common in the seventh decade (Hum Pathol 2003;34:369, Case Rep Pathol 2017;2017:9752908)
Sites
- Pelvis is the most common site (Am J Surg Pathol 1988;12:737, Cancer 1989;64:1336, Am J Clin Pathol 2021;155:853)
- Ovarian surface, peritoneal cul de sac and rectal serosa are frequently involved
- ~50% also involve the abdominal peritoneum (Am J Surg Pathol 1988;12:737, Cancer 1989;64:1336, Am J Clin Pathol 2021;155:853)
- Minority (~20%) also involve the retroperitoneum (Cancer 1989;64:1336)
- Rare cases involve splenic capsule or perinephric region (Am J Surg Pathol 1997;21:334, Arch Pathol Lab Med 2000;124:766)
Pathophysiology
- Unclear if neoplastic or reactive
- Factors favoring neoplastic pathogenesis (Am J Surg Pathol 1988;12:737, Eur J Surg Oncol 2018;44:1100)
- Progressive nature of untreated cases
- Rare transformation to malignant mesothelioma
- Inconstant association with prior surgery or inflammatory process
- Occasional co-occurrence with adenomatoid tumor or well differentiated papillary mesothelioma (Histopathology 1996;29:375, Curr Probl Cancer 2017;41:340, Ann Diagn Pathol 2019;38:43, Medicine (Baltimore) 2019;98:e15746)
- Factors favoring reactive pathogenesis (Cancer 1989;64:1336, Am J Surg Pathol 1986;10:844, Hum Pathol 2003;34:369)
