Table of Contents
Definition / general | Essential features | Terminology | ICD coding | Epidemiology | Sites | Pathophysiology | Etiology | Clinical features | Diagnosis | Laboratory | Radiology description | Prognostic factors | Case reports | Treatment | Gross description | Gross images | Microscopic (histologic) description | Microscopic (histologic) images | Virtual slides | Cytology description | Positive stains | Negative stains | Molecular / cytogenetics description | Sample pathology report | Differential diagnosis | Board review style question #1 | Board review style answer #1 | Board review style question #2 | Board review style answer #2Cite this page: Short E, Varma M. Adenocarcinoma. PathologyOutlines.com website. https://www.pathologyoutlines.com/topic/prostateadenoNOS.html. Accessed April 29th, 2024.
Definition / general
- Most common malignancy of the prostate gland
- Originates from prostatic secretory epithelium
Essential features
- Clinical and radiological features neither sensitive nor specific
- Often diagnosed by nontargeted needle biopsies investigating raised serum prostate specific antigen (PSA)
- Absence of basal cell layer is a pathognomonic histological feature
- Pathognomonic diagnostic features: circumferential perineural invasion, glomerulations and collagenous micronodules (mucinous fibroplasia)
- Other histological features: infiltrative architecture, nucleolar prominence, amphophilic cytoplasm and some intraluminal contents (crystalloids, blue mucin, pink amorphous material)
Terminology
- Prostate cancer
- Prostate adenocarcinoma
- Subtypes of prostatic adenocarcinoma: acinar adenocarcinoma, ductal adenocarcinoma, atrophic adenocarcinoma, pseudohyperplastic adenocarcinoma, microcystic adenocarcinoma, foamy gland adenocarcinoma, mucinous adenocarcinoma, signet ring variant of adenocarcinoma, pleomorphic giant cell adenocarcinoma, sarcomatoid adenocarcinoma
Epidemiology
