Table of Contents
Definition / general | Essential features | Terminology | ICD coding | Epidemiology | Sites | Clinical features | Diagnosis | Prognostic factors | Case reports | Treatment | Microscopic (histologic) description | Microscopic (histologic) images | Peripheral smear description | Peripheral smear images | Positive stains | Flow cytometry description | Molecular / cytogenetics description | Molecular / cytogenetics images | Sample pathology report | Differential diagnosis | Additional references | Practice question #1 | Practice answer #1 | Practice question #2 | Practice answer #2Cite this page: Gupta S, Courville E. AML with myelodysplasia defining abnormalities. PathologyOutlines.com website. https://www.pathologyoutlines.com/topic/leukemiaamlmulti.html. Accessed September 21st, 2025.
Definition / general
- ≥ 20% myeloid blasts in the blood or bone marrow with 1 or more of the following: features of myelodysplasia, prior history of myelodysplastic syndrome (MDS) or MDS / myeloproliferative neoplasm (MPN) or MDS related cytogenetic abnormalities
- Requires absence of prior cytotoxic / radiation therapy and absence of a recurrent cytogenetic abnormality
- In de novo acute myeloid leukemia (AML) with mutated NPM1 or biallelic mutation of CEBPA, multilineage dysplasia alone or del(9q) alone are insufficient
- Includes refractory anemia with excess blasts in transformation defined by French-American-British (FAB) classification
