Table of Contents
Definition / general | Essential features | Terminology | ICD coding | Epidemiology | Sites | Pathophysiology | Etiology | Clinical features | Diagnosis | Laboratory | Prognostic factors | Treatment | Clinical images | Gross description | Gross images | Microscopic (histologic) description | Microscopic (histologic) images | Cytology description | Positive stains | Videos | Sample pathology report | Differential diagnosis | Additional references | Practice question #1 | Practice answer #1 | Practice question #2 | Practice answer #2 | Practice question #3 | Practice answer #3Cite this page: Karamchandani DM. Barrett related dysplasia. PathologyOutlines.com website. https://www.pathologyoutlines.com/topic/esophagusdysplasia.html. Accessed September 15th, 2025.
Definition / general
- Unequivocally neoplastic epithelium without invasion, associated with Barrett esophagus (WHO: Digestive System Tumours, 5th Edition, 2019)
Essential features
- Presence of Barrett esophagus related dysplasia remains greatest risk factor for development of esophageal adenocarcinoma (Am J Surg Pathol 2017;41:e8)
- Pathologic diagnosis and grading of dysplasia is the gold standard marker for assessing risk of neoplastic progression (Am J Surg Pathol 2016;40:e83)
- Atypia in Barrett esophagus dysplasia can be interpreted as negative for dysplasia, indefinite for dysplasia, low grade dysplasia and high grade dysplasia (WHO: Digestive System Tumours, 5th Edition, 2019, Hum Pathol 1988;19:166)
- Diagnosis and grading of Barrett esophagus related dysplasia can be challenging, especially with coexisting inflammation (Hum Pathol 1988;19:166)
- Both low grade dysplasia and high grade dysplasia managed by endoscopic therapy, per the latest American College of Gastroenterology (ACG) guidelines (Am J Gastroenterol 2016;111:30)
Terminology
- Indefinite for dysplasia, low grade Barrett dysplasia and high grade Barrett dysplasia (WHO: Digestive System Tumours, 5th Edition, 2019, Hum Pathol 1988;19:166)