- Second most common cancer and second leading cause of cancer related death in American men (SEER data available at NIH: Cancer Stat Facts - Common Cancer Sites [Accessed 10 December 2021])
- 92% of U.S. cases diagnosed in men aged 55+ years; 19.5% in men aged 75+ years (SEER data available at NIH: Cancer Stat Facts - Prostate Cancer [Accessed 10 December 2021])
- Found at autopsy in 40% of men age 60+ years (J Natl Cancer Inst 2013;105:1050)
- Incidental prostate cancer reported in about 25% of cystoprostatectomies performed for treatment of bladder cancer (J Urol 2017;197:385)
- Globally, highest age standardized rates in Oceania, North America, Europe (World J Oncol 2019;10:63)
- Lower rates in developing countries: may be due to different screening programs and diagnostic pathways
- Higher incidence in men of African heritage (World J Oncol 2019;10:63)
Sites
- Most tumors are multifocal (Eur Urol 2019;75:498)
- 75 - 80% are posterior / posterolateral peripheral zone
- Approximately 13 - 20% are in transition (periurethral) zone (Prostate 1997;30:130, Urology 1994;43:11)
- Most clinically significant cancers arise in the peripheral zone that is sampled by needle biopsies
- Transition zone prostate cancer is associated with favorable pathologic features and better recurrence free survival (Prostate 2015;75:183)
- Less frequently involved anterior prostate most likely due to inadequate sampling using standard biopsy approach (Histopathology 2012;60:142)
Pathophysiology
- Germline variants can increase risk of prostate adenocarcinoma; see Molecular / cytogenetics description
- Somatic mutations in genes such as ERG, ETV1/4, FLI1, SPOP, FOXA1, IDH1, PTEN, TP53, MYC, CDH1 (Cell 2015;163:1011, Adv Anat Pathol 2020;27:11)
- Most common somatic genomic rearrangement is fusion of the androgen regulated gene TMPRSS2 with a member of the ETS transcription family (Adv Anat Pathol 2020;27:11)
Etiology
- Obesity increases risk (Oncotarget 2018;9:34485)
