Table of Contents
Definition / general | Terminology | Epidemiology | Sites | Pathophysiology and etiology | Clinical features | Laboratory | Prognostic factors | Case reports | Treatment | Gross description | Gross images | Microscopic (histologic) description | Microscopic (histologic) images | Positive stains | Electron microscopy description | Molecular / cytogenetics description | Differential diagnosis | Additional referencesCite this page: Rane S. Gonadoblastoma. PathologyOutlines.com website. https://www.pathologyoutlines.com/topic/testismixed.html. Accessed April 20th, 2024.
Definition / general
- Rare benign gonadal tumor composed of germ cells and sex cord stromal derivatives resembling immature granulosa cells and Sertoli cells
Terminology
- First described by Scully (Cancer 1953;6:455, Cancer 1970;25:1340)
- Resembles normal developing gonad (hence the name)
Epidemiology
- Most cases occur within first 2 decades; nearly 94% by age 30
- Those with with partial dysgenesis / mosaicism are diagnosed prior to puberty; those with complete androgen insensitivity are diagnosed after puberty
- Almost always associated with disorders of sex development
- ~10% with dysgenetic gonads develop gonadoblastoma / seminoma by age 10 (Obstet Gynecol Surv 1986;41:74)
- ~25% with mixed gonadal dysgenesis and Y component develop gonadoblastoma and germ cell tumor by age 40 (Am J Obstet Gynecol 1976;124:293)
Sites
- Usually abdominal / inguinal undescended gonads / testis
- May occur in ovaries of phenotypically and genetically normal females (see ovary tumor chapter)
Pathophysiology and etiology
Disorders of sexual development (DSD) clearly associated with increased risk of gonadoblastoma include:
- Complete androgen insensitivity (46,XY) (30 - 66% incidence)
- Pure gonadal dysgenesis / Swyer syndrome (46,XY)
- Mixed gonadal dysgenesis (45,X / 46,XY) (55% incidence)
- Turner syndrome (45,XO) with XY mosaicism
- Molecular presence of Y chromosome (6 - 9% incidence) results in 43% incidence of gonadoblastoma
- Frasier syndrome (rare 46,XY DSD caused by mutation in WT1 gene)
- Maternal exposure to androgens, drugs, alcohol or illness during first trimester is associated with gonadal dysgenesis and intersex disorders; however, association with gonadoblastoma development is not established
Clinical features
- ~80% are phenotypic females with subtle abnormalities in sexual development; cases with pure gonadal dysgenesis can be completely normal females
- Patients with clitoromegally, abnormal hirsutism, etc. are candidates for karyotype analysis to exclude intersex disorders
- ~20% are phenotypic males with gynecomastia, hypospadias, cryptorchidism; usually 46XY or 45X / 46XY
- 1/3 have bilateral gonadoblastoma
- Commonly associated with primary amenorrhea, developmental delay of genitalia and secondary sexual characters
- 50% have abdominal mass with malignant transformation into seminoma or with metastatic disease
- Gonadoblastoma not identified during neonatal period may go undetected until puberty when patients present with primary amenorrhea
- Features of intersex disorders:
- Presence of inguinal hernia in phenotypically female neonates associated with male pseudohermaphrodites in 1.6%
- Bimanual abdominal palpation can be used to confirm presence of uterus in initial few days of neonatal life (due to effects of maternal hCG)
- Patients with complete androgen insensitivity / male pseudohermaphroditism not diagnosed during neonatal period usually go unnoticed until puberty
- Normal breast development but secondary sexual characters like pubic and axillary hair growth are rudimentary; vagina may be hypoplastic
- Patients with mixed gonadal dysgenesis (45XO / 46XY) have
- Ambiguous genitalia with varying degrees of phallic enlargement
- Undescended testis
- Urogenital sinus with labioscrotal fusion
- Nearly all have a uterus, vagina and fallopian tubes in addition to an ovary / streak and a contralateral testicle