- Vienna classification uses terms noninvasive low grade neoplasia (low grade adenoma / dysplasia) for low grade dysplasia and noninvasive high grade neoplasia for high grade dysplasia (Gut 2000;47:251)
- World Health Organization recognizes both classifications (WHO: Digestive System Tumours, 5th Edition, 2019)
ICD coding
Epidemiology
- Dysplasia develops typically in the setting of Barrett esophagus (Am J Gastroenterol 2016;111:30)
- Barrett esophagus detected in ~ 10- 15% of patients with chronic gastroesophageal reflux disease (GERD) (Am J Gastroenterol 2016;111:30, Gastroenterol Hepatol (NY) 2016;12:449)
- Incidence of low grade dysplasia should be ~2 - 3% but not > 5% in prospectively evaluated patients (Am J Gastroenterol 2010;105:1523)
- Indefinite for dysplasia category should be used in only a small percentage of cases (< 3 - 5%) (Ann Diagn Pathol 2018;37:75)
- For low grade dysplasia, the annual risk of progression to cancer is ~ 0.7% per year (Am J Gastroenterol 2016;111:30)
- For high grade dysplasia, the annual risk of progression to cancer is ~ 7% per year (Am J Gastroenterol 2016;111:30)
Sites
- Distal esophagus, gastroesophageal junction (restricted to metaplastic esophageal mucosa)
Pathophysiology
- Sequential progression from inflammation to metaplasia, dysplasia and carcinoma (Am J Surg Pathol 2016;40:e45)
- Accumulation of multiple genetic and epigenetic alterations causes development and progression of dysplasia (Nat Genet 2014;46:837)
- C-myc and cyclins D1, E and B implicated as oncogenes in neoplastic progression in Barrett esophagus (Am J Surg Pathol 2016;40:e45)
- Inactivation of tumor suppressor proteins p53, p16, p15, p27 and adenomatous polyposis coli (APC) also implicated in Barrett esophagus carcinogenesis (Am J Surg Pathol 2016;40:e45)