- Frequent association with prior surgery, pelvic inflammatory disease, endometriosis or peritoneal dialysis
Clinical features
- May present with abdominal fullness, pain, urinary / bowel symptoms or palpable mass (Cancer 1982;50:1615)
- Up to half present as incidental finding (Am J Surg Pathol 1988;12:737, Cancer 1989;64:1336, Am J Clin Pathol 2021;155:853)
Diagnosis
- Laparoscopic exploration with tissue sampling (Cancer 1982;50:1615)
Laboratory
- CA125 may be mildly elevated (Case Rep Obstet Gynecol 2014;2014:852583, Obstet Gynecol Surv 2009;64:321)
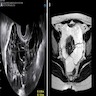
Radiology description
- Ultrasound: multiloculated anechoic cysts (Curr Probl Cancer 2017;41:340, Obstet Gynecol Surv 2009;64:321)
- CT: multilocular low density, thin walled multicystic mass (Tumori 2008;94:14, Obstet Gynecol Surv 2009;64:321)
- MRI: multiple cysts that are hypointense on T1, hyperintense on T2 (Clin Imaging 2017;42:133, Obstet Gynecol Surv 2009;64:321)
Prognostic factors
- Peritoneal recurrence rates range from 3% in a population based series with more frequent small unilocular cysts to 50% in a consultation heavy series enriched for florid multicystic lesions (Am J Clin Pathol 2021;155:853, Cancer 1982;50:1615, Cancer 1989;64:1336)
