Table of Contents
Definition / general | Essential features | Terminology | ICD coding | Epidemiology | Sites | Pathophysiology | Etiology | Clinical features | Diagnosis | Radiology description | Prognostic factors | Case reports | Treatment | Gross description | Gross images | Microscopic (histologic) description | Microscopic (histologic) images | Positive stains | Negative stains | Molecular / cytogenetics description | Sample pathology report | Differential diagnosis | Board review style question #1 | Board review style answer #1 | Board review style question #2 | Board review style answer #2Cite this page: Skala SL. Atypical polypoid adenomyoma. PathologyOutlines.com website. https://www.pathologyoutlines.com/topic/uterusapa.html. Accessed April 26th, 2024.
Definition / general
- Polypoid lesion composed of atypical, architecturally complex endometrial glands within benign myomatous / fibromyomatous stroma (Am J Surg Pathol 2015;39:1148)
Essential features
- Biphasic neoplasm composed of crowded atypical endometrial glands (with or without squamous morules) in myomatous / fibromyomatous stroma
- Stroma typically positive for SATB2, in contrast to most examples of myoinvasive carcinoma
- Risk of recurrence or concurrent or subsequent endometrial endometrioid carcinoma
Terminology
- Atypical polypoid adenomyofibroma
ICD coding
- ICD-11: XH7ZB1 - atypical polypoid adenomyoma
Epidemiology
- Usually diagnosed in women of reproductive age; range of 25 - 73 years (Am J Surg Pathol 1996;20:1)
- Associated with obesity, infertility, nulliparity
Sites
- Lower uterine segment > uterine corpus
Pathophysiology
- Prolonged estrogenic stimulation plays a role in some cases (Int J Gynecol Pathol 1987;6:104)
- Progesterone may have a role in development of the stroma of atypical polypoid adenomyoma (Hum Pathol 2014;45:33)
- Overlapping molecular and immunohistochemical features with atypical hyperplasia (Am J Surg Pathol 2015;39:1148)
Etiology
- Unknown
Clinical features
- Abnormal vaginal bleeding