Essential features
- AML with morphological features of myelodysplasia in > 50% of the cells of 2 or more lineages or a prior history of MDS or MDS / MPN or MDS related cytogenetic abnormalities
- Generally poorer prognosis with lower remission rates and shorter overall survival than other AML subtypes (Am J Clin Pathol 2020;154:731)
- Commonly presents with severe pancytopenia
Terminology
- Acute myeloid leukemia with multilineage dysplasia
- Acute myeloid leukemia with prior myelodysplastic syndrome
- Acute myeloid leukemia with myelodysplasia related changes (AML-MRC)
ICD coding
- ICD-O: 9895/3 - acute myeloid leukemia with myelodysplasia related changes
Epidemiology
- Mainly elderly patients; median age 70 years (Am J Hematol 2020;95:612)
- Rare in children
- Up to 48% of AML (Surg Pathol Clin 2010;3:1153)
Sites
- Blood and bone marrow
Clinical features
- Frequently presents with severe pancytopenia
- Cases with 20 - 29% blasts may have a slowly progressive course with less marked cytopenias (Am J Hematol 2014;89:E193)
Diagnosis
- Made based on the following (Surg Pathol Clin 2010;3:1153):
- Microscopic features, including bone marrow blast percentage and the presence and degree of dysplasia
- Immunophenotyping to confirm myeloid lineage
- Results of cytogenetic studies