- Based on inclusion criteria for these studies, the true recurrence rate is likely closer to 3% than 50%, though data remains limited and the WHO (5th edition) retains the 50% figure
- Recurrence free interval may range from 6 months to 21 years (Cancer 1989;64:1336, Am J Clin Pathol 2021;155:853)
- Recurrences are frequently multiple (Cancer 1989;64:1336, Hum Pathol 2003;34:369)
- High peritoneal carcinomatosis index and incomplete resection are associated with increased recurrence risk (Cancer 1989;64:1336, Eur J Surg Oncol 2018;44:1100)
- In surgical series, complete cytoreduction with hyperthermic intraperitoneal chemotherapy has been associated with reduced recurrence risk (21% at 10 years) (Eur J Surg Oncol 2018;44:1100)
- Death from disease is extremely rare (Am J Surg Pathol 1988;12:737, Cancer 1989;64:1336, Hum Pathol 2003;34:369)
- Very rare cases present with or progress to epithelioid malignant mesothelioma (Am J Surg Pathol 1988;12:737, BMJ Case Rep 2014;2014:bcr2013200212, J Surg Oncol 2002;79:243)
Case reports
- 28 year old woman with peritoneal inclusion cyst of the pelvis (Obstet Gynecol Sci 2013;56:126)
- 47 year old woman with peritoneal inclusion cyst presenting as a pelvic mass (Obstet Gynecol Sci 2018;61:170)