- Incidental finding during evaluation of infertility
- May occur in the setting of Turner syndrome (Int J Gynecol Pathol 1987;6:104)
- Rare report of occurrence in patient with Cowden syndrome (Gynecol Oncol Case Rep 2012;2:29)
- Reference: Pathol Res Pract 2019;215:766
Diagnosis
- Based on histologic features
Radiology description
- T2 weighted MRI images: slightly hyperintense polypoid mass mixed with markedly hyperintense foci corresponding to endometrial glands
- T1 weighted MRI images: isointense with myometrium, occasional hyperintense cystic foci
- Solid portions, other than cystic foci, show contrast enhancement in the arterial phase and washout or plateau pattern in the late phase
- Reference: J Comput Assist Tomogr 2015;39:32
Prognostic factors
- Approximately 30 - 44% recur after conservative treatment; higher risk of recurrence if sampling is not under hysteroscopic visualization (Int J Gynaecol Obstet 2021 Dec 18 [Epub ahead of print], Medicine (Baltimore) 2020;99:e20491)
- Concurrent diagnosis of endometrial endometrioid carcinoma in ~11%; diagnosis of endometrial carcinoma during follow up in ~14% (Medicine (Baltimore) 2020;99:e20491)
- High likelihood of successful pregnancy after conservative treatment (Medicine (Baltimore) 2020;99:e20491)
Case reports
- 29 year old woman with low grade endometrioid adenocarcinoma arising from atypical polypoid adenomyoma; diagnosed in early pregnancy and managed conservatively (Gynecol Oncol Rep 2019;28:101)
- 35 year old woman with atypical polypoid adenomyoma treated by hysteroscopy with photodynamic diagnosis using 5-aminolevulinic acid (Photodiagnosis Photodyn Ther 2019;27:295)
- 36 year old woman with endometrioid adenocarcinoma developing 8 years after conservative management for atypical polypoid adenomyoma (Gynecol Oncol Case Rep 2014;8:21)
- 45 year old woman with atypical polypoid adenomyoma treated with local resection (Pathologica 2020;112:214)
Treatment
- Conservative management and close follow up can be considered for women that desire fertility preservation; addition of progestin does not seem to improve oncologic outcomes (Int J Gynaecol Obstet 2021 Dec 18 [Epub ahead of print])