- Clinical correlation to evaluate for prior history of MDS or MDS / MPN and exclude prior cytotoxic or radiation therapy for an unrelated disease
Prognostic factors
- Generally poorer prognosis with lower remission rates and shorter overall survival than other AML subtypes (Am J Clin Pathol 2020;154:731)
- Diagnosis of AML-MRC solely based on multilineage dysplasia or previously untreated MDS or MDS / MPN have significantly better outcomes than those with MDS defining cytogenetic abnormalities or disease secondary to a previously treated MDS or MDS / MPN (Am J Hematol 2020;95:612)
- In de novo AML cases lacking myelodysplasia related cytogenetic abnormalities, select dysplastic features may carry more prognostic significance than others with micromegakaryocytes and hypogranulated myeloid cells identified as significantly associated with shorter event free survival (Mod Pathol 2015;28:965)
Case reports
- 47 year old man with a history of Crohn's disease and new onset acute myeloid leukemia with myelodysplasia related changes (Case Rep Oncol 2018;11:573)
- 54 year old man with a prior history of acute myeloid leukemia with myelodysplasia related changes presenting with new onset pleural effusion with leukemic blasts (Diagn Cytopathol 2011;39:451)
- 77 year old man with acute myeloid leukemia with myelodysplasia related changes (Intern Med 2011;50:3037)
- 77 year old man with acute myeloid leukemia with myelodysplasia related changes demonstrating mixed lineage phenotype (Blood 2016;128:1663)