- 47 year old woman peritoneal inclusion cyst of the pelvis (Int J Surg Case Rep 2020;74:152)
- 68 year old man with peritoneal inclusion cyst of the abdomen and pelvis (Case Rep Pathol 2017;2017:9752908)
- 84 year old man with peritoneal inclusion cyst (Diagn Interv Imaging 2016;97:361)
Treatment
- Early complete surgical resection is first line therapy (Am J Surg Pathol 1988;12:737, Eur J Surg Oncol 2018;44:1100)
- Recurrence(s) is managed by repeat resection
- Hyperthermic intraperitoneal chemotherapy is advocated by some for select clinical scenarios (Eur J Surg Oncol 2018;44:1100, Br J Surg 2011;98:60)
- Other options include hormonal therapy, sclerotherapy or laser ablation for select clinical scenarios (Korean J Radiol 2001;2:164, Curr Probl Cancer 2017;41:340)
- No role for radiotherapy or systemic chemotherapy
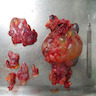
Gross description
- Uniloculated or multiloculated cystic lesion ranging from 0.1 cm to > 30 cm (Cancer 1982;50:1615, Cancer 1989;64:1336, Am J Clin Pathol 2021;155:853)
- In one population based study, ~66% were unifocal and unilocular while 33% were multifocal or multilocular (Am J Clin Pathol 2021;155:853)