- One proposed strategy: transvaginal ultrasonography plus hysteroscopic biopsy every 3 months for 2 years, every 4 - 6 months for another 3 years and then once a year (Arch Gynecol Obstet 2016;293:177)
- Small case series suggests a potential role for photodynamic therapy to ensure complete resection (Photodiagnosis Photodyn Ther 2021;36:102583)
- Hysterectomy, if peri or postmenopausal (Int J Gynaecol Obstet 2021 Dec 18 [Epub ahead of print])
Gross description
- Firm polypoid lesion
- Typically unifocal
- Average size of 2 cm
- Reference: Am J Clin Pathol 1986;86:139
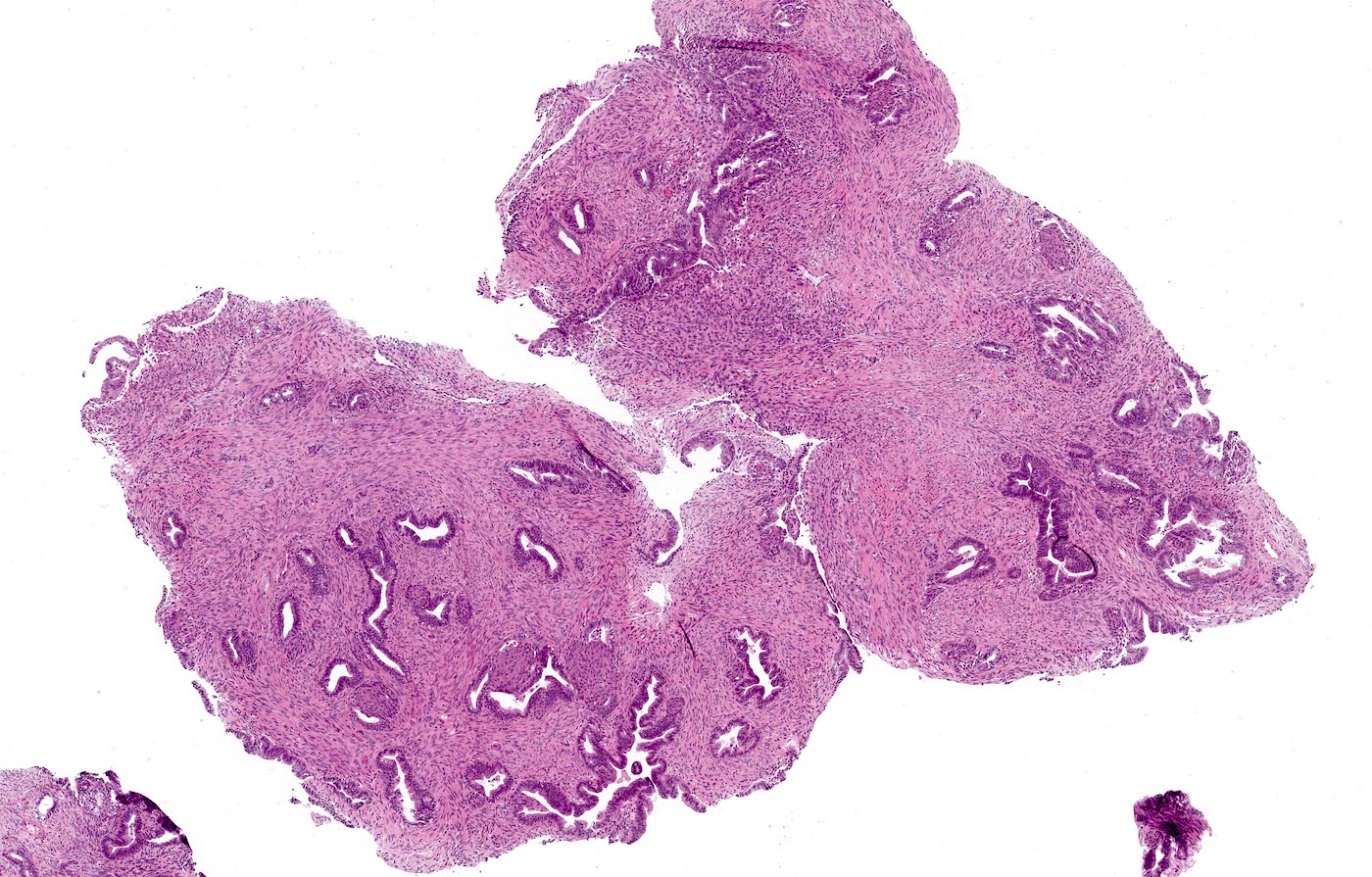
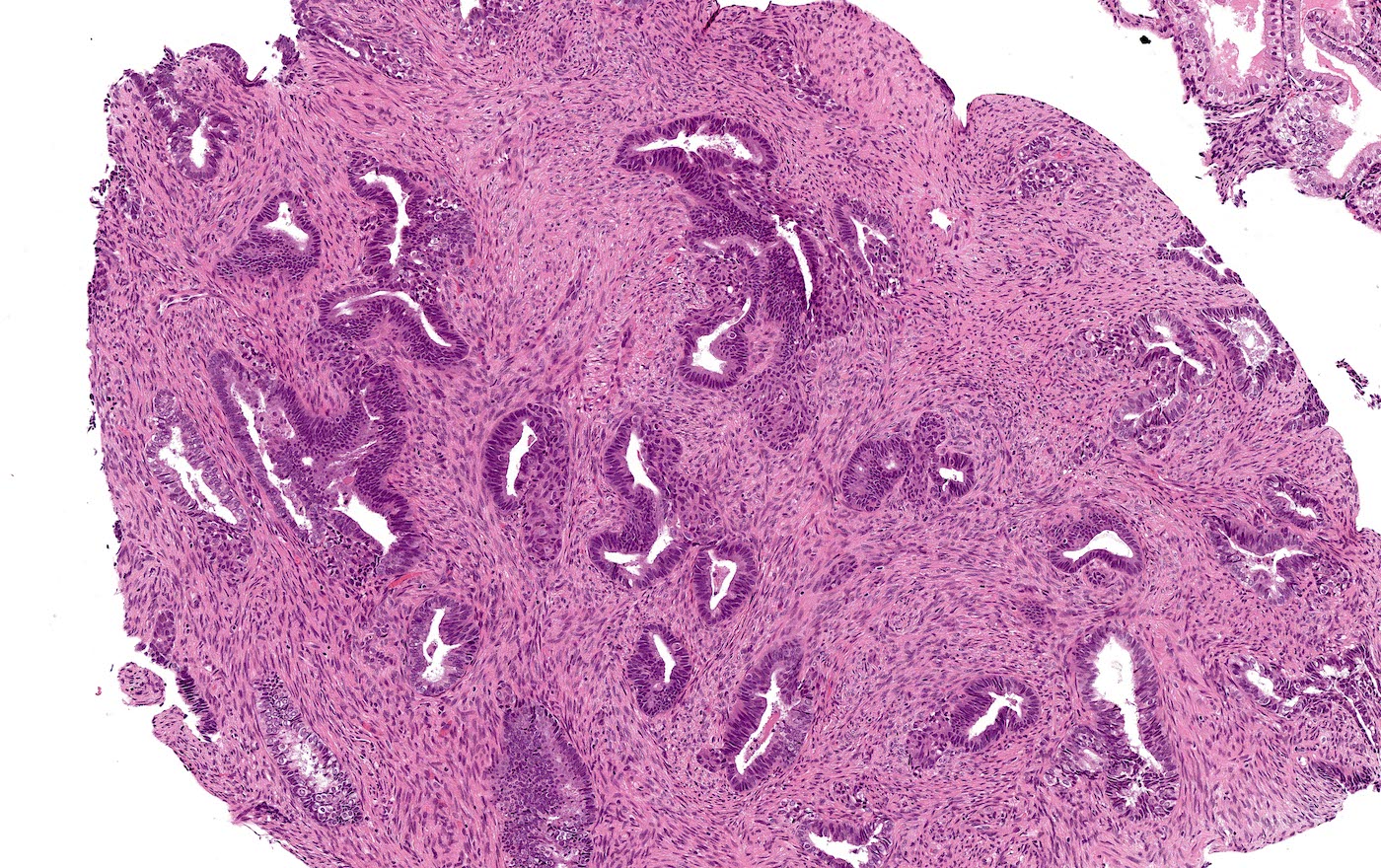
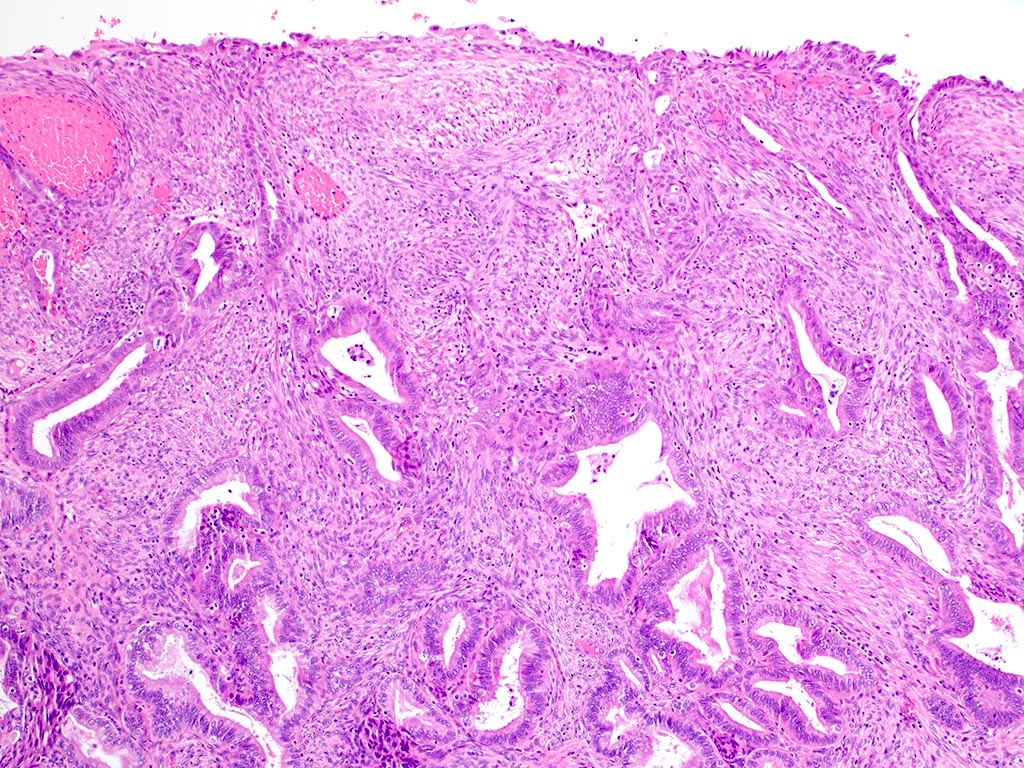
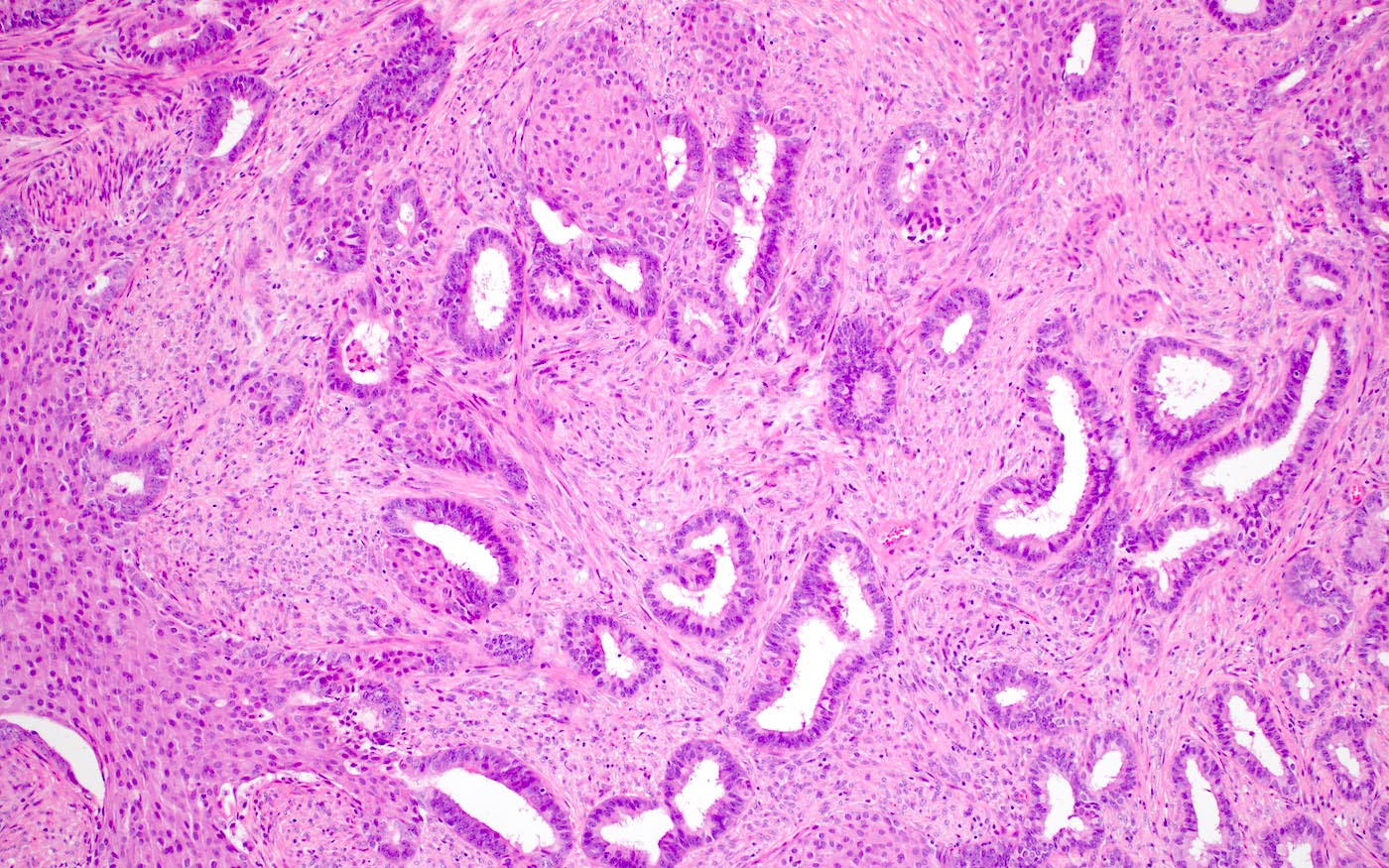
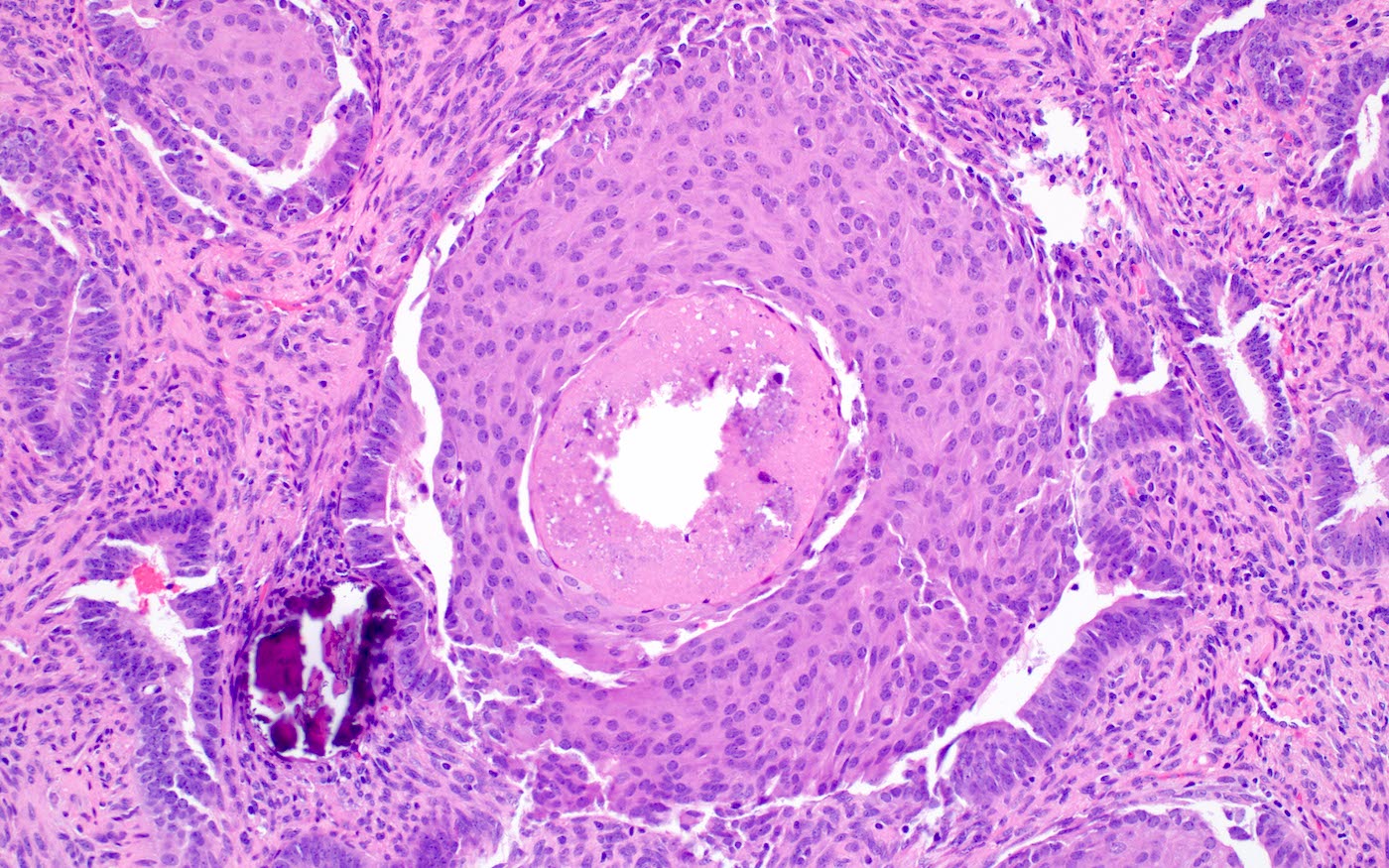
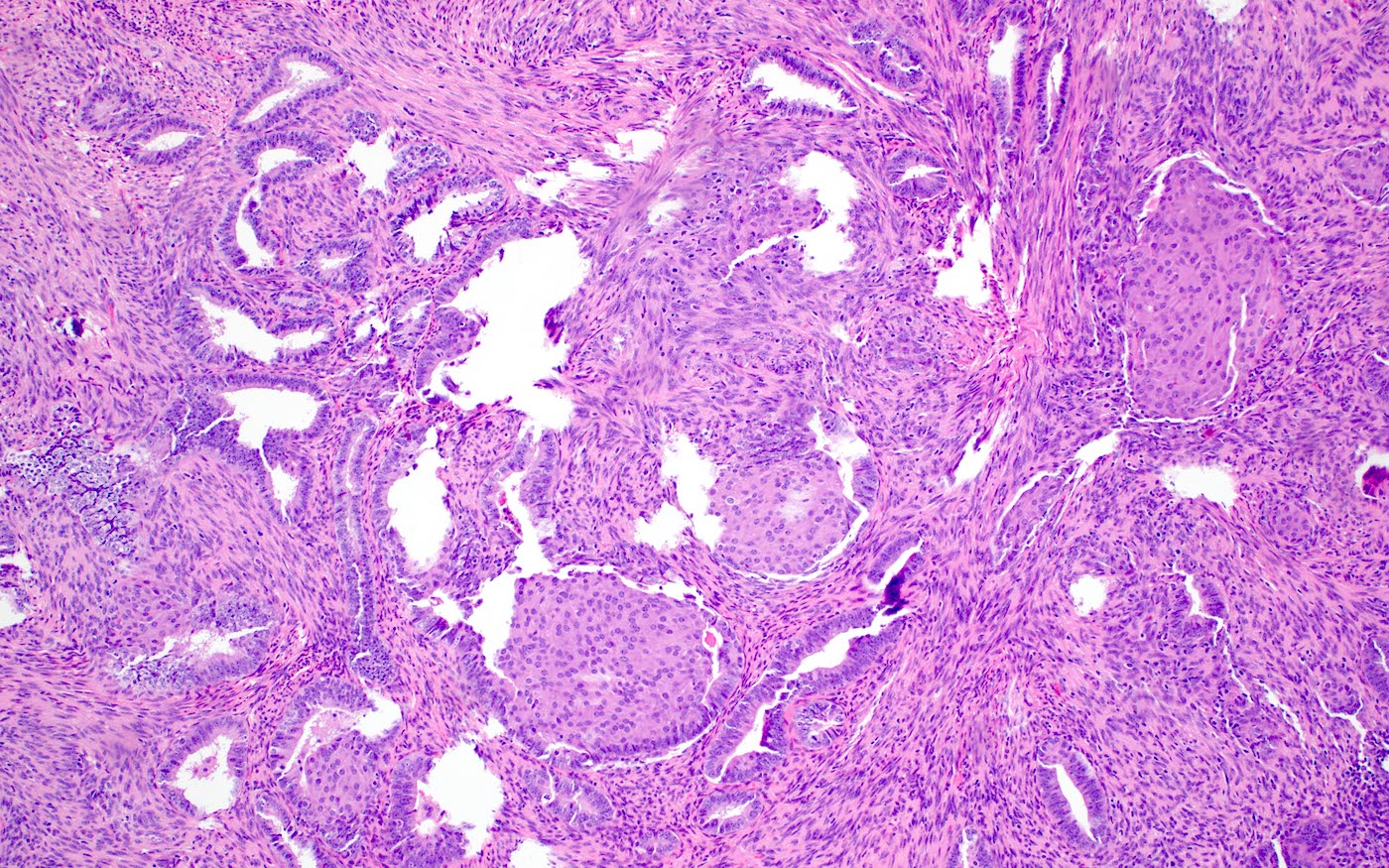
Microscopic (histologic) description
- Biphasic tumor:
- Endometrioid glands, usually with complex architecture and sometimes with cytologic atypia
- Glandular component often shows lobulated architecture
- Benign fibromyomatous stroma, rarely with myxoid change
- Squamous morular metaplasia is frequently seen and may show central necrosis
- Well circumscribed or slightly irregular margin
- Reference: Am J Surg Pathol 2015;39:1148
Microscopic (histologic) images
Positive stains
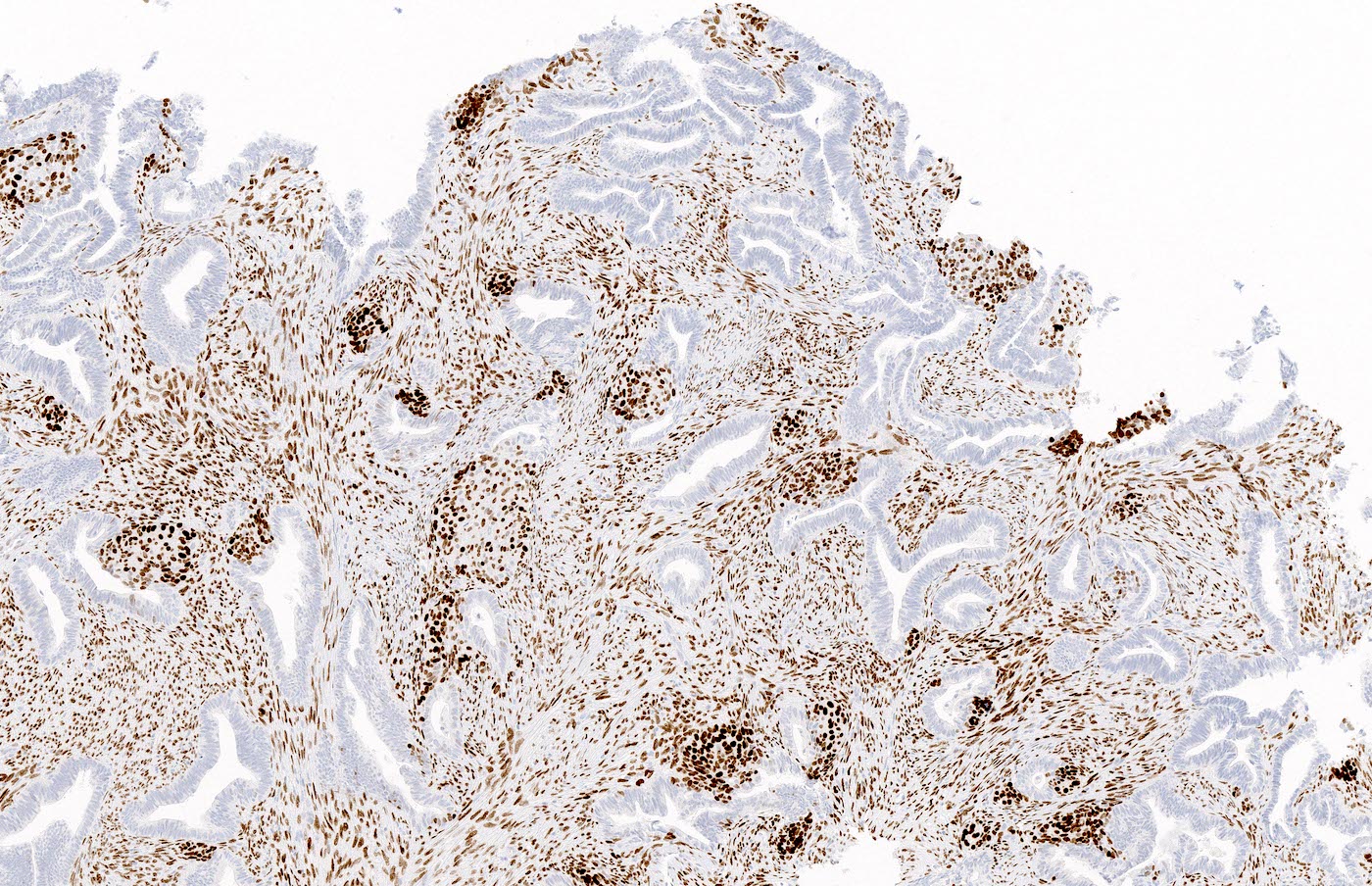
- Beta catenin nuclear positivity (squamous morules > glands) (Hum Pathol 2014;45:33)
- SATB2 reactivity in the stromal component (Histopathology 2021;79:96, Int J Gynecol Pathol 2019;38:397)
- Pancytokeratin, estrogen receptor and progesterone receptor typically positive in glands
- Smooth muscle actin and desmin positive in stromal muscle (Appl Immunohistochem Mol Morphol 2020;28:646)
- CDX2 and p16 positive in squamous morules (Appl Immunohistochem Mol Morphol 2020;28:646, Histopathology 2021;79:96)
Negative stains
- CD10 and caldesmon negative in stroma (Appl Immunohistochem Mol Morphol 2020;28:646)