- 84 year old woman with acute myeloid leukemia with myelodysplasia related changes showing basophilic differentiation (Am J Hematol 2014;89:1082)
Treatment
- Intensive induction chemotherapy followed by allogeneic stem cell transplant (Expert Opin Emerg Drugs 2021;26:245)
- Chemotherapy with hypomethylating agents and with or without venetoclax (Blood 2019;133:7)
- Liposomal formulation of daunorubicin and cytarabine (J Clin Oncol 2018;36:2684)
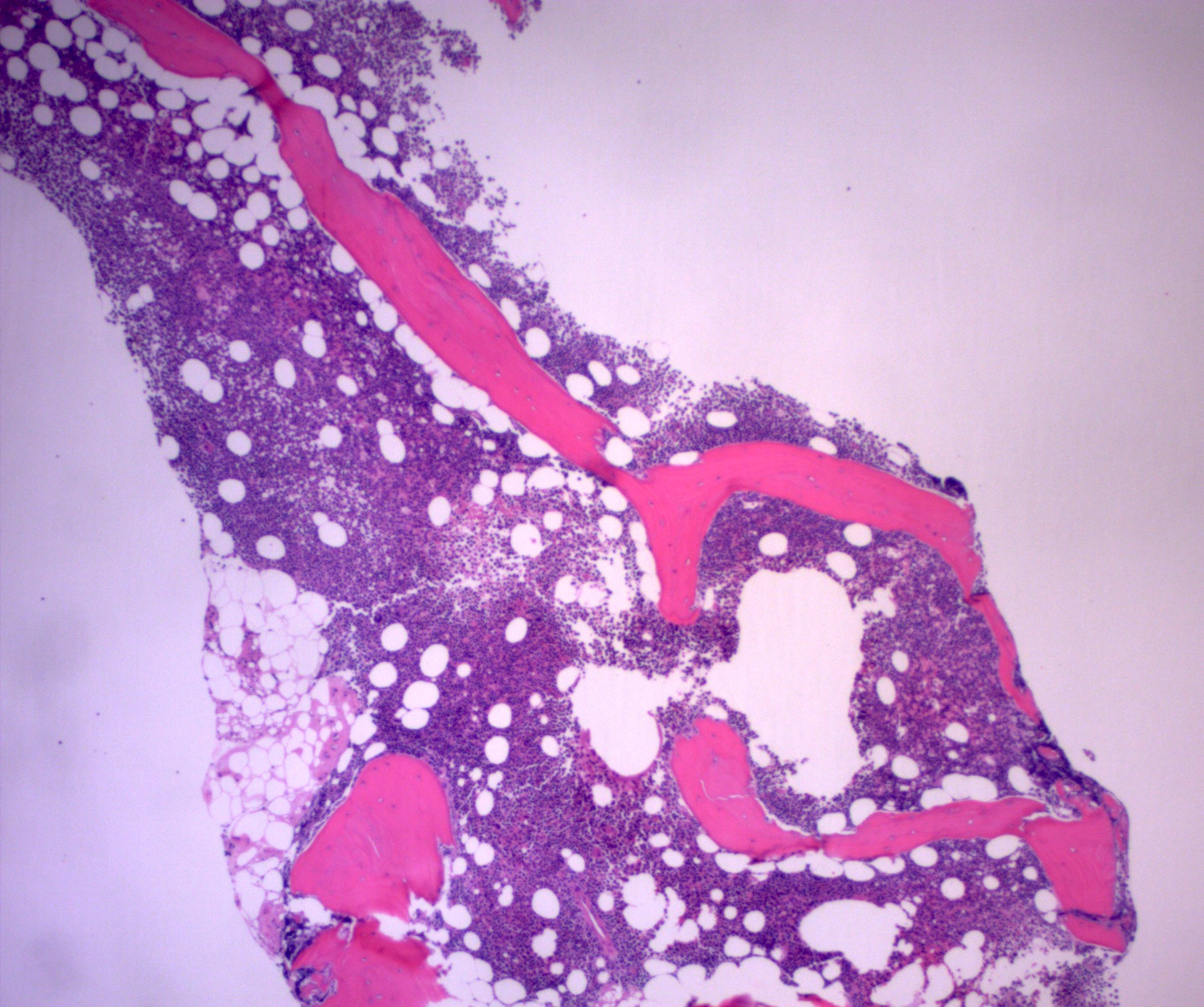
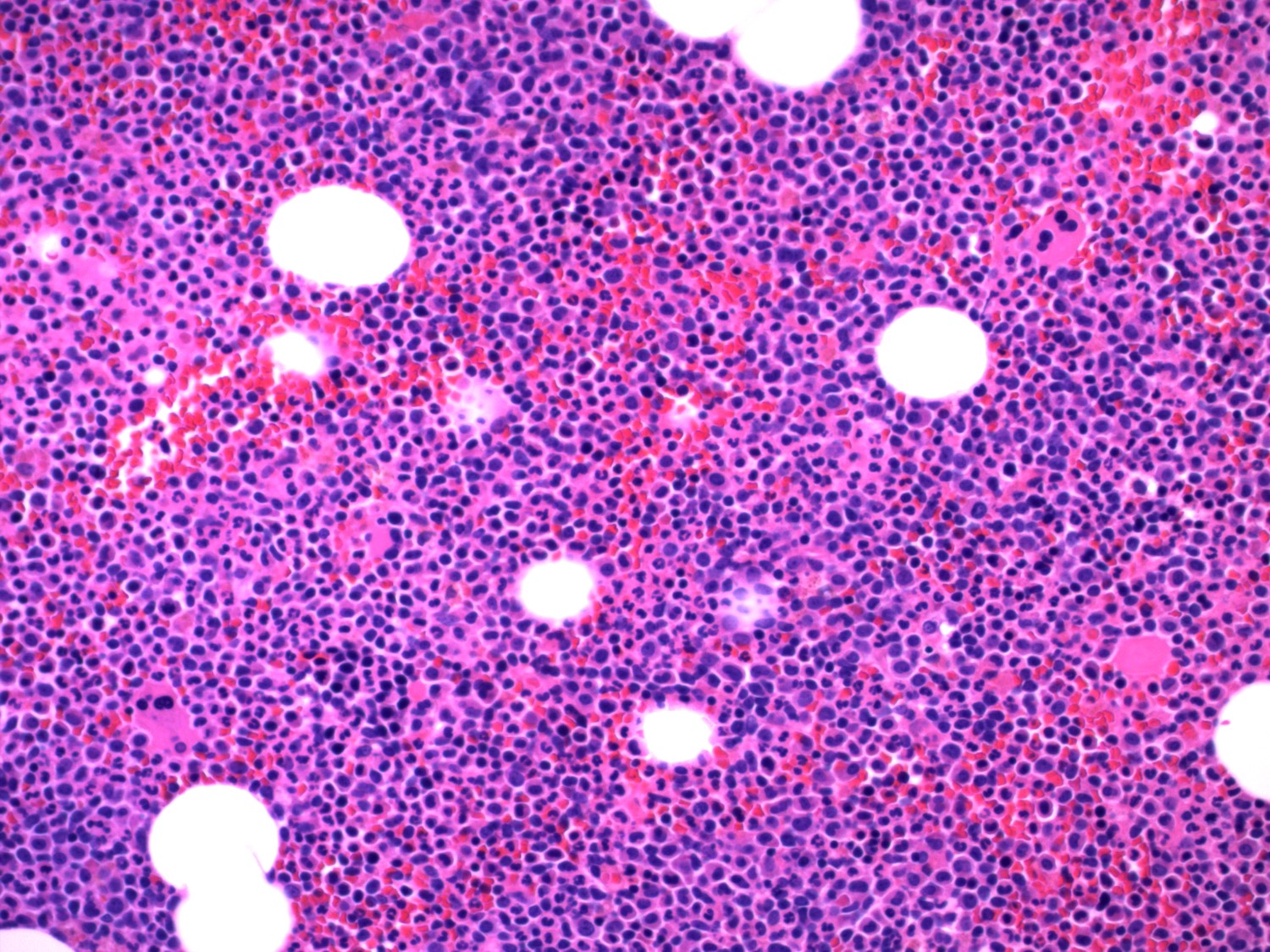
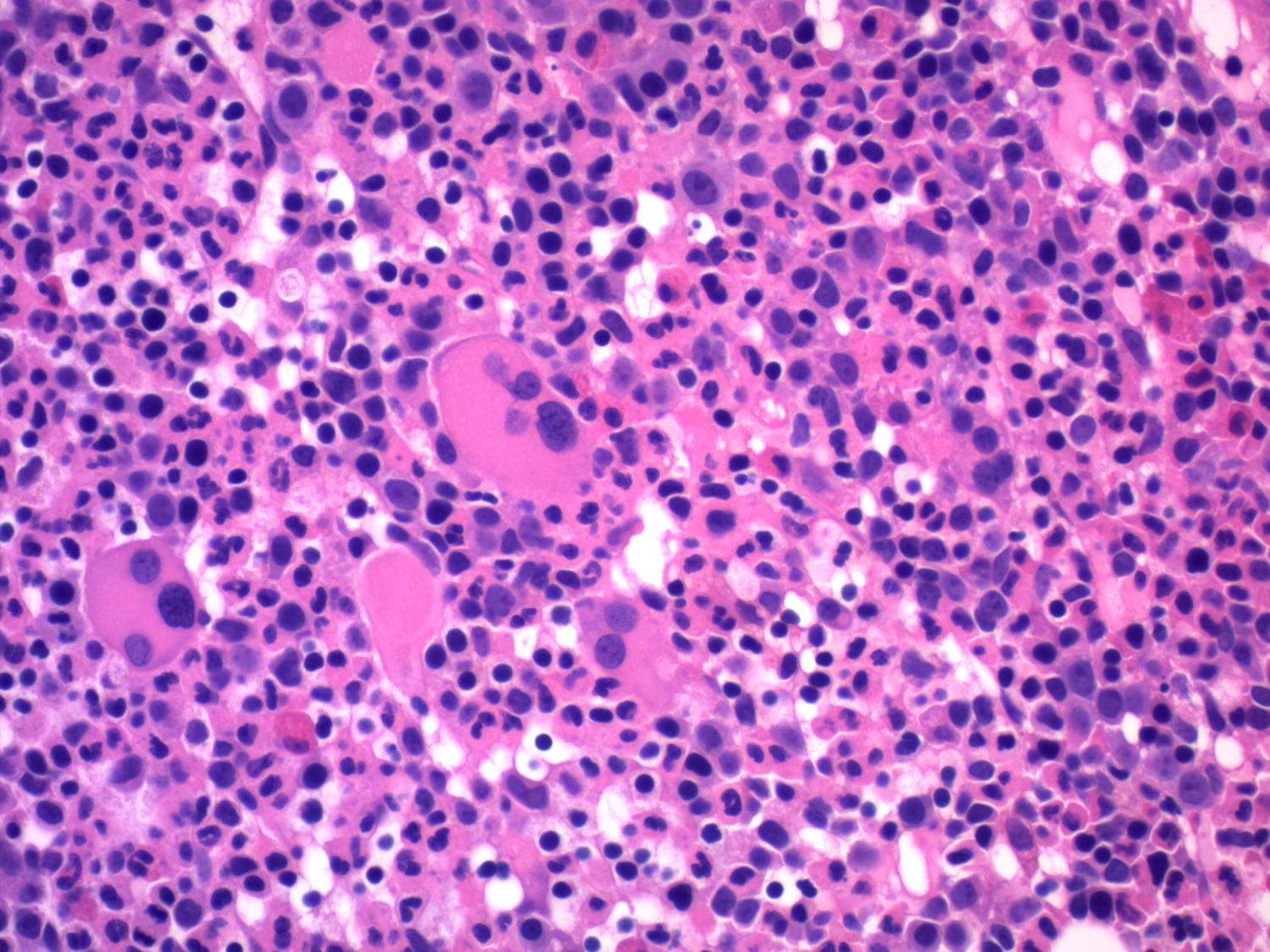
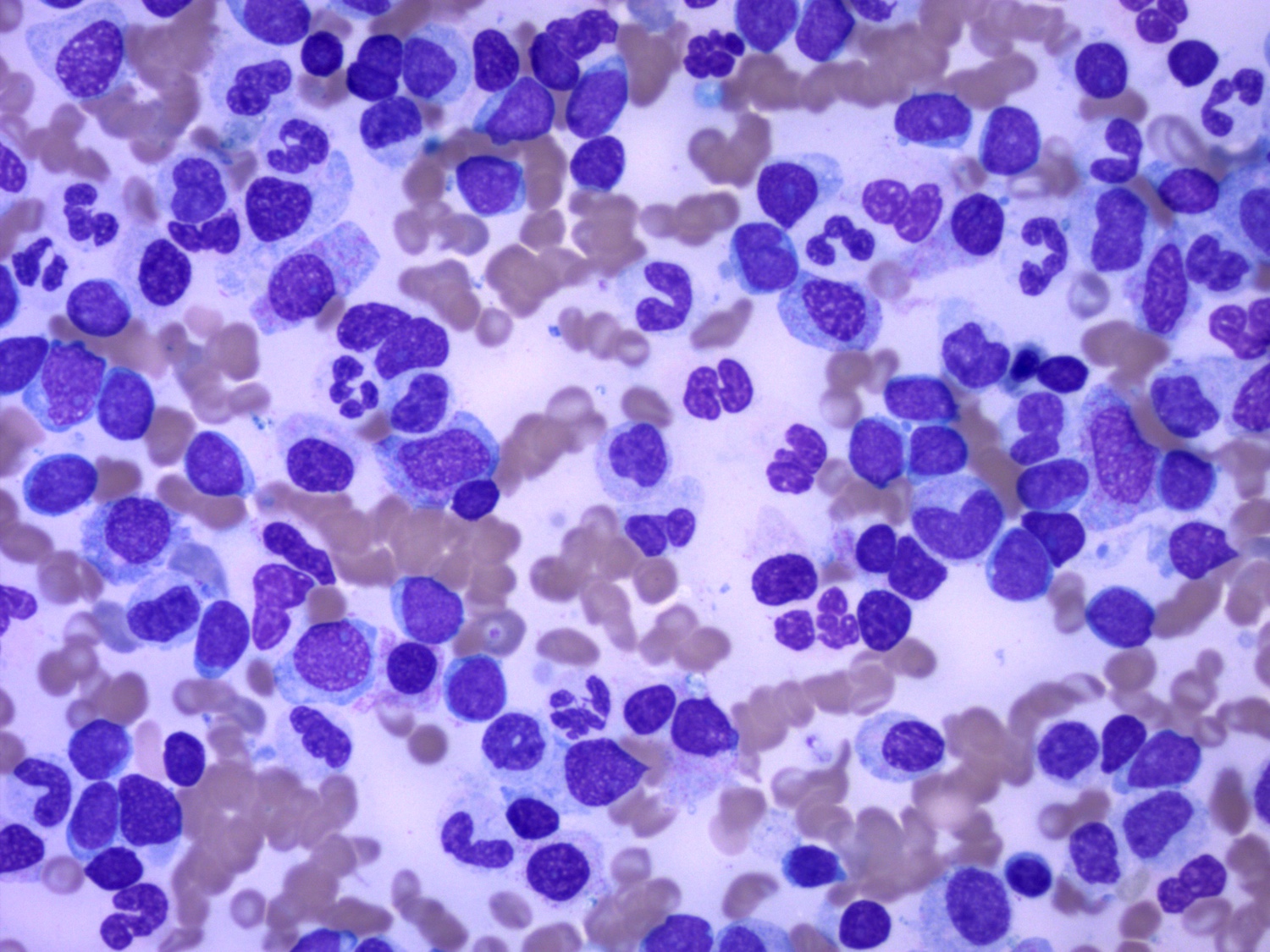
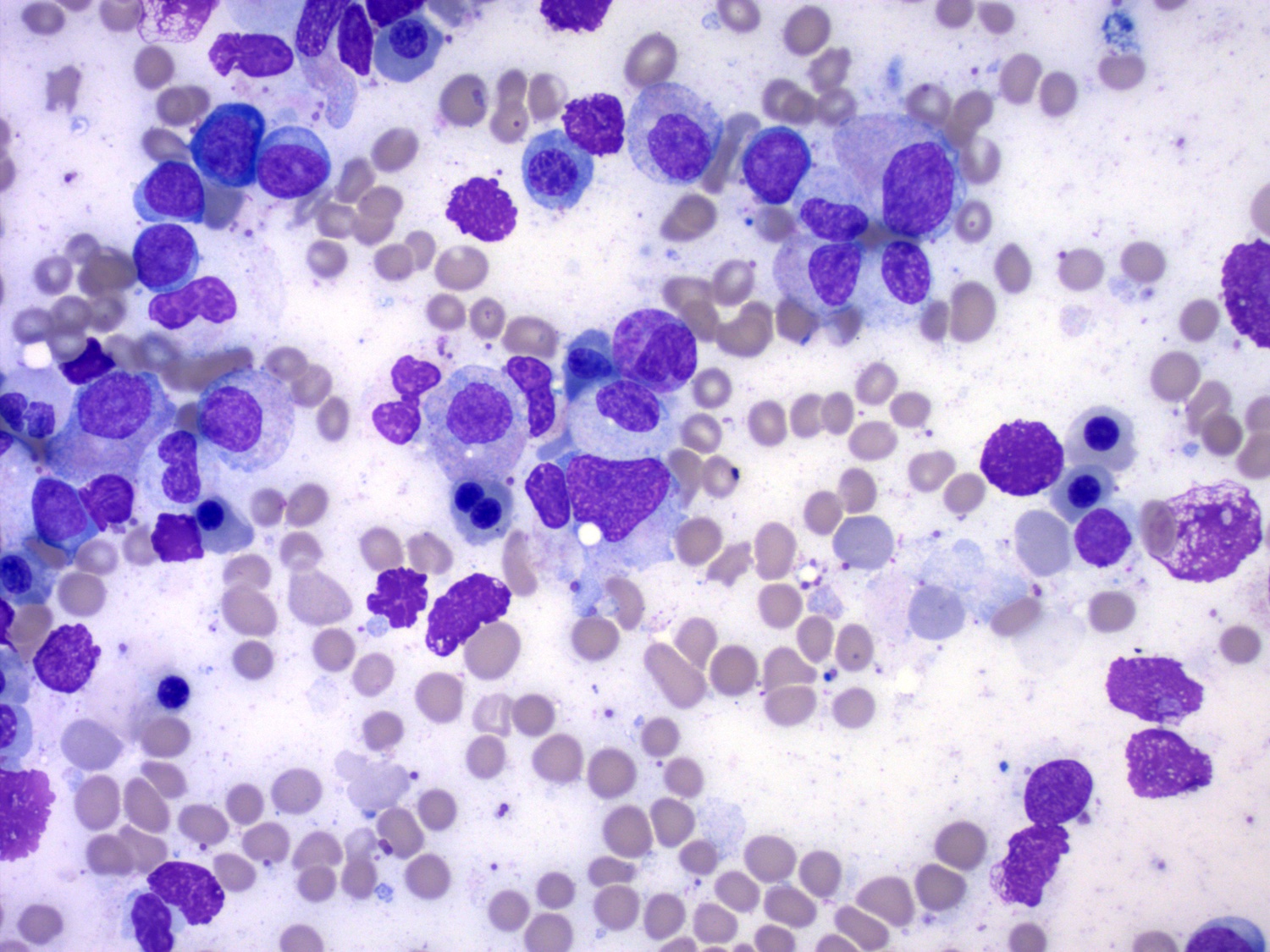
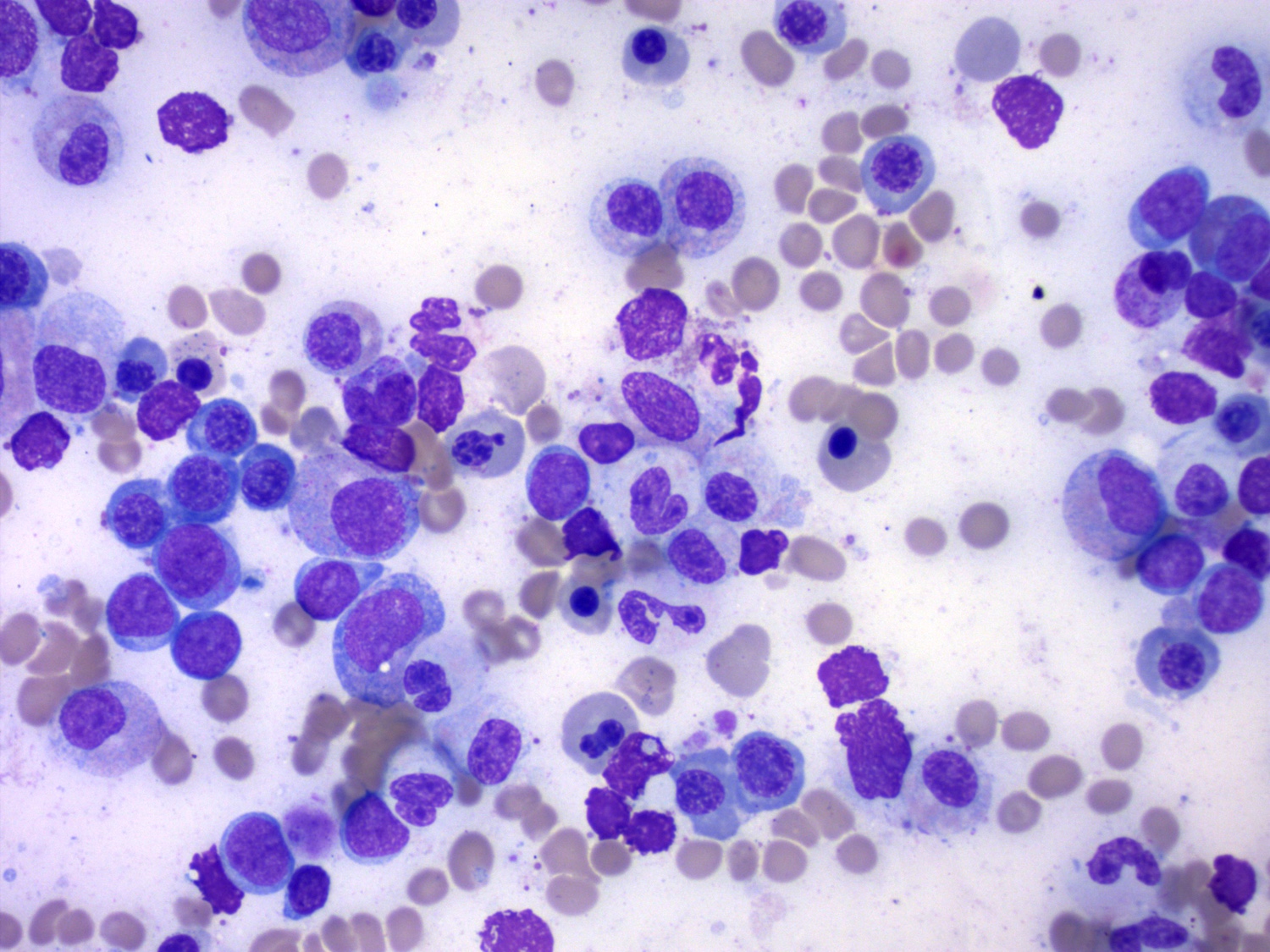
Microscopic (histologic) description
- Blasts 20% or more
- Dysplasia in > 50% of the cells in 2 or more lineages
- Dyserythropoiesis: nuclear budding, megaloblastosis, multinucleation, nuclear budding, irregular nuclear contours, cytoplasmic vacuoles, karyorrhexis, ring sideroblasts
- Dysgranulopoiesis: hypogranular cytoplasm, hyposegmented nucleus, abnormal nuclear segmentation
- Dysmegakaryopoiesis: micromegakaryocytes, nuclear hyposegmentation, nuclear hypersegmentation, separated nuclear lobes
- References: Surg Pathol Clin 2010;3:1153, Am J Clin Pathol 2020;154:731
Microscopic (histologic) images
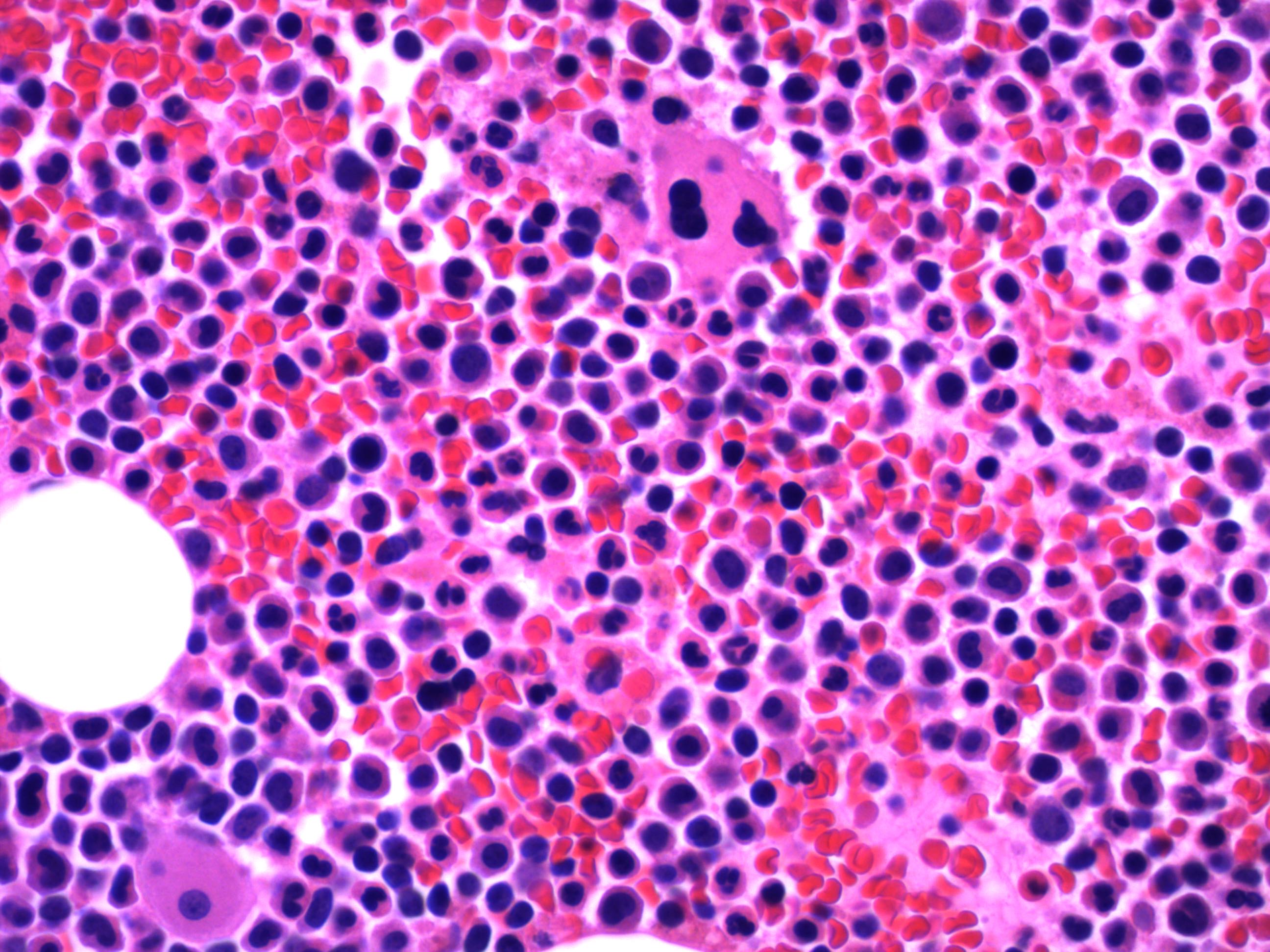
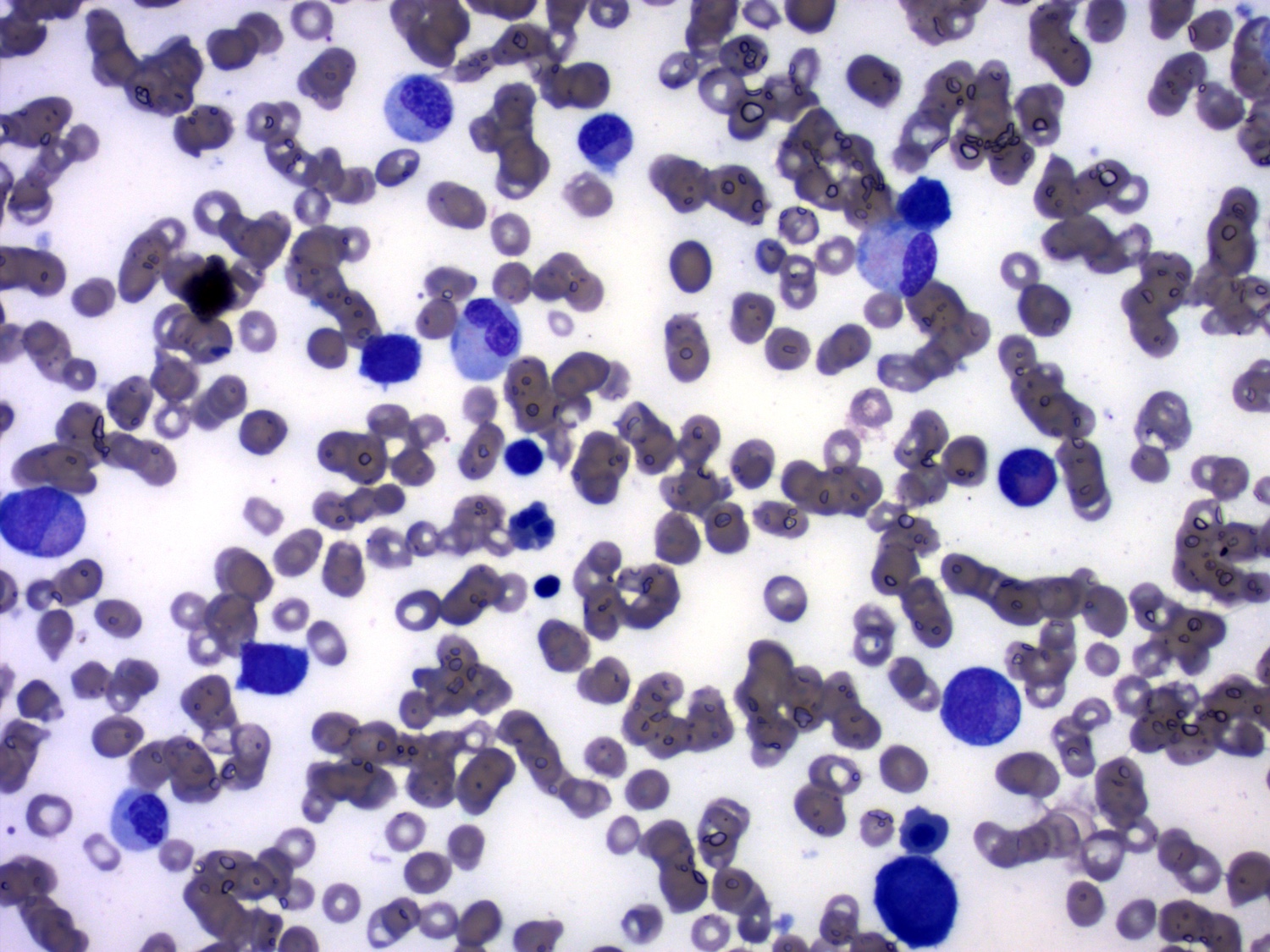