- Other mechanisms proposed include increased telomerase expression, increased VEGFA and C, decreased membrane E-cadherin, increased MMP-7 and MMP-9, increased markers of epithelial-mesenchymal transition such as 2EB1/2EB2 and TGF-B1 (Am J Surg Pathol 2016;40:e45)
- As Barrett esophagus epithelial cells progress to cancer, they typically manifest aneuploidy, a marker of genomic instability (Am J Surg Pathol 2016;40:e45)
Etiology
- Known risk factors for development of neoplasia in Barrett esophagus (Am J Gastroenterol 2016;111:30):
- Advancing age
- Increasing length of Barrett esophagus
- Central obesity
- Tobacco usage
- Lack of nonsteroidal anti inflammatory agent use
- Lack of PPI use
- Lack of statin use
Clinical features
- Patients typically have gastroesophageal reflux disease symptoms
- No distinct clinical or radiologic manifestations of Barrett esophagus dysplasia
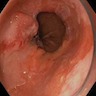
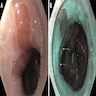
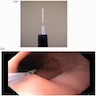
- Endoscopically, Barrett esophagus dysplasia may be visible as thickened, flat, irregular or plaque-like area, distinct from adjacent nondysplastic Barrett mucosa (Dig Dis Sci 2003;48:1537, Am J Gastroenterol 2016;111:30)
- Mucosal abnormalities (ulceration, stricture, mass, nodules, plaques) associated with increased risk of cancer (Am J Gastroenterol 2016;111:30)
Diagnosis
- Recommended for Barrett esophagus with dysplasia to be reviewed by 2 pathologists, at least 1 with specialized expertise in GI pathology (Am J Gastroenterol 2016;111:30)
- Barrett esophagus surveillance performed with high definition / high resolution white light endoscopy
- Routine use of advanced imaging techniques other than electronic chromoendoscopy not recommended for endoscopic surveillance at this time
- Endoscopic surveillance should employ 4 quadrant biopsies at 1 cm intervals in patients with prior dysplasia (Am J Gastroenterol 2016;111:30)
- Mucosal abnormalities to be sampled separately, preferably with endoscopic mucosal resection (EMR)