- Multilocular lesions are larger than unilocular (average, 4.5 cm versus 2.0 cm, respectively)
- Occasional cases present with numerous discrete cysts studding the peritoneum (Am J Surg Pathol 1988;12:737)
- Rare cases present with free floating intraperitoneal cysts (Cancer 1989;64:1336)
- Individual locules range from 0.1 - 15 cm (Cancer 1982;50:1615, Cancer 1989;64:1336, Am J Surg Pathol 1988;12:737, Hum Pathol 2003;34:369)
- Pink-tan to green-gray to purple-red
- Cysts filled with serous fluid; intracystic hemorrhage is common
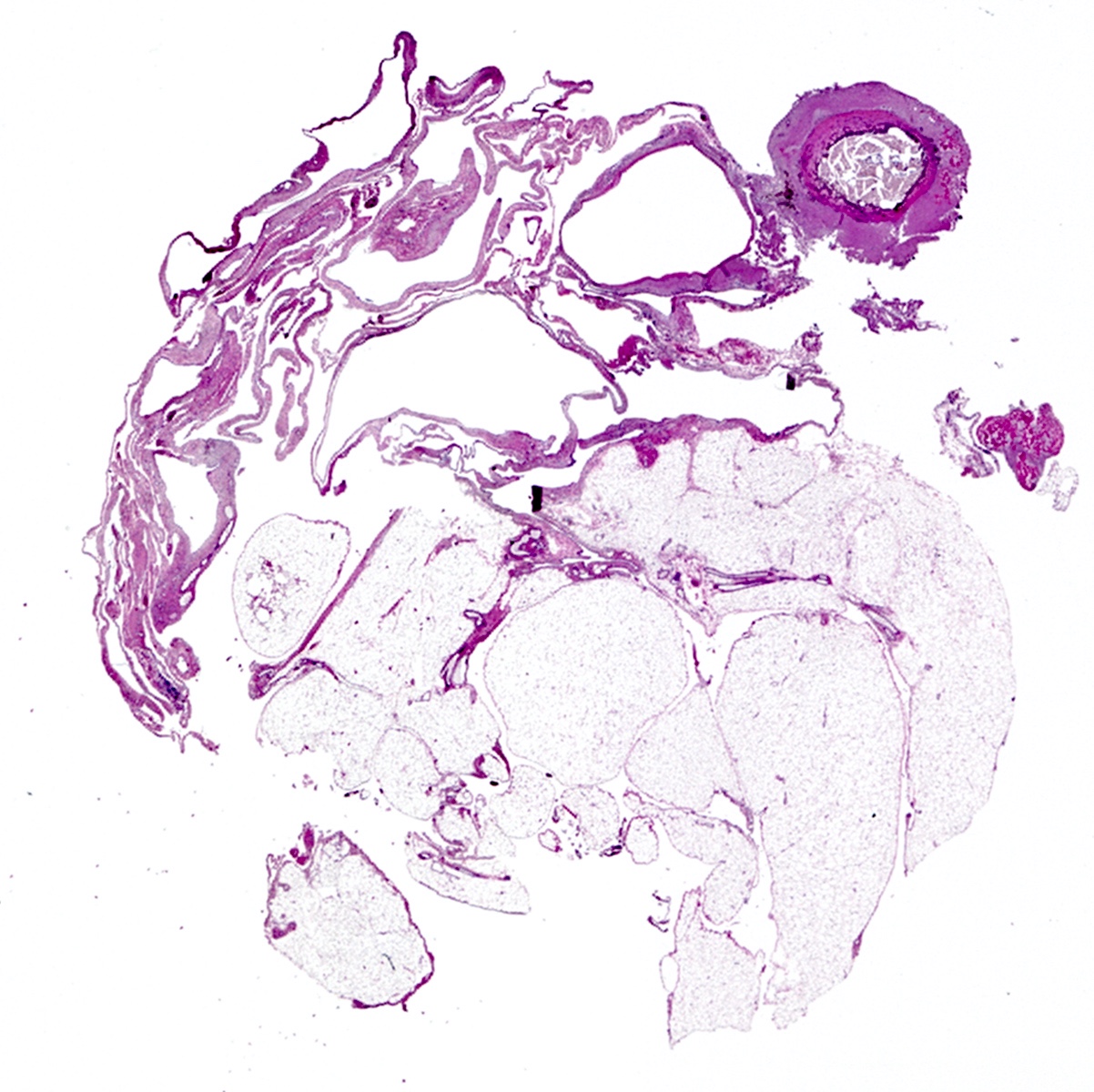
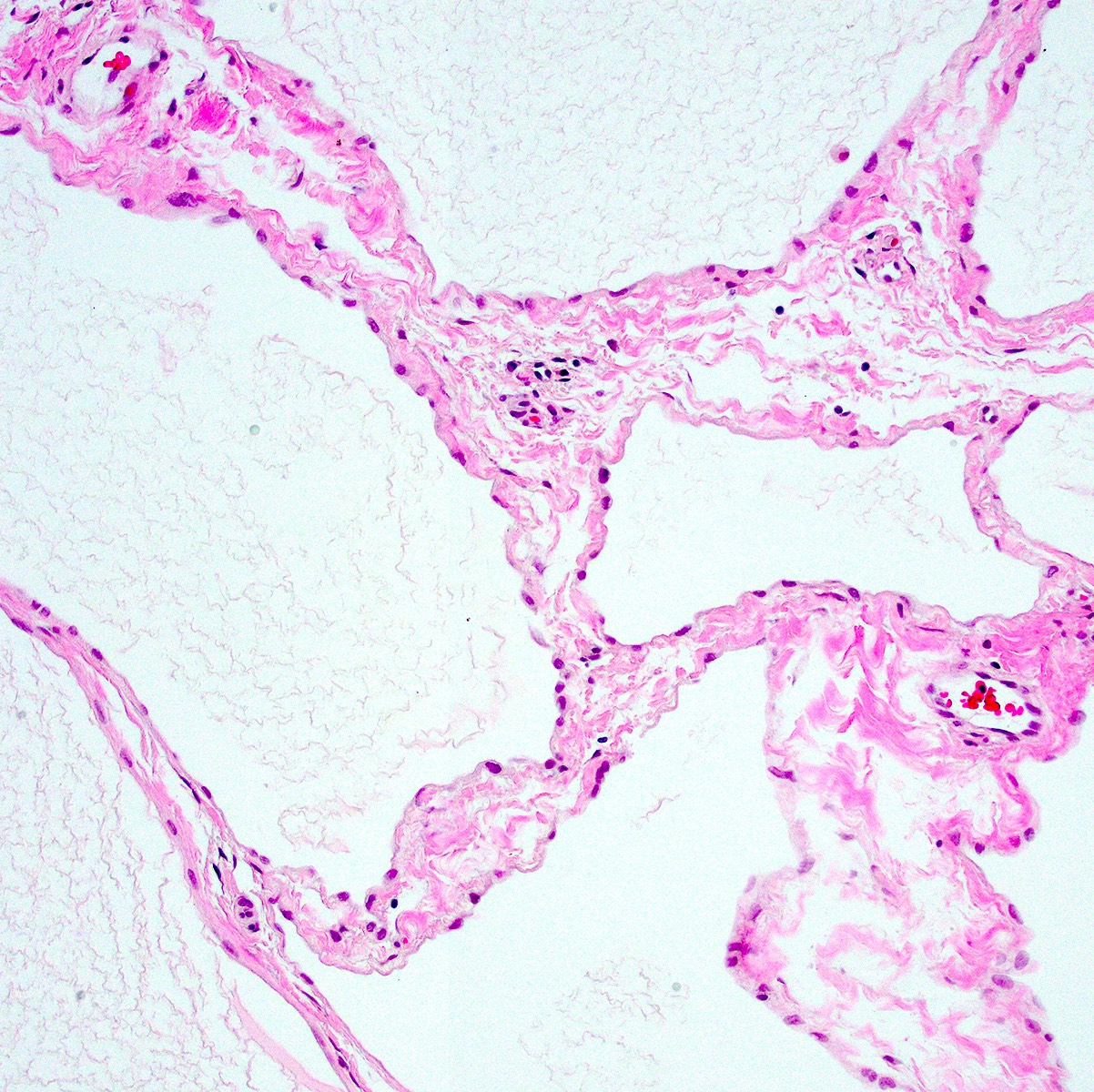
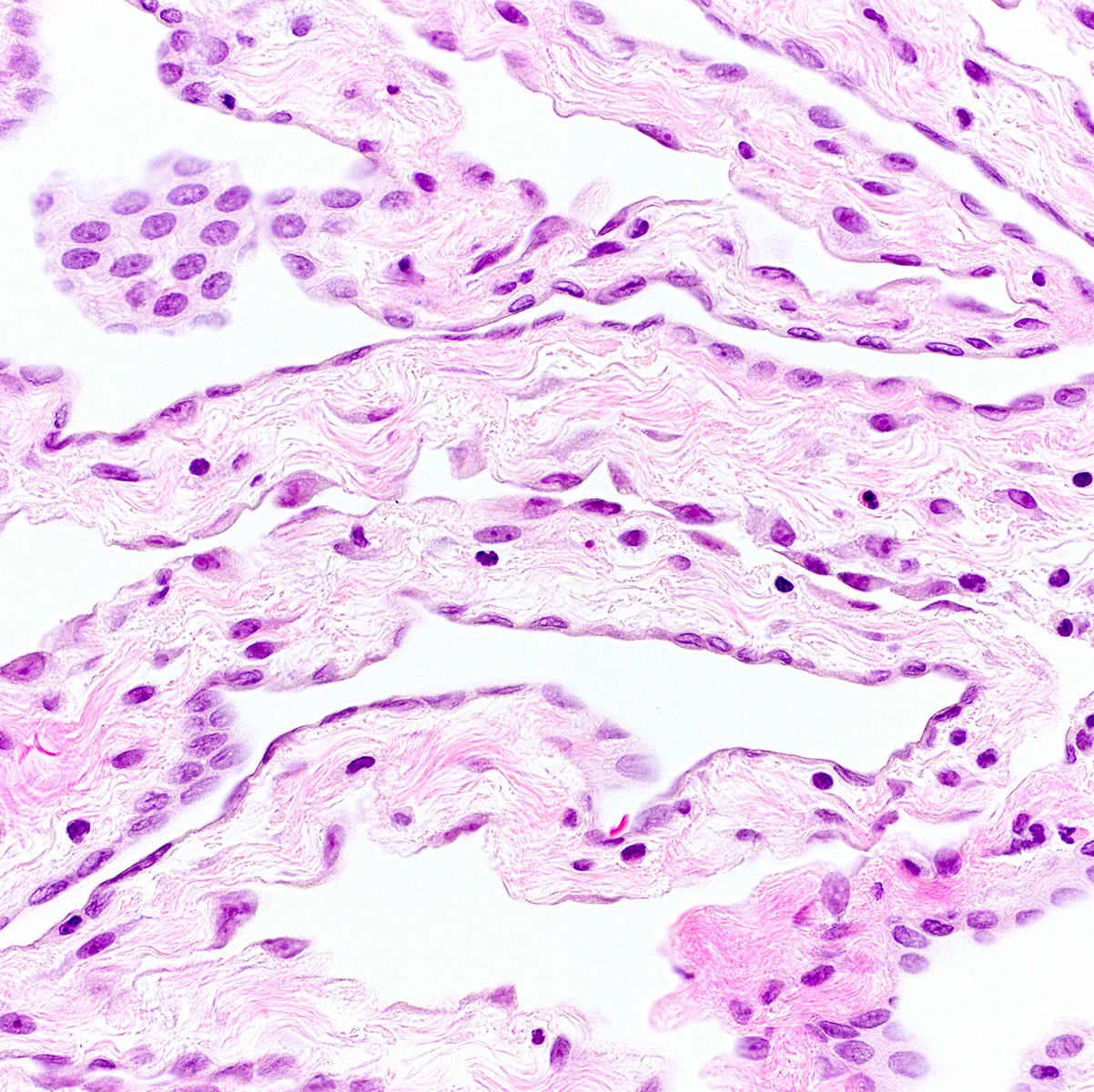
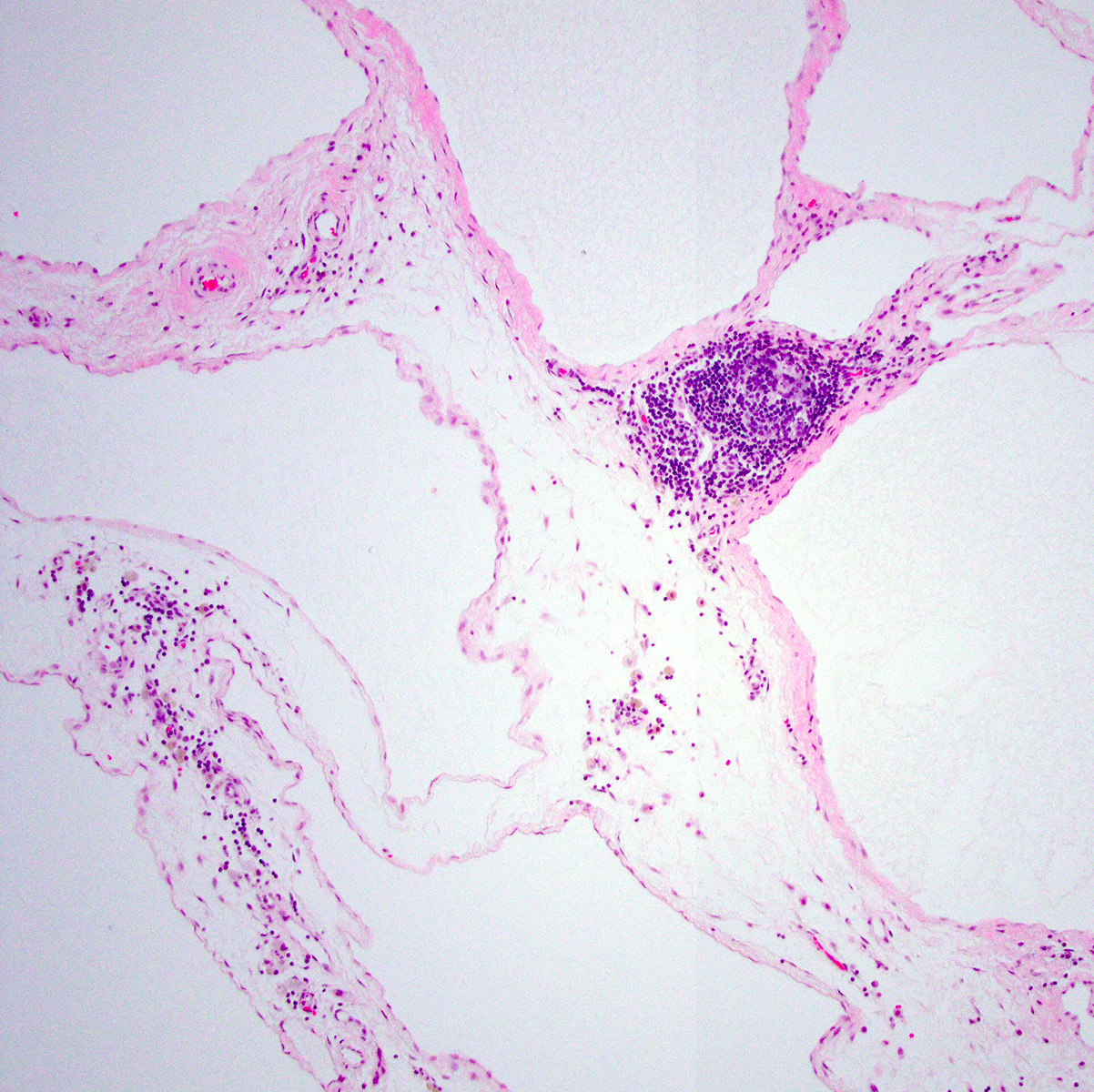
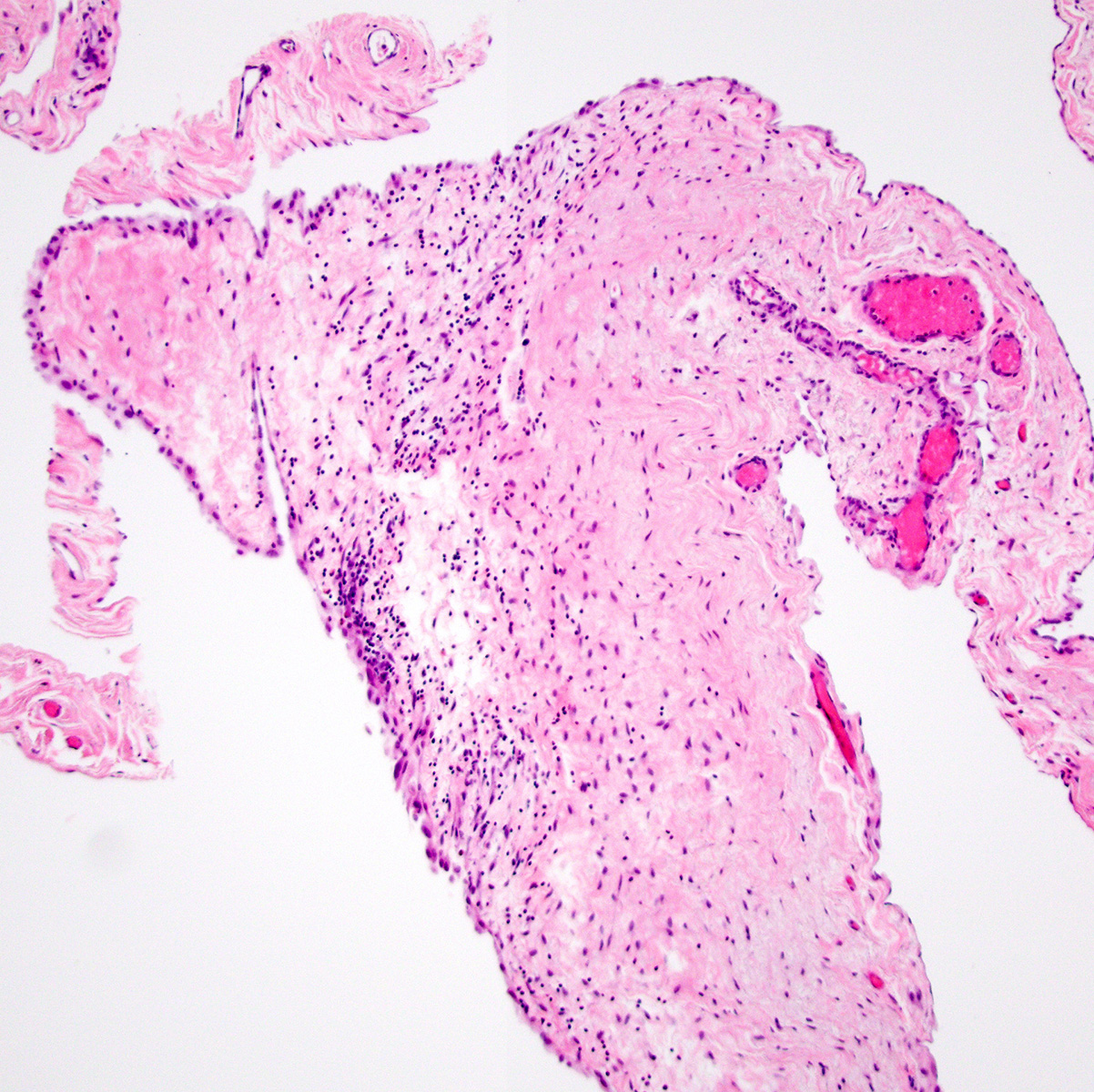
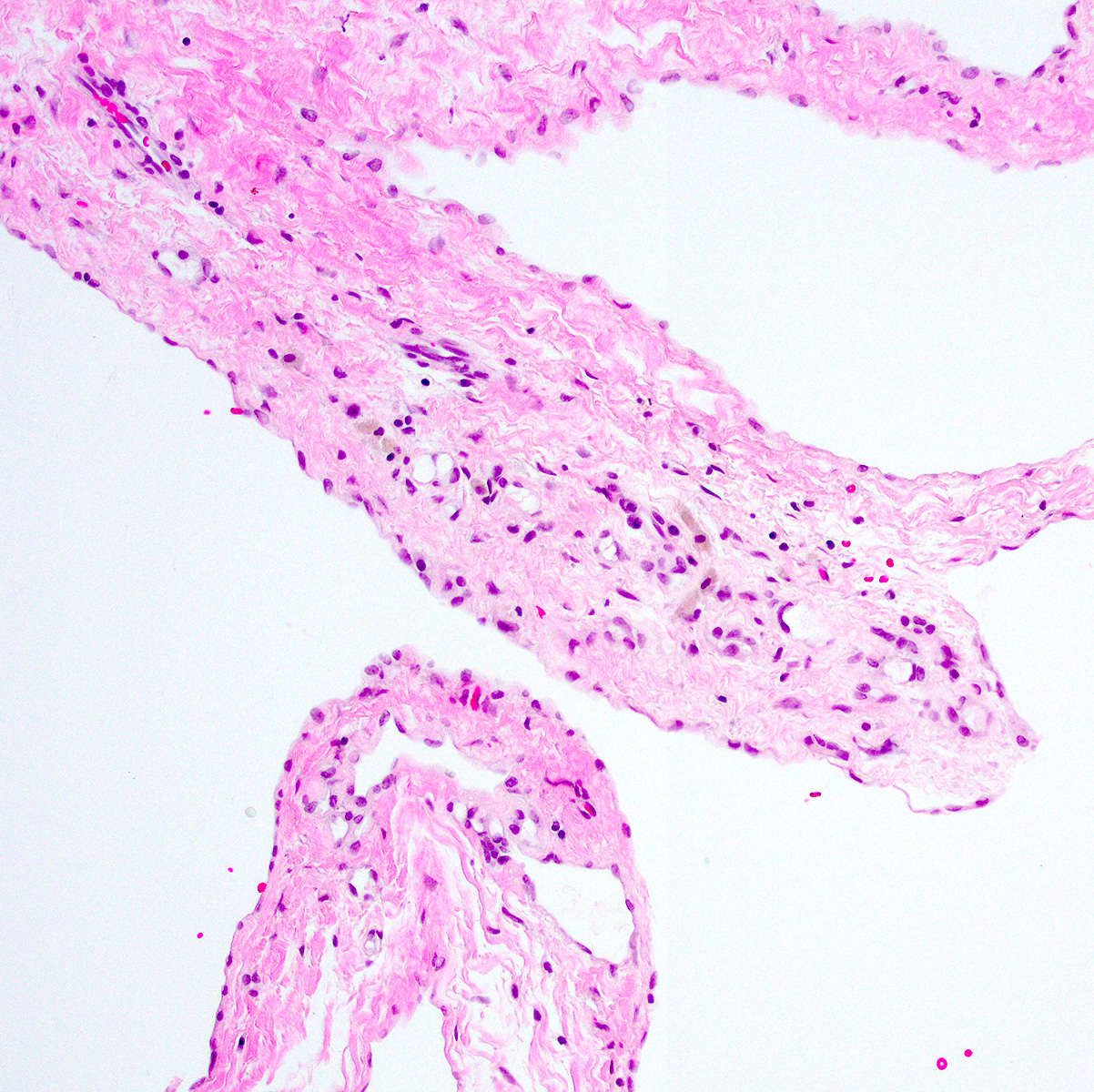
Microscopic (histologic) description
- 1 or multiple small cysts lined by a single layer of bland, flat to cuboidal cells
- Delicate mesothelial papillae or buds may project into cyst lumina (Am J Surg Pathol 1988;12:737, Cancer 1989;64:1336)
- Cysts separated by scant loose to collagenous stromal septa
- No infiltrative invasion of underlying tissues (e.g., fat, bowel)
- Chronic inflammation and hemorrhage are common
- Uncommon features (Am J Surg Pathol 1988;12:737)