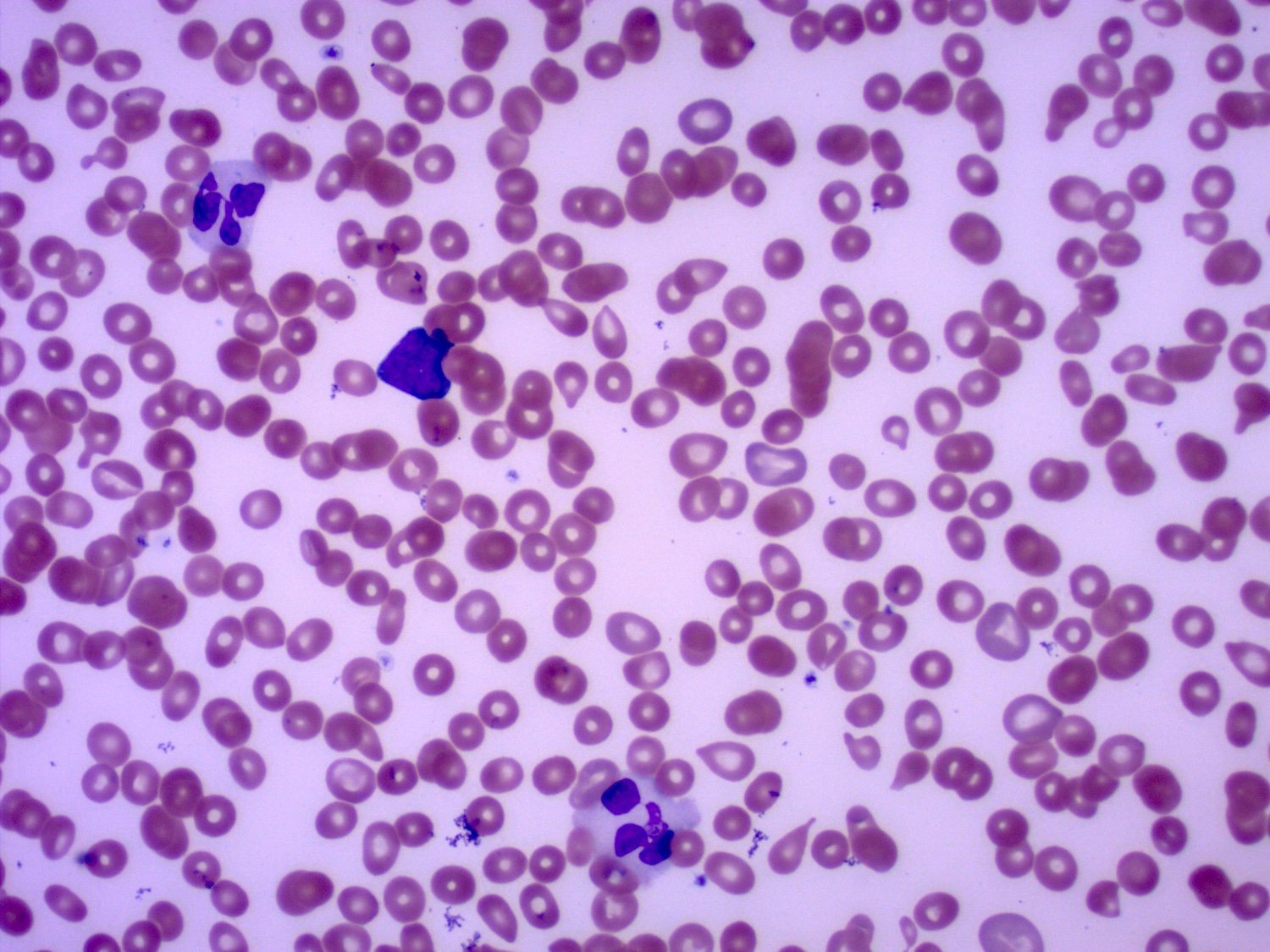
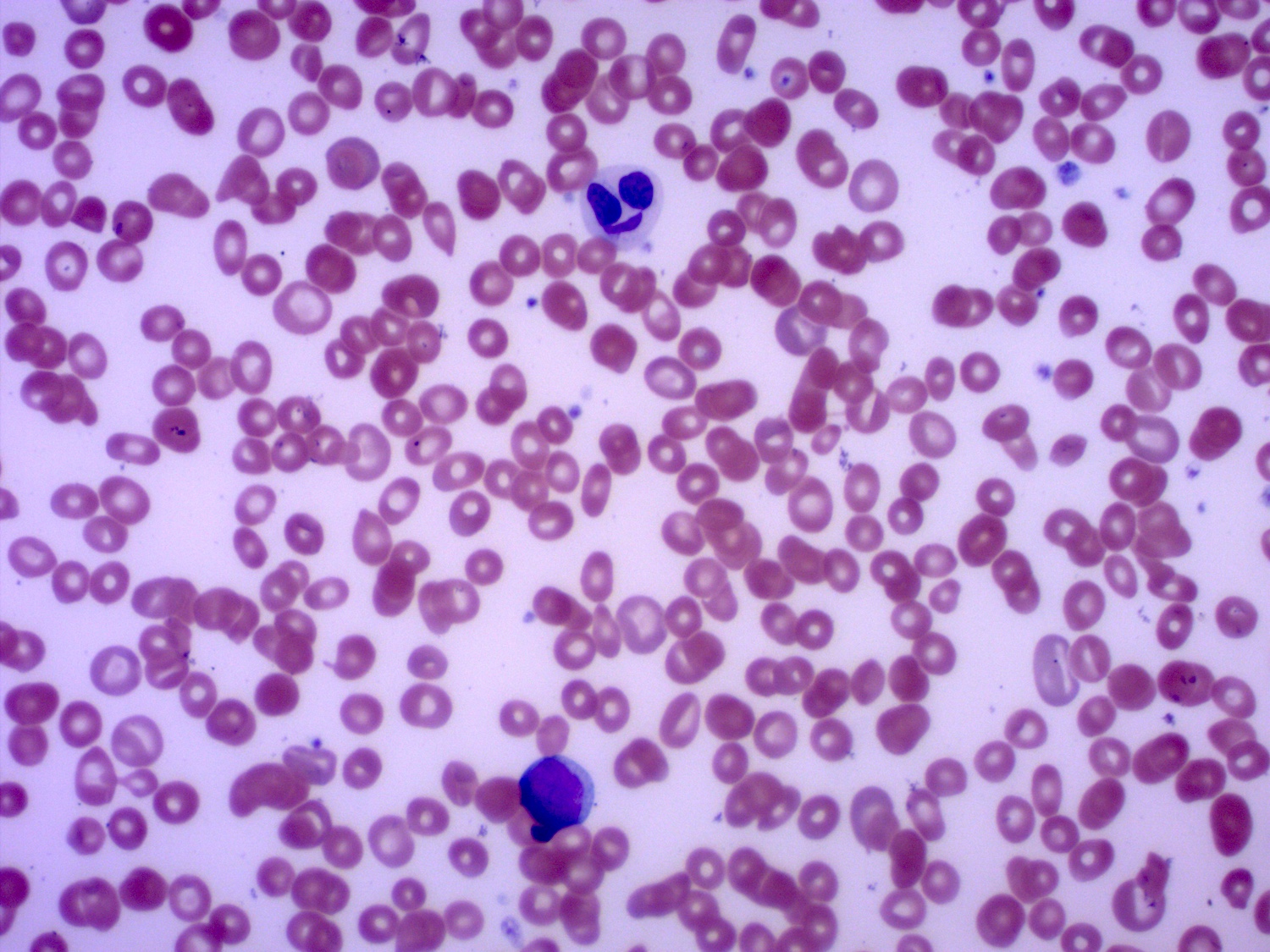
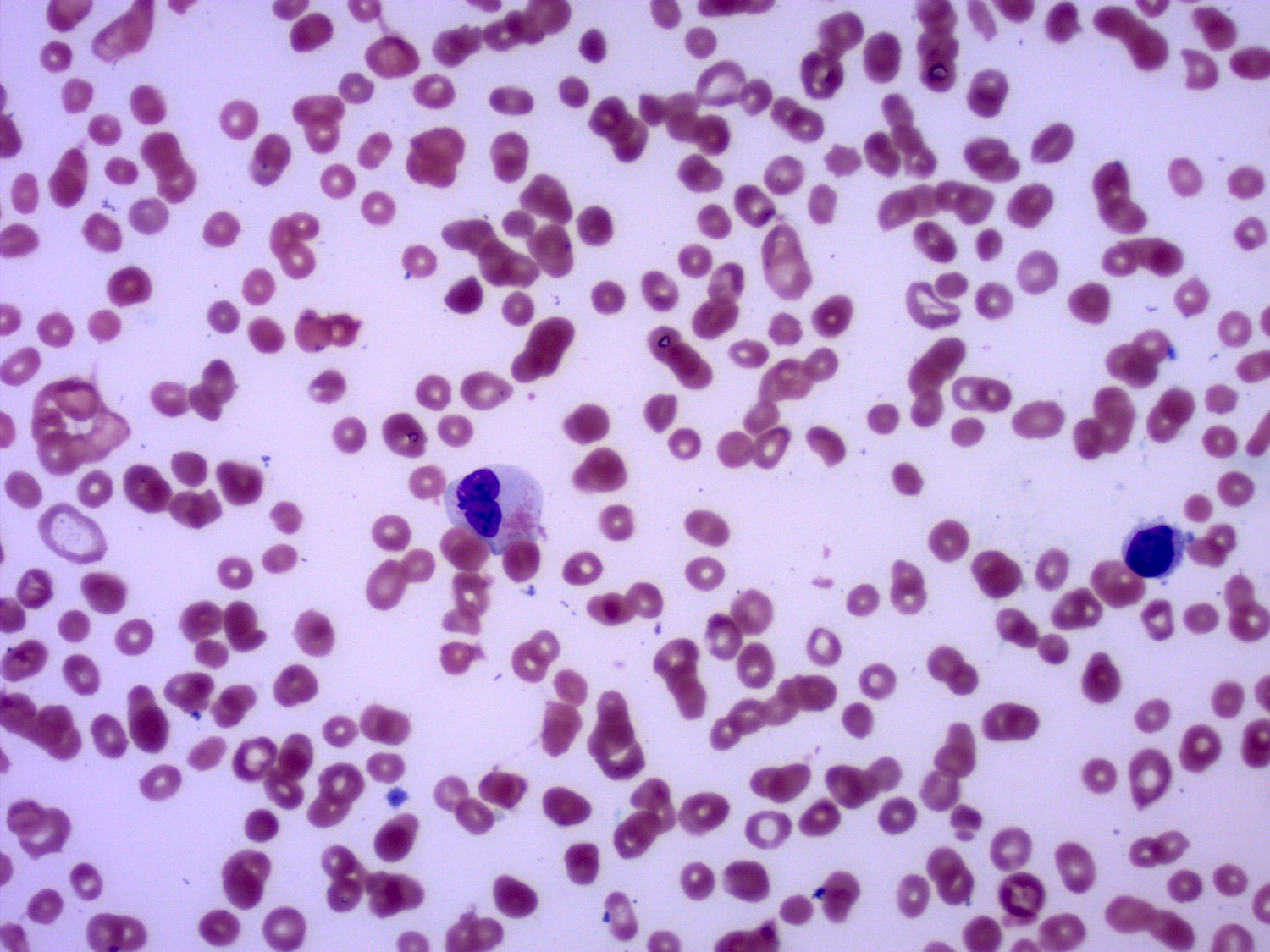
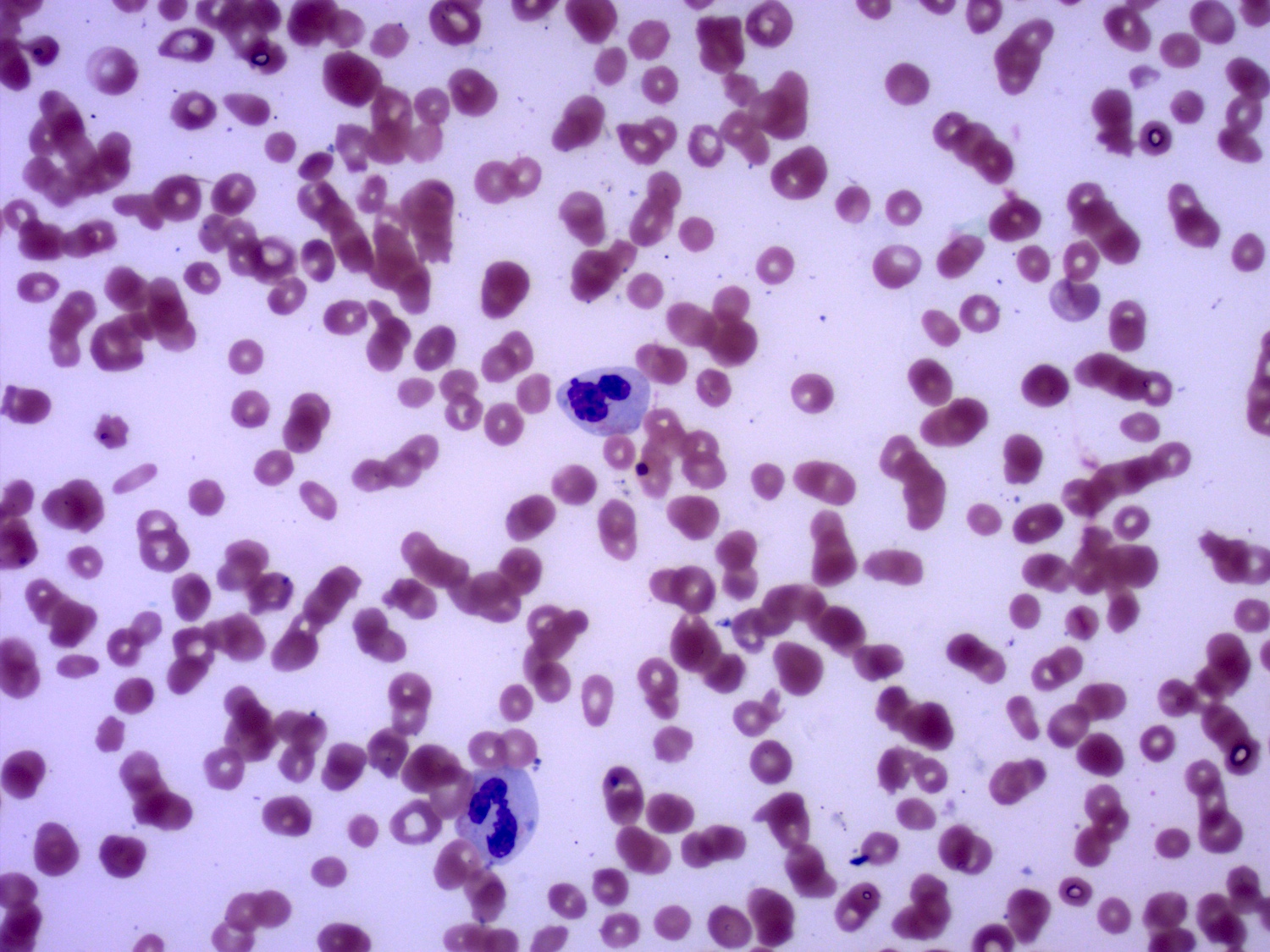
Peripheral smear description
- Nucleated red cells with dysplastic features in the form of nuclear budding and nuclear irregularities
- Increased red blood cell anisopoikilocytosis
- Large, hypogranular platelets
- Circulating blasts may be present
- Hypogranular or hypolobated myeloid elements
- Cytopenias
- Reference: Jaffe: Hematopathology, 2nd Edition, 2016
Peripheral smear images
Positive stains
- CD61 may highlight micromegakaryocytes
- CD34 and CD117 may help with blast percentage estimation on the core biopsy
- Reference: Jaffe: Hematopathology, 2nd Edition, 2016
Flow cytometry description
- Variable immunophenotypic features
- Immunophenotypic features are those reported in myelodysplastic syndromes, including reduced side scatter reflecting hypogranularity of neutrophils, aberrant differentiation patterns, loss of hematogones and aberrant expression of CD56 or CD7 (Acta Haematol 2019;141:232)
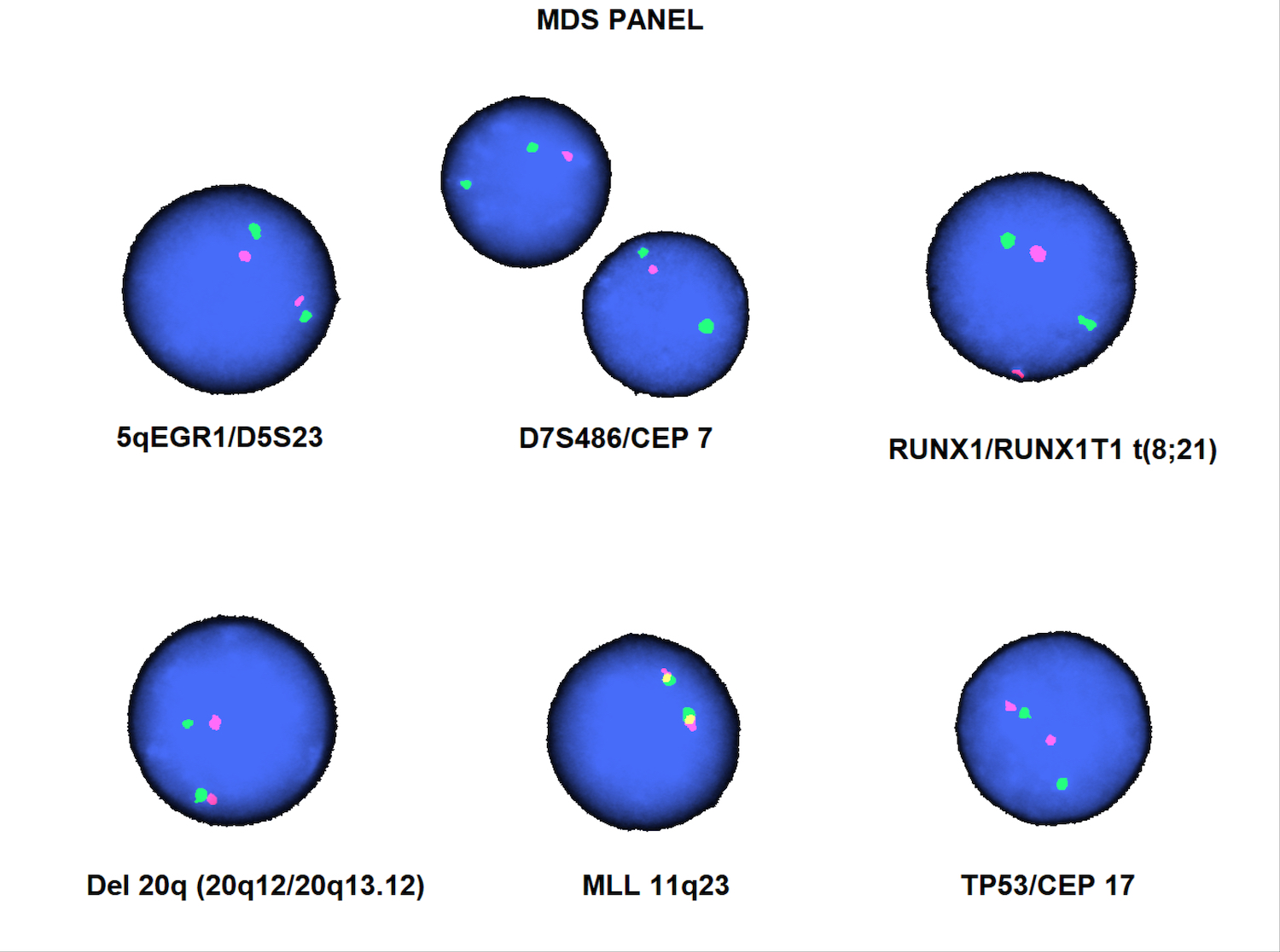
Molecular / cytogenetics description
- Complex karyotypes, loss of chromosome 7 / del(7q), del(5q) and unbalanced translocations involving 5q are the most common cytogenetic abnormalities
- Balanced translocations are less common but frequently involve 5q32-33