- Adjunct use of wide area transepithelial sampling with computer assisted 3 dimensional analysis (WATS) to forceps biopsy markedly improves detection of esophageal dysplasia (United European Gastroenterol J 2018;6:529)
- Overall detection of dysplasia by WATS reported to be increased by 242% (Dis Esophagus 2019;32:doy099)
Laboratory
- No laboratory tests assist in the detection of Barrett esophagus dysplasia
Prognostic factors
- Diagnosis of Barrett esophagus associated dysplasia remains a marker of increased risk of progression to esophageal adenocarcinoma (Am J Surg Pathol 2017;41:e8, Am J Surg Pathol 2016;40:e45)
- Progression rates of low grade dysplasia to high grade dysplasia and dysplasia to carcinoma directly proportional to number of pathologists who agree on dysplasia diagnosis (Am J Surg Pathol 2016;40:e45, Am J Gastroenterol 2007;102:483)
Treatment
- Indefinite for dysplasia:
- Repeat endoscopy after acid suppressive medication optimization for 3 - 6 months
- If indefinite for dysplasia is reconfirmed, surveillance interval of 12 months is recommended (Am J Gastroenterol 2016;111:30, Gastroenterol Hepatol (NY) 2016;12:449)
- Low grade dysplasia:
- Endoscopic eradication therapy preferred treatment modality, endoscopic surveillance every 12 months an acceptable alternative (Am J Gastroenterol 2016;111:30)
- High grade dysplasia:
- Endoscopic therapy (Am J Gastroenterol 2016;111:30, Gastroenterol Hepatol (NY) 2016;12:449)
- Nodular Barrett esophagus: recommend endoscopic mucosal resection of the nodular lesion as initial diagnostic and therapeutic maneuver (Am J Gastroenterol 2016;111:30)
- If endoscopic mucosal resection confirms high grade dysplasia, endoscopic ablative therapy of remaining Barrett esophagus (Am J Gastroenterol 2016;111:30)
- Nonnodular dysplastic Barrett esophagus: radiofrequency ablation currently preferred endoscopic ablative therapy (Am J Gastroenterol 2016;111:30)