- Adenomatoid tumor-like foci
- Squamous metaplasia of the mesothelial lining
- Hobnail change of mesothelial lining
- Brisk acute inflammation with mesothelial denudation and granulation type change
- Small mesothelial nests, cords and single cells in lesional stroma; often associated with brisk inflammation
- Mesothelial nests, cords and single cells may be seen in stromal septa; often associated with brisk inflammation (termed mural mesothelial proliferation) (Cancer 1989;64:1336, Am J Surg Pathol 1986;10:844, Hum Pathol 2003;34:369)
- Considered to be a superimposed reactive change
- No diffuse sheet-like growth or infiltration of underlying tissues
- Recurrences histologically similar to primary lesions (Cancer 1982;50:1615, Cancer 1989;64:1336)
Microscopic (histologic) images
Cytology description
- Nonspecific sheets or strips of flattened or squamoid cells with distinct cell borders and small bland nuclei (Diagn Cytopathol 2010;38:192, Cytopathology 2008;19:224, Obstet Gynecol Surv 2009;64:321)
Positive stains
- Cytokeratin
- Calretinin (Hum Pathol 2003;34:369, J Pediatr Adolesc Gynecol 2009;22:41)
- CK5/6
- WT1
- D2-40
- BAP1 (normal / retained)
- MTAP (normal / retained)
Negative stains
- ER, PR (focal weak staining in ~10%) (Hum Pathol 2003;34:369)
- Claudin4
- MOC31 / BerEP4
Electron microscopy description
- Well developed desmosomal attachments
- Numerous luminal microvilli
- Abundant cytoplasmic keratin filaments
- Ovoid nuclei with even chromatin
- Thin, intact basal lamina (Cancer 1982;50:1615, Cancer 1989;64:1336)
Sample pathology report
- Pelvic mass, excision:
- Multiloculated peritoneal inclusion cysts (6.5 cm) (see comment)
- Comment: On microscopic examination of this multiloculated cystic lesion, individual cysts are lined by bland, flattened mesothelial cells. Mitoses are not identified. The cysts are separated by fibrous septa with patchy chronic inflammation. There is no infiltration of underlying tissues. The gross and microscopic findings are most consistent with a diagnosis of multiloculated peritoneal inclusion cyst (previously termed multicystic mesothelioma). Although peritoneal inclusion cysts are generally regarded as benign, local recurrence has been documented in up to 50% of cases. Recurrence is more common after incomplete excision. Progression to malignant mesothelioma has been reported in exceptionally rare cases. Clinical and radiographic correlation is advised with appropriate clinical follow up.