- Presence of a specific genetic abnormality characteristic of AML with recurrent genetic abnormalities as defined in the WHO excludes a diagnosis of AML-MRC
- NPM1 mutation and biallelic mutation of CEBPA are fairly uncommon and are rarely associated with MDS related cytogenetic abnormalities
- MDS related mutations (including in U2AF1, ASXL1, TP53) are more frequent in this entity than in AML, NOS (Mod Pathol 2015;28:706)
Table 1: MDS related cytogenetic abnormalities (Am J Clin Pathol 2020;154:731)
| Balanced translocation |
|
| Unbalanced translocation |
|
| Complex cytogenetic abnormality | 3 or more unrelated cytogenetic abnormalities, excluding the recurrent cytogenetic abnormalities seen in AML |
Sample pathology report
- Posterior iliac crest, left / right, bone marrow aspirate, clot section and biopsy:
- Acute myeloid leukemia with myelodysplasia related changes (see comment)
- Comment: The patient's specimen demonstrates a hypercellular marrow with myeloid predominant trilineage hematopoiesis and increased blasts (> 20% by aspirate count and immunohistochemical stains). The peripheral blood shows leukopenia, thrombocytopenia and normocytic normochromic anemia. Flow cytometry analysis identified a large population of abnormal myeloid blasts. Overall, the findings in the current specimen are consistent with acute myeloid leukemia. Given the patient's history of myelodysplastic syndrome, acute