- In patients with preablation low grade dysplasia, endoscopic surveillance recommended every 6 months in the first year following complete elimination of intestinal metaplasia (CIEM) and annually thereafter (Am J Gastroenterol 2016;111:30)
- Endoscopic surveillance following complete elimination of intestinal metaplasia for patients with preablation high grade dysplasia recommended every 3 months for the first year, every 6 months in the second year and annually thereafter (Am J Gastroenterol 2016;111:30)
Clinical images
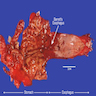
Gross description
- Normal appearing or nodule, erosion or polyp
Microscopic (histologic) description
- Barrett esophagus dysplasia divided into low grade dysplasia and high grade dysplasia; indefinite for dysplasia also a valid interpretation (but not part of a histologic spectrum of progression) (Hum Pathol 1988;19:166)
- Indefinite for dysplasia (Hum Pathol 1988;19:166):
- Histologic features too marked for reactive atypia but not sufficient for a definitive diagnosis of dysplasia
- Cases with significant inflammation or ulceration in which inflammation obscures the findings
- Artifact limits interpretation (thermal effect, denuded surface epithelium, rarely technical issues)
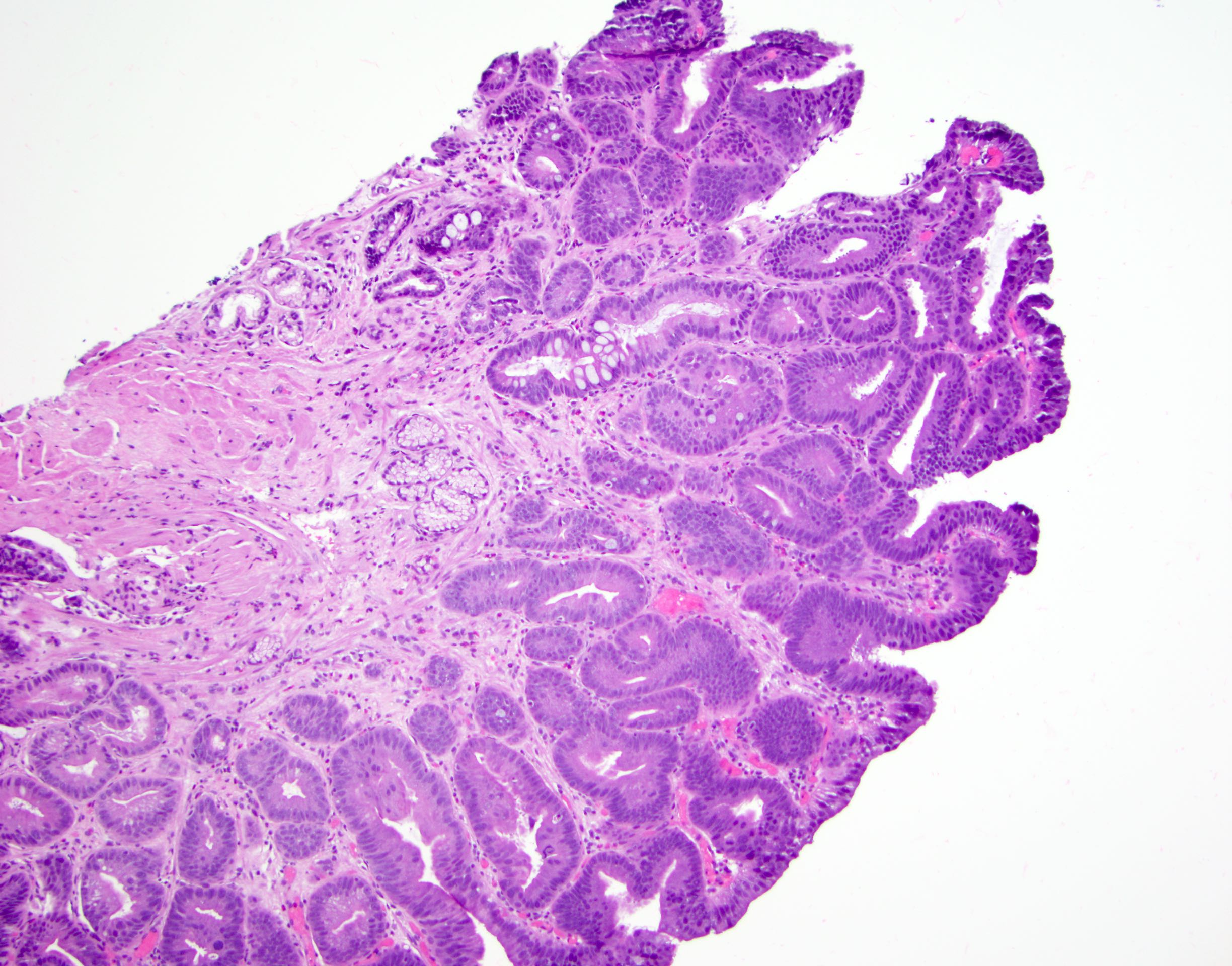
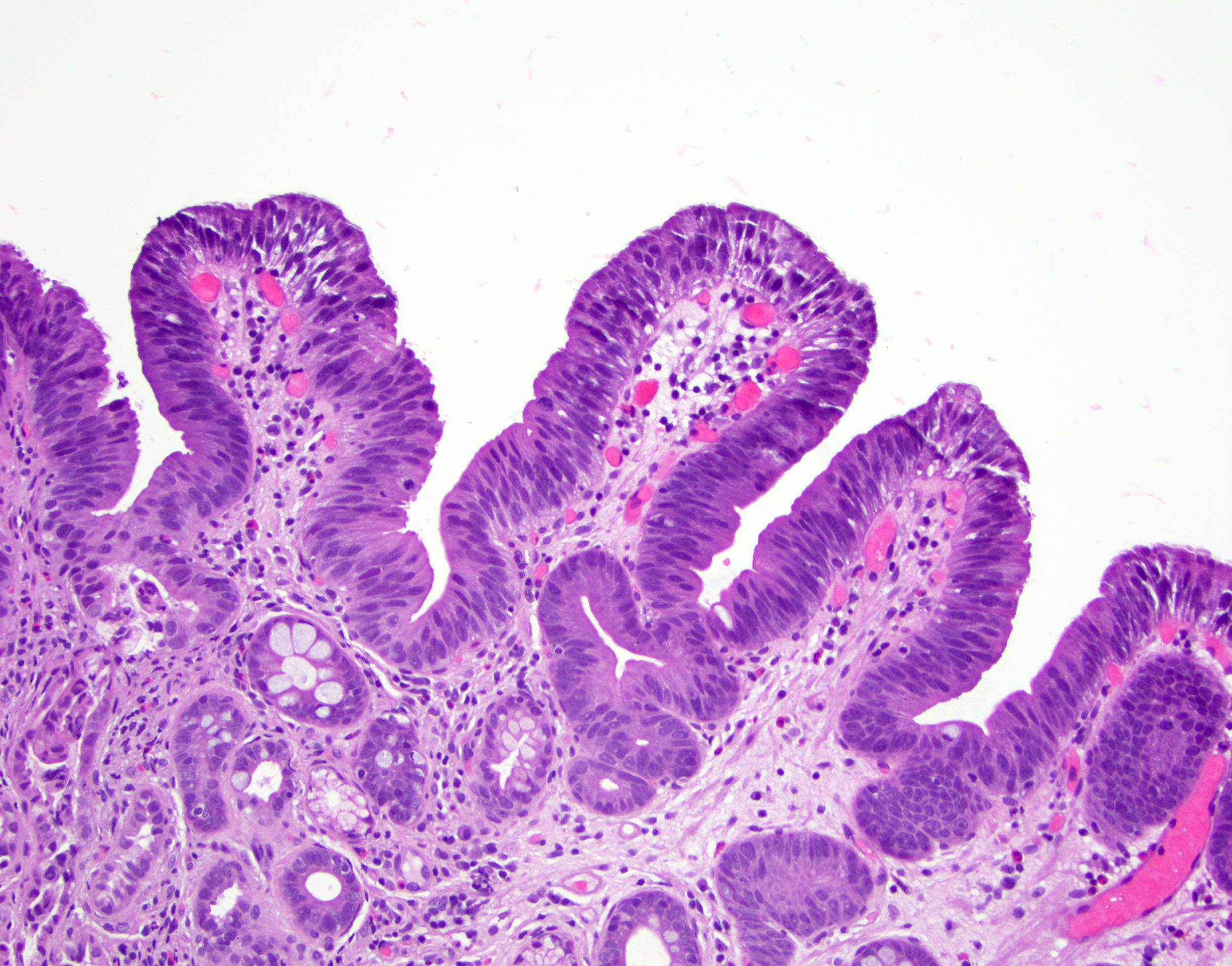
- Low grade dysplasia (Hum Pathol 1988;19:166):
- Unequivocally neoplastic
- Nuclear hyperchromasia, enlargement and stratification identified in the deeper glands and involving the surface epithelium
- Maintenance of nuclear polarity
- Abrupt transition between dysplastic and nondysplastic zones
- Effacement or loss of the 4 surface lines that characterize nondysplastic Barrett mucosa (Surg Pathol Clin 2017;10:781)
- Little (if any) architectural abnormalities
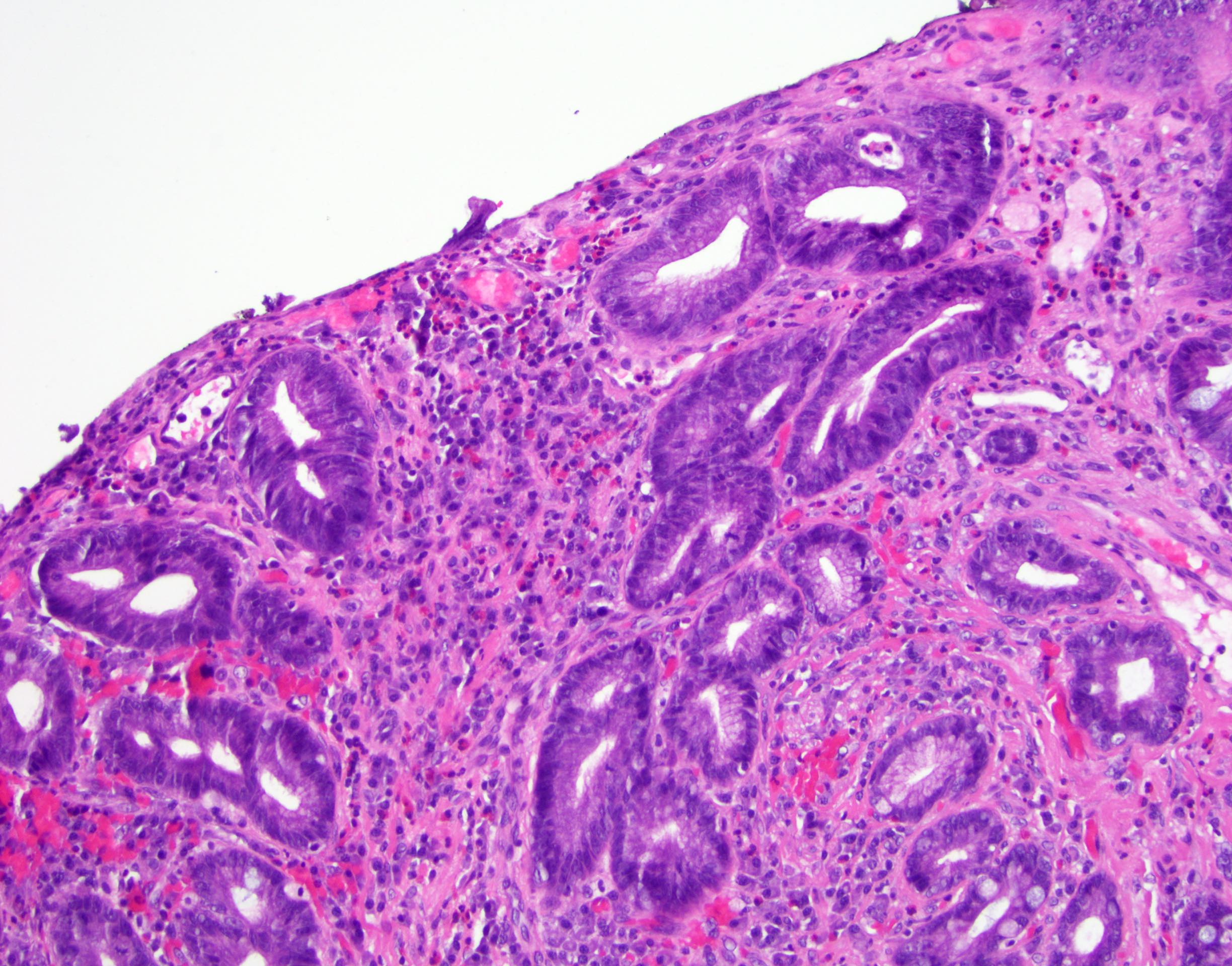
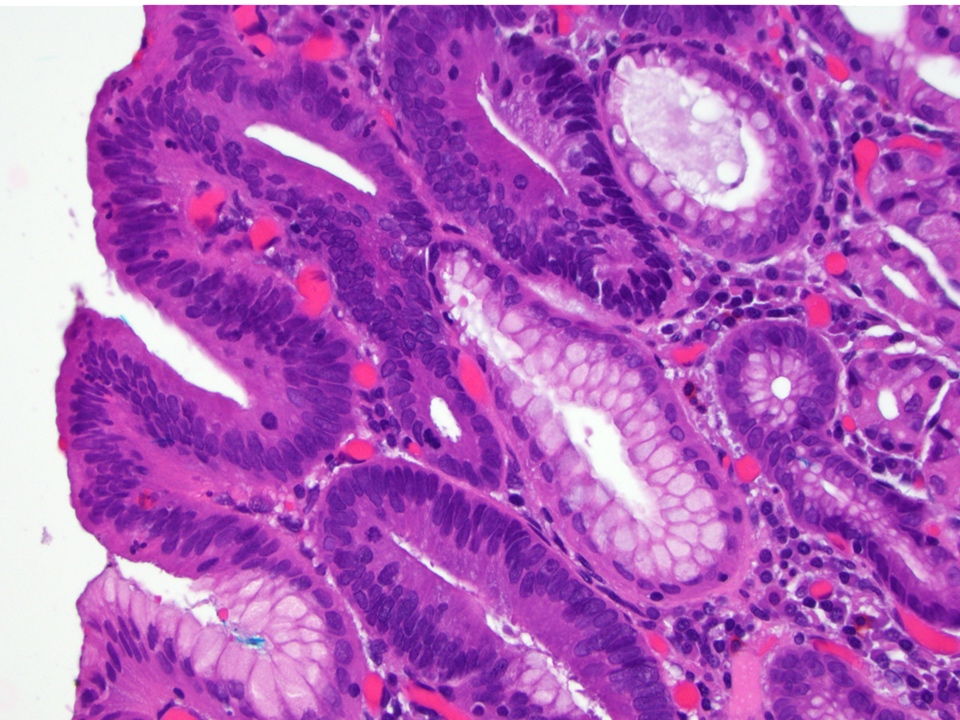
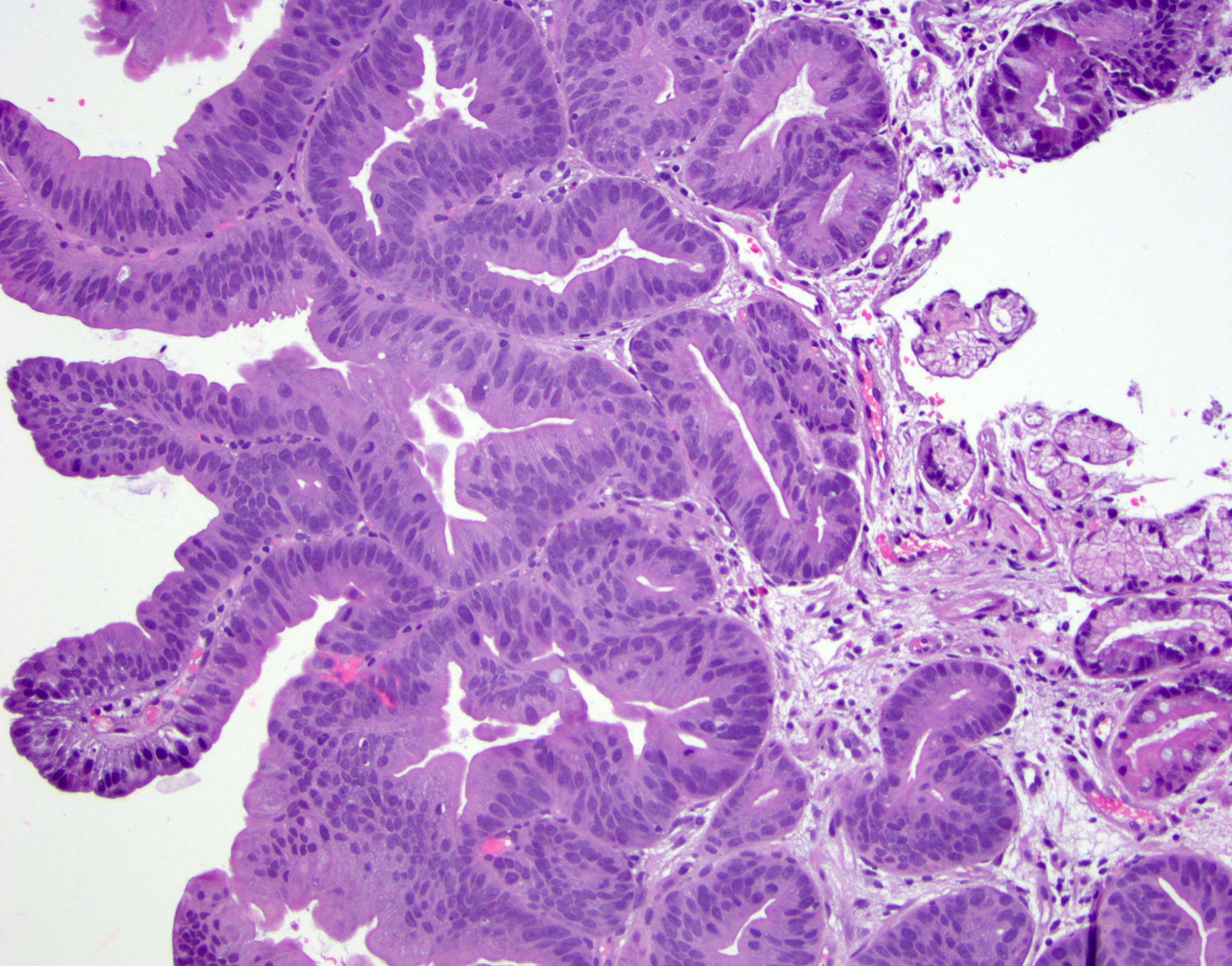
- High grade dysplasia (Hum Pathol 1988;19:166, Am J Gastroenterol 2008;103:2333):
- Greater degree of cytologic atypia in addition to architectural abnormalities
- Architectural abnormalities:
- Irregular size and shape of crypts, crowded crypts with little intervening lamina propria, intraluminal budding or cribriforming, rare dilated glands with intraluminal necrotic debris
- Cytologic features:
- Lack of surface maturation, loss of nuclear polarity, marked nuclear enlargement, pleomorphism and hyperchromasia, irregular nuclear contours
- Loss of nuclear polarity considered an important objective criterion to diagnose high grade dysplasia
- Basal crypt dysplasia (Am J Surg Pathol 2006;30:423):
- Controversial diagnosis
- Cytologic atypia in the basal pits with surface maturation
- Cytologic atypia may be low grade or high grade
- Some authors prefer to use the indefinite for dysplasia category for such lesions, especially with low grade atypia confined to the deep mucosa
- Finding of crypt dysplasia in one biopsy suggests high likelihood of finding conventional dysplasia in other biopsies
- Performing further deeper levels recommended
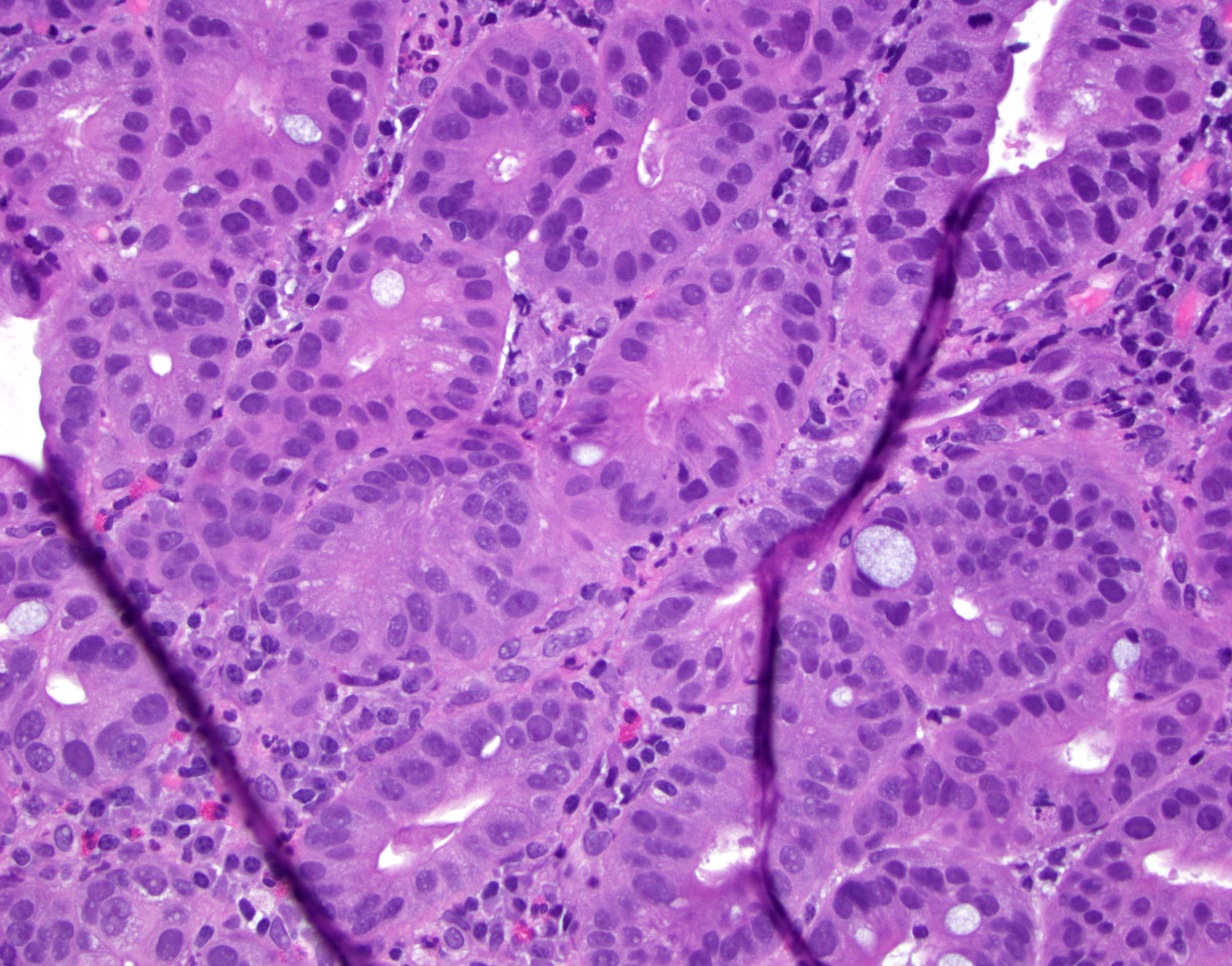
- Gastric foveolar dysplasia (non adenomatous dysplasia):
- Full thickness atypia with non stratified nuclei (Mod Pathol 2010;23:1, Hum Pathol 2013;44:1146)
- Nuclear enlargement, variably prominent nucleoli
- May appear bland and reactive at low magnification but nuclear alterations seen at high magnification
- Low grade dysplasia:
- Nuclear size 2 - 3 times the size of a small, mature lymphocyte, mostly mucinous cytoplasm and variably prominent nucleoli
- High grade dysplasia:
- Nuclear size at least 3 - 4 times the size of a small, mature lymphocyte, most cases have prominent nucleoli (Mod Pathol 2010;23:1, Hum Pathol 2013;44:1146)
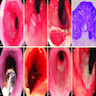
Microscopic (histologic) images
Contributed by Dipti M. Karamchandani, M.D.
Cytology description
- Used as an adjunct to biopsy in diagnosis of Barrett esophagus and associated neoplasms (Diagn Cytopathol 2003;29:130)
- High degree of diagnostic accuracy of cytology for the diagnosis of Barrett associated high grade dysplasia, with reported sensitivity of 82% and specificity of 95% (Diagn Cytopathol 2003;29:130)
- Observed sensitivity for low grade dysplasia is low (about 31%)
- Cytologically, dysplasia shows haphazard arrangement of cells, nuclear enlargement, nuclear hyperchromasia, nuclear membrane irregularity
Positive stains
- Ancillary stains (such as AMACR and p53) have been studied for diagnosing Barrett related dysplasia; neither proved sufficiently sensitive or specific to be clinically useful in detection and classification of Barrett related dysplasia (Am J Surg Pathol 2016;40:e83, Am J Surg Pathol 2017;41:e8, Am J Surg Pathol 2016;40:e83)
- Both strong or absent (null phenotype) p53 staining considered significant (Am J Surg Pathol 2016;40:e83, Am J Surg Pathol 2017;41:e8)
- Gastrointestinal Pathology Society (GIPS) recommendation for p53 in diagnosing Barrett esophagus dysplasia:
- Additional studies are needed to develop and validate precise criteria before p53 staining can be fully endorsed and incorporated into the morphologic dysplasia diagnosis algorithm (Am J Surg Pathol 2017;41:e8)
- p53 appears promising marker in predicting disease progression but not recommended for routine use at present
Videos
Update on recently developed quality metrics
Sample pathology report
- Esophagus, 36 cm, biopsy:
- Barrett esophagus with low grade dysplasia
- Esophagus, 35 cm, biopsy:
- Barrett esophagus with high grade dysplasia
- Esophagus, 34 cm, biopsy:
- Barrett esophagus with epithelial alterations indefinite for dysplasia
Differential diagnosis
- Reactive atypia versus low grade foveolar dysplasia:
- Full thickness nuclear atypia with nonstratified nuclei suggests low grade gastric foveolar type dysplasia
- Reactive atypia is usually limited to upper mucosa
- Indefinite for dysplasia versus reactive atypia:
- Maintenance of 4 lines in a background of inflammation suggests reactive atypia (Surg Pathol Clin 2017;10:781)
- Indefinite for dysplasia versus low grade dysplasia:
- Nuclear changes extending to the surface epithelium in a background of significant inflammation suggests a diagnosis of indefinite for dysplasia
- High grade dysplasia versus intramucosal adenocarcinoma:
- Histologic features of lamina propria invasion (single cells in more than one focus, never ending glandular pattern, solid sheets of cells, significant cribriforming) is diagnostic of intramucosal adenocarcinoma (Am J Gastroenterol 2008;103:2333)
Additional references
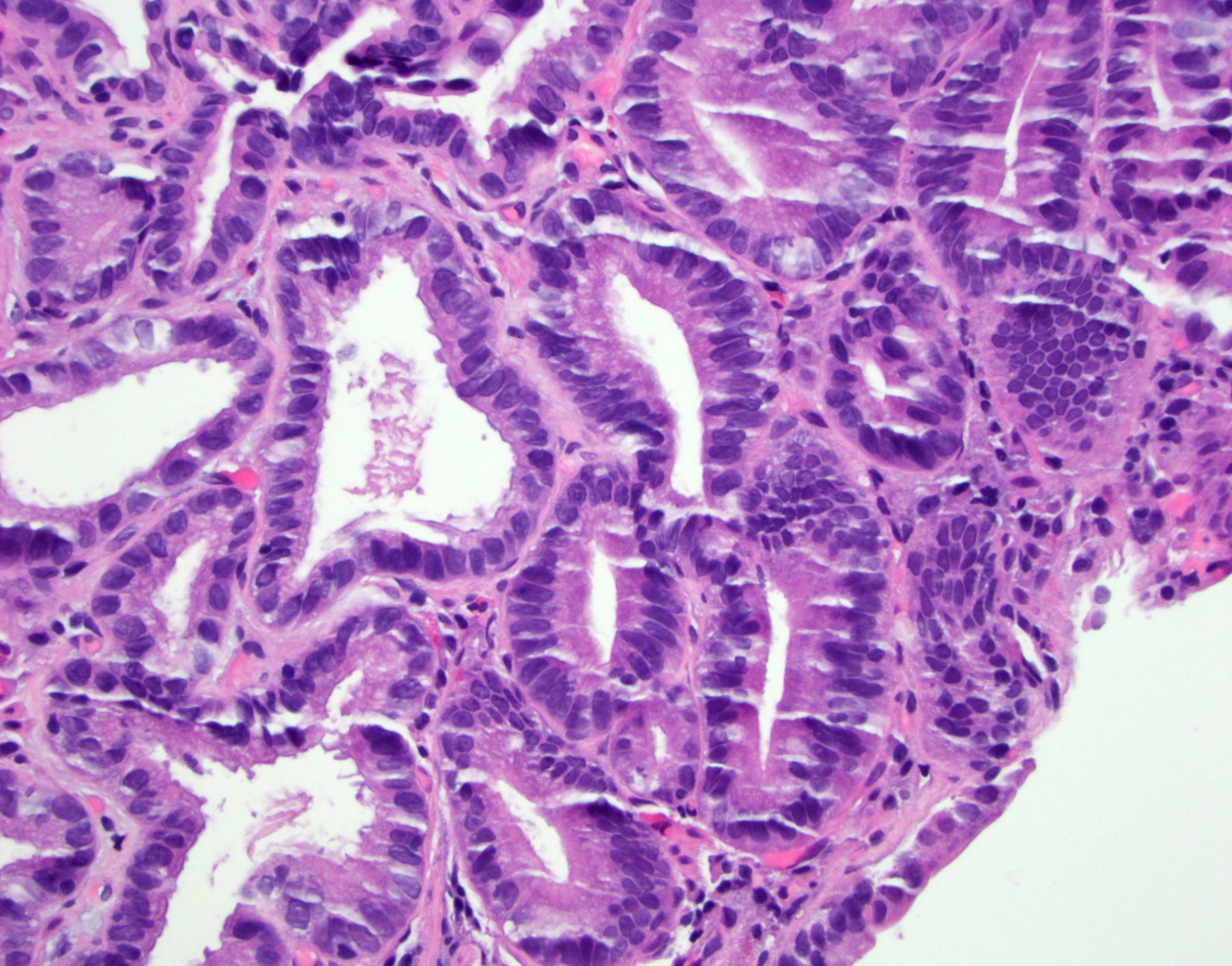
Practice question #1
Above is a photomicrograph taken from a biopsy obtained from salmon colored mucosa in the distal esophagus extending to about 2 cm proximal to the gastroesophageal junction. What is your diagnosis?
- Barrett esophagus, negative for dysplasia
- Barrett esophagus with epithelial alterations indefinite for dysplasia
- Barrett esophagus with high grade dysplasia
- Barrett esophagus with low grade dysplasia
Practice answer #1
Practice question #2
A nodule is found on surveillance endoscopy of a 60 year old patient with a longstanding history of Barrett esophagus. What should be the next step in patient management?
- Endoscopic mucosal resection of the nodule
- Esophagectomy
- Radiofrequency ablation of the nodule
- Use of use of wide area transepithelial sampling with computer assisted 3 dimensional analysis (WATS)
Practice answer #2
Practice question #3
- A biopsy diagnosis of low grade dysplasia was made by a pathologist and confirmed by a subspecialized GI pathologist. What is the preferred appropriate management based on the ACG 2016 guidelines?
- Endoscopic ablation therapy
- Endoscopic mucosal resection
- Endoscopic surveillance at 6 months interval
- Endoscopic surveillance at 12 months interval
Practice answer #3