Molecular / cytogenetics description
- CTNNB1 mutation, PTEN deletion and KRAS mutation (Am J Surg Pathol 2015;39:1148, Hum Pathol 2014;45:33)
- MLH1 promoter hypermethylation and microsatellite instability (Hum Pathol 2003;34:784)
Sample pathology report
- Uterus, total hysterectomy:
- Atypical polypoid adenomyoma
Differential diagnosis
- Endometrial atypical hyperplasia (complex atypical hyperplasia / endometrioid intraepithelial neoplasia):
- Lacks muscular stroma
- Involves flat endometrium or polyps without myomatous stroma
- Myoinvasive endometrioid carcinoma:
- Fragments of myometrium with invasive carcinoma often admixed with separate fragments of endometrioid carcinoma
- May show desmoplasia
- Muscular stroma negative for SATB2 by immunohistochemistry
- Endometrial polyp:
- Lacks muscular stroma
- Some cases may show endometrial hyperplasia with or without squamous morules
- Adenosarcoma:
- Benign endometrial glands and malignant stroma
- Carcinosarcoma:
- Malignant endometrial glands and stroma
Board review style question #1
The tumor above is seen in endometrial curettings from a 36 year old patient. Which of the following statements is true?
- Atypical polypoid adenomyoma can progress to endometrioid carcinoma
- Atypical polypoid adenomyoma is associated with TP53 mutations
- Comedonecrosis of squamous morules implies poor prognosis
- Hysterectomy is always the first line treatment for atypical polypoid adenomyoma
- Progestin typically leads to regression of atypical polypoid adenomyoma
Board review style answer #1
A. Atypical polypoid adenomyoma can progress to endometrioid carcinoma
Comment here
Reference: Atypical polypoid adenomyoma
Comment here
Reference: Atypical polypoid adenomyoma
Board review style question #2
Patients with atypical polypoid adenomyoma that are diagnosed on endometrial biopsy are at an increased risk for which of the following?
- Carcinosarcoma
- Clear cell carcinoma
- Endometrioid carcinoma
- Endometriosis
- Serous carcinoma
Board review style answer #2