myeloid leukemia with myelodysplastic related changes is a compatible diagnosis if a therapy related AML is clinically excluded and a defined recurrent cytogenetic abnormality is not identified (see WHO). Correlation with cytogenetic studies is recommended.
Differential diagnosis
- Therapy related acute myeloid leukemia:
- Characterized by history of cytotoxic chemotherapy or relevant radiation therapy for an unrelated disease
- AML with recurrent cytogenetic abnormalities:
- AML with t(8;21), inv(3) or t(6;9) may show evidence of multilineage dysplasia
- These entities are defined by the presence of defined recurrent cytogenetic abnormalities
- Acute panmyelosis with myelofibrosis:
- Marrow is hypercellular and shows panmyelosis within a diffusely fibrotic stroma
- No prior history of MDS or MDS / MPN
- No AML-MRC defining cytogenetic abnormalities
- Distinction from AML-MRC with myelofibrosis may be difficult
- Acute megakaryoblastic leukemia:
- No prior history of MDS or MDS / MPN
- No AML-MRC defining cytogenetic abnormalities
- No accompanying granulocytic or erythroid dysplasia
- Pure erythroid leukemia:
- Characterized by erythroid precursors representing > 80% of the bone marrow cells, with ≥ 30% proerythroblasts
- Some of the cases previously classified as erythroleukemia (erythroid / myeloid type) may be categorized as AML-MRC according to the current classification system (Blood 2016;127:2391)