Differential diagnosis
- Malignant mesothelioma:
- Lymphangioma (Cancer 1982;50:1615, Am J Surg Pathol 1988;12:737, Hum Pathol 2003;34:369):
- Unifocal lesion
- Primarily seen in in retroperitoneum, mesentery or omentum
- Uniform cyst size without intraluminal mesothelial budding
- Endothelial lining positive for CD31, CD34, ERG
- Negative for cytokeratin, calretinin, WT1
Additional references
Practice question #1
A 36 year old woman presents with pelvic discomfort, worsening over 6 months. Transvaginal ultrasound shows a multicystic lesion involving the cul de sac, left uterine adnexa and rectal serosa. The lesion is resected by laparoscopy. A representative H&E stained slide is shown. The cyst lining cells are positive for cytokeratin AE1 / AE3, calretinin and WT1 and negative for MOC31. The lesion is extensively sampled and no infiltrative growth is identified. Which of following is true?
- ~70% of such lesions harbor BAP1 mutations
- Complex copy number alterations are characteristic of such lesions
- Excision is curative; no clinical follow up is required
- Local recurrence affects ~50% of patients
- Men are affected ~4 times as often as women
Practice answer #1
D. Local recurrence affects ~50% of patients. This is a multiloculated peritoneal inclusion cyst. The published literature and WHO blue book indicate a local recurrence rate as high as 50% (answer D). Answer A is incorrect as peritoneal inclusion cysts lack BAP1 mutations; rather, ~70% of peritoneal mesotheliomas harbor BAP1 mutations. Answer B is incorrect because peritoneal inclusion cysts lack recurrent copy number alterations. The molecular underpinnings of these lesions remain uncertain. Answer C is incorrect as the potential for local recurrence (particularly in larger multiloculated lesions) warrants clinical follow up. Answer E is incorrect as peritoneal inclusion cysts predominantly affect women, not men.
Comment Here
Reference: Peritoneal inclusion cyst
Comment Here
Reference: Peritoneal inclusion cyst
Practice question #2
Which of the following statements regarding peritoneal inclusion cysts is true?
- ~20% of patients progress to malignant mesothelioma over 10 years
- 40 - 50% of lesions harbor homozygous deletion of CDKN2A
- > 90% of lesions express estrogen receptor and progesterone receptor
- Destructive infiltration of underlying structures (e.g., omental fat) is seen in ~33% of cases and does not affect prognosis
- Surgical excision is considered the mainstay of treatment
Practice answer #2
E. Surgical excision is considered the mainstay of treatment. Answer A is incorrect. Although the literature contains rare reports of apparent progression from peritoneal inclusion cyst to malignant mesothelioma, the rate of progression is much lower than 20%. Answer B is incorrect as peritoneal inclusion cysts lack CDKN2A deletions. Instead, ~40 - 50% of pleural mesotheliomas harbor CDKN2A deletion. Answer C is incorrect as ER and PR expression are rarely seen in peritoneal inclusion cysts. Answer D is incorrect as peritoneal inclusion cysts lack destructive invasion of underlying tissues. The presence of destructive invasion is consistent with a diagnosis of mesothelioma.
Comment Here
Reference: Peritoneal inclusion cyst
Comment Here
Reference: Peritoneal inclusion cyst