- Nonmodifiable risk factors: age, race and family history (Rev Urol 2002;4:S3)
- Genetic susceptibility linked to African heritage (Rev Urol 2002;4:S3)
- Increased risk with first degree relative with prostate cancer (Rev Urol 2002;4:S3)
- BRCA2 mutations increase risk by 5 fold; BRCA2 associated cancers occur at a lower age and have worse survival outcomes (Saudi Med J 2020;41:9, Adv Anat Pathol 2020;27:11)
- Additional germline variants associated with increased cancer risk occur in HOXB13 (Adv Anat Pathol 2020;27:11)
- Increased risk in Lynch syndrome (Genet Med 2014;16:553)
- Numerous single nucleotide polymorphisms (SNPs) that have a low to moderate effect on risk / progression have been identified (Am J Transl Res 2021;13:3868)
- High levels of IGF1 may confer increased risk (Novartis Found Symp 2004;262:193)
Clinical features
- Generally asymptomatic unless locally advanced or metastatic
- Often discovered following investigation of nonspecific lower urinary tract symptoms
- Digital rectal examination (DRE): prostate may feel normal or may be enlarged / asymmetrical / hard / have a palpable nodule present
Diagnosis
- Generally diagnosed by systematic transrectal ultrasound guided prostate biopsies
- Transperineal needle biopsies increasingly used as associated with lower risk of infection
- Prebiopsy MRI followed by systematic biopsies supplemented with targeted biopsies from any radiological abnormality leads to better identification of clinically significant prostate cancer than systematic prostate biopsy alone (Lancet 2017;389:815)
- Incidental prostate cancer sometimes diagnosed in transurethral resections
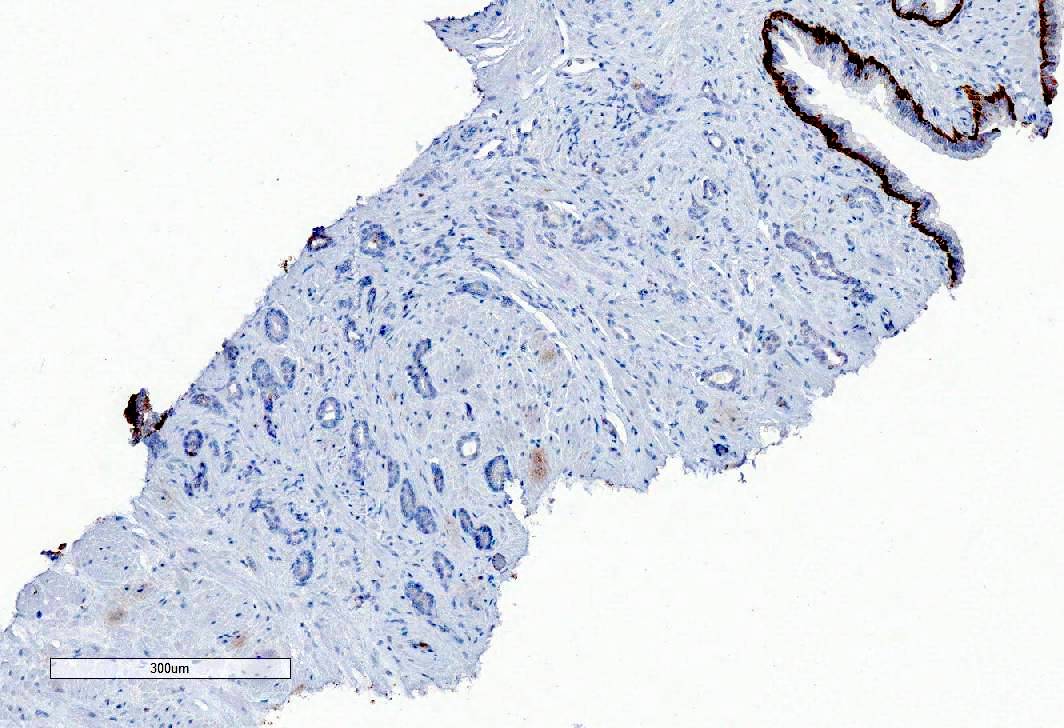
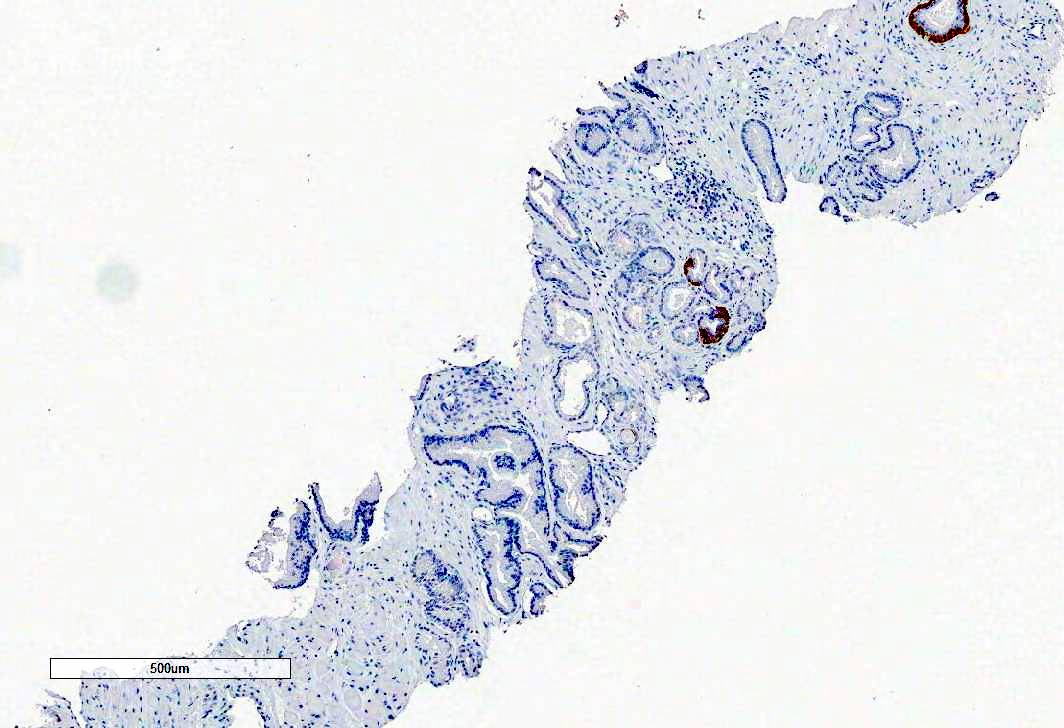
- Immunohistochemistry with basal cell markers (HMWCK, p63) and AMACR used to establish the diagnosis in equivocal cases
Laboratory
- Raised serum PSA
- Different PSA cutoffs have been used to prompt prostate needle biopsy
- Age specific cutoffs, PSA velocity (rate of change in PSA over time) and PSA density (PSA per unit prostate volume - ng/mL/cc) may increase sensitivity and specificity of PSA testing (NCCN Guidelines: Prostate Cancer Early Detection [Accessed 10 December 2021])
- U.S. Preventative Services Task Force (USPSTF) recommends against PSA based screening for prostate cancer in men 70 years and older
- For men aged 55 - 69 years, periodic PSA based screening should be an individual choice
- Screening in this age group offers a small potential benefit of reducing the chance of death from prostate cancer in some men; however, many men will experience potential harm (USPSTF: Recommendation - Prostate Cancer - Screening [Accessed 27 January 2022])
- American Urological Association (AUA) does not recommend PSA screening in men under age 40 years or in men aged 40 - 54 years at average risk
- For men age 55 - 59 years, shared decision making is desirable
- For men aged 70 years and over or men with < 10 - 15 year life expectancy, PSA screening is not recommended (J Urol 2013;190:419)
- Potential urine biomarker for prostate cancer is PCA3 (Adv Anat Pathol 2020;27:11)
Radiology description
- Ultrasound scan (USS) generally used to guide prostate biopsies; prostate cancer may appear hypoechoic but USS neither sensitive nor specific
- Multiparametric MRI commonly used for local tumor staging; may also be used to identify abnormalities for targeting at biopsy
- MRI abnormalities generally reported using either PI-RADS (Prostate Imaging - Reporting and Data System) or Likert score
- CT scan used to identify metastatic disease in lymph nodes
- Bone scan used to detect bony metastases
- PET scan used to detect micrometastatic disease in selected patients, such as men with raised PSA levels after treatment
Prognostic factors
- Biopsy: tumor extent (mm or percentage core involvement), grade (Gleason score and grade group), perineural invasion, extraprostatic extension
- Radical prostatectomy: tumor size, Gleason score and grade group, stage, margin status
- Cribriform morphology and intraductal carcinoma associated with invasive prostate cancer are adverse prognostic indicators (Transl Androl Urol 2018;7:145)
- Small cell carcinoma component is associated with aggressive behavior and treated differently
- Some expert groups recommend incorporating intraductal component into the Gleason score while others recommend reporting it separately in a comment (Am J Surg Pathol 2020;44:e87, Arch Pathol Lab Med 2021;145:461, Histopathology 2021;78:231)
Case reports
- 53 year old man presented with prostate adenocarcinoma with retained basal cells in lymph node metastases (Histopathology 2016;69:338)
- 56 year old man with gait disturbance was found to have disseminated prostate carcinoma with bone metastases and a germline mutation in BRCA2 (J Korean Med Sci 2017;32:377)
- 60 year old man presented with metastatic prostate adenocarcinoma to supraclavicular lymph nodes (J Clin Diagn Res 2017;11:PD06)
- 64 year old man with asymptomatic case of oncocytic variant of prostate adenocarcinoma (World J Clin Oncol 2017;8:289)
Treatment
- Preoperative risk stratification based on serum PSA, clinical stage, biopsy parameters (tumor extent, grade, cribriform morphology, intraductal carcinoma, perineural invasion)
- Primary treatment options based on preoperative risk stratification:
- Active surveillance
- Focal therapy (cryotherapy, high intensity ultrasound)
- Radical prostatectomy
- Brachytherapy
- External beam radiotherapy
- Hormone therapy (e.g., luteinizing hormone releasing hormone [LHRH] analogues, antiandrogens)
- Orchidectomy (rare in contemporary practice)
- Chemotherapy (for metastatic disease)
- Postprostatectomy options:
- Generally, PSA monitoring and early salvage therapy if rising serum PSA
- Less commonly adjuvant therapy for high stage disease or margin positivity
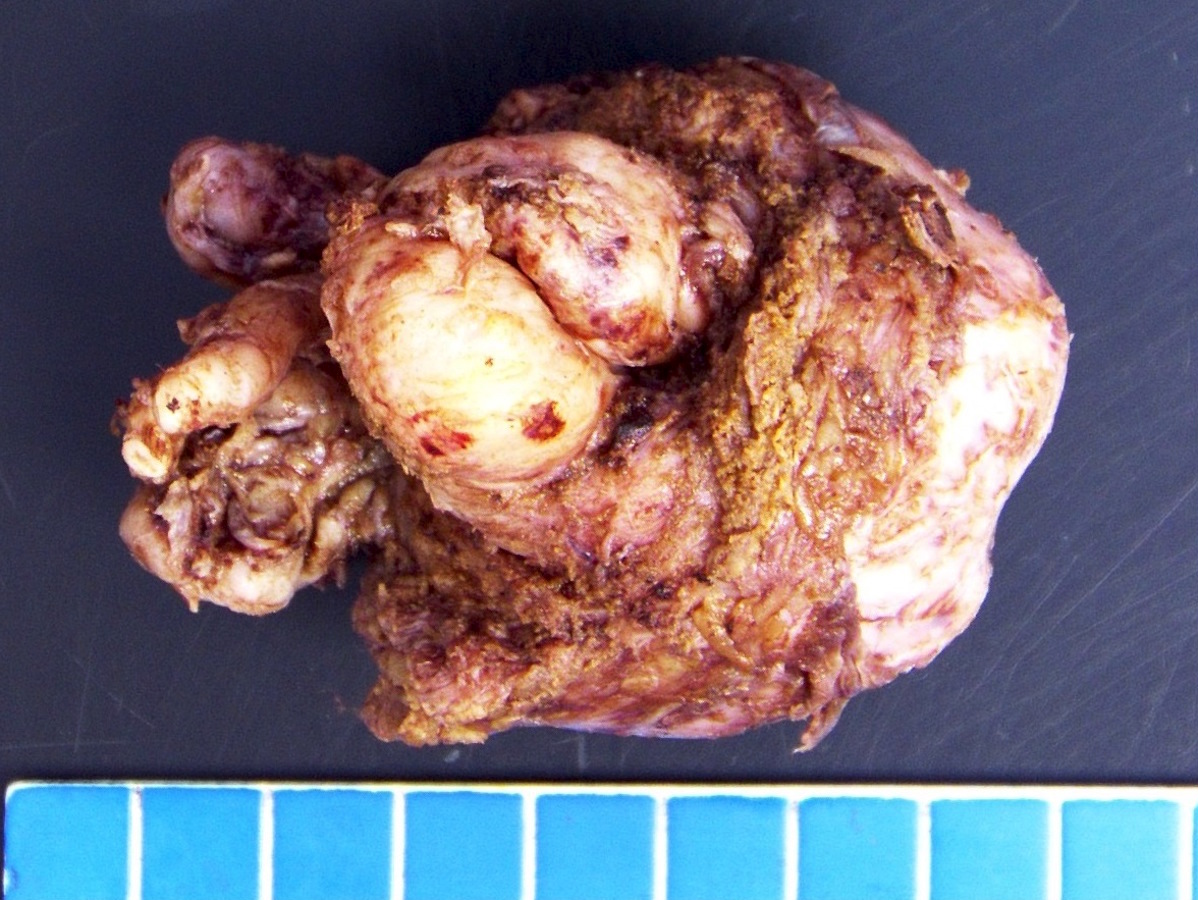
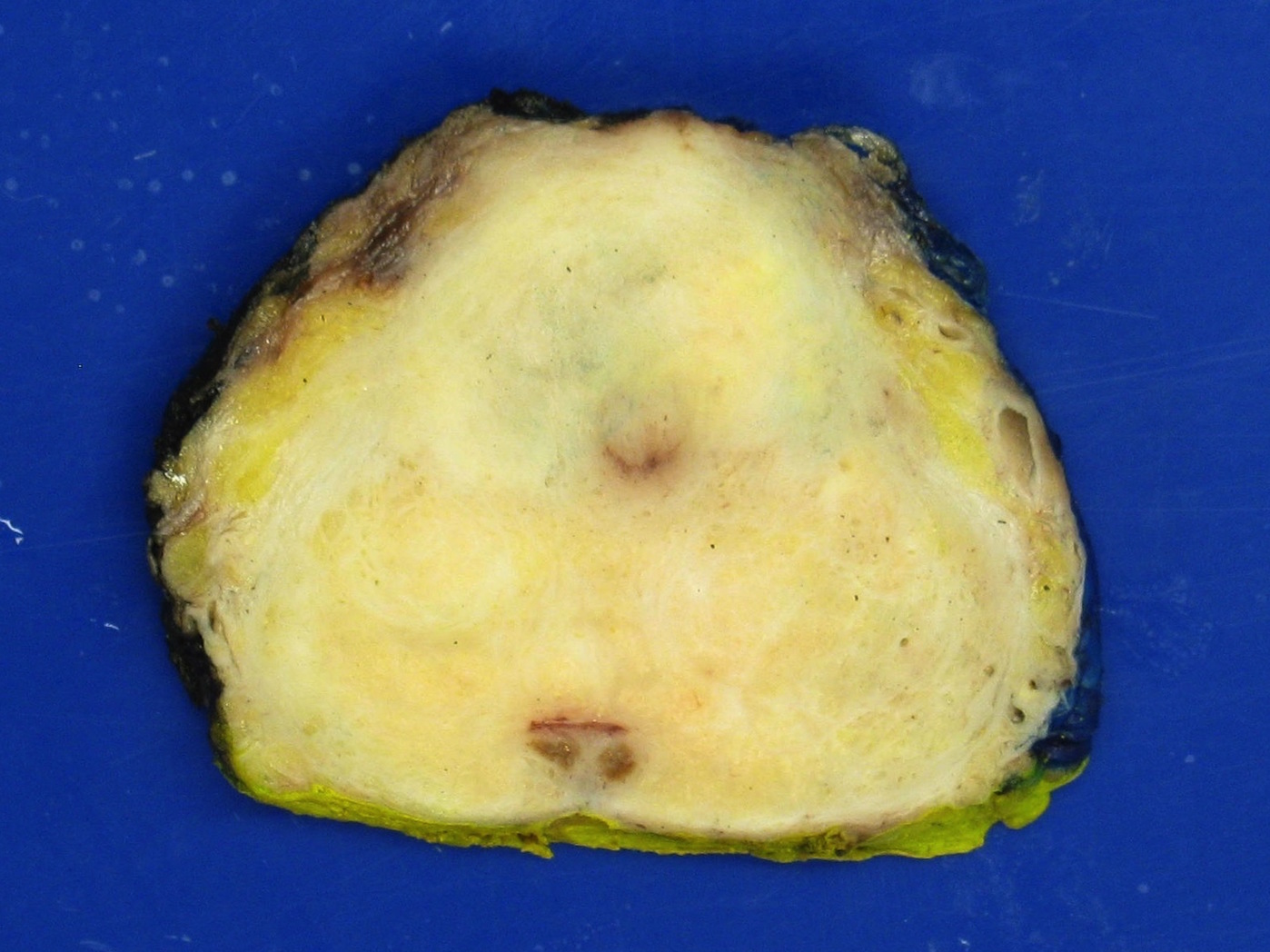