- Pure erythroid leukemia should not be diagnosed as AML with myelodysplasia related changes, even if there is a history of a prior myeloid neoplasm, as AML-MRC by definition requires myeloblasts whereas the neoplastic cells in pure erythroid leukemia are erythroid blasts
Additional references
Practice question #1
A patient is diagnosed with de novo myeloid neoplasm. There is multilineage dysplasia (> 50% in 2 of the lineages) and > 20% myeloid blasts, as you can see in the above image from the bone marrow aspirate. The karyotype is normal and an NPM1 mutation is identified. What is the most appropriate diagnosis?
- Acute erythroid leukemia
- Acute myeloid leukemia with myelodysplasia related changes
- Acute myeloid leukemia, NOS
- Therapy related myeloid neoplasm (acute myeloid leukemia)
Practice answer #1
Practice question #2
A 69 year old woman is diagnosed with acute myeloid leukemia. Which of the following supports a diagnosis of acute myeloid leukemia with myelodysplasia related changes?
- Biallelic mutation of CEBPA
- History of below the knee amputation for diabetic complications
- History of cytotoxic chemotherapy for breast carcinoma
- History of myelodysplastic syndrome (MDS)
Practice answer #2
D. History of myelodysplastic syndrome (MDS)
Comment Here
Reference: AML with myelodysplasia related changes
Comment Here
Reference: AML with myelodysplasia related changes