- 50% are short and ~33% appear similar to individuals with Turner syndrome
Laboratory
- Karyotyping is most important investigation for identifying those with intersex disorders who are at increased risk of developing gonadoblastoma
- May have abnormalities in serum electrolytes, may be life threatening
- Hormonal evaluation is necessary to rule out other causes of abnormal sexual development and to plan hormonal replacement
Prognostic factors
- Excellent prognosis prior to malignant transformation, with no deaths reported
- 30% of patients have malignant germ cell tumor in dysgenetic or contralateral gonad
- Cure rates > 80% have been reported, including those with seminomatous overgrowth or metastases
Case reports
- 10 year old girl, Turner mosaic with Y chromosome material identified by restriction fragment analysis and unilateral microscopic gonadoblastoma (Am J Clin Pathol 1988;90:622)
- 12 year old girl with minute Y chromosome derived marker (J Med Genet 1992;29:542)
Treatment
- Some cases undergo spontaneous involution
- Treatment of choice is excision of gonadoblastoma and dysgenetic gonads prior to neoplastic transformation; however, timing of surgery varies with underlying disorder of sexual development
- For mixed gonadal dysgenesis and mosaicism, surgery recommended before puberty, as early as possible
- For complete androgen insensitivity (pure gonadal dysgenesis), surgery recommended after completion of puberty but before age 20:
- Allows development of female breast by peripheral conversion of testosterone by aromatase into estrogens
- Incidence of gonadoblastoma or other germ cell tumors before age 20 is negligible in these patients
- Patients are usually diagnosed at puberty when they present with primary amenorrhea
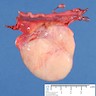
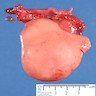
Gross description
- Tumor size varies from microscopic to 8 cm
- Yellow to tan nodules with gritty cut surface
- No hemorrhage or necrosis unless transformation to malignant germ cell tumor
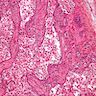
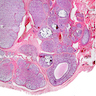
Microscopic (histologic) description
- Three growth patterns
- Most common: round / irregular clusters of immature Sertoli cells and germ cells surrounded by basement membranes, with Sertoli cells encircling rounded hyaline nodules
- Sertoli cells surround large germ cells
- Germ cells occupy center of nests with peripheral ring of Sertoli cells
- Stroma may contain large polygonal cells indistinguishable from Leydig cells: common in postpubertal patients
- Calcification common: may be focal involving hyaline bodies or extensive
- 50% have coexisting or subsequent seminoma
- 8 - 10% develop other germ cell malignancies (Am J Obstet Gynecol 1972;113:410)
Positive stains
- Germ cells express p53 (Mod Pathol 1997;10:1101), PLAP, CD117 / c-kit, VASA protein (Lab Invest 2002;82:159), testis specific Y protein encoded (Cytogenet Cell Genet 2000;91:160)
- Stromal cells express inhibin, WT1
Electron microscopy description
- Some cells show Charcot-Böttcher filaments of Sertoli cells
Molecular / cytogenetics description
- Germ cells are aneuploid (Histopathology 1997;30:177)
- Tsuchiya identified gonadoblastoma locus near Y centromere; TSPY gene is most likely candidate
- TSPY is expressed in adult spermatogonia and fetal gonocytes and has vital functions in male stem / germ cell proliferation
- It is ectopically expressed in gonadoblastoma, seminoma, ITGCN, some nonseminomatous germ cell tumors
- TSPY expression correlates with PLAP, c-kit, OCT3 / 4
- Seminomas and nonseminomatous tumors arising within dysgenetic gonad are usually diploid, unlike those arising from normal gonad (Genes Chromosomes Cancer 1999;25:134, Cancer Genet Cytogenet 1991;57:219, Cancer Res 1998;58:3105)
Differential diagnosis
- Sertoli cell tumor with nodules with entrapped germ cells in a may be mistaken for a gonadoblastoma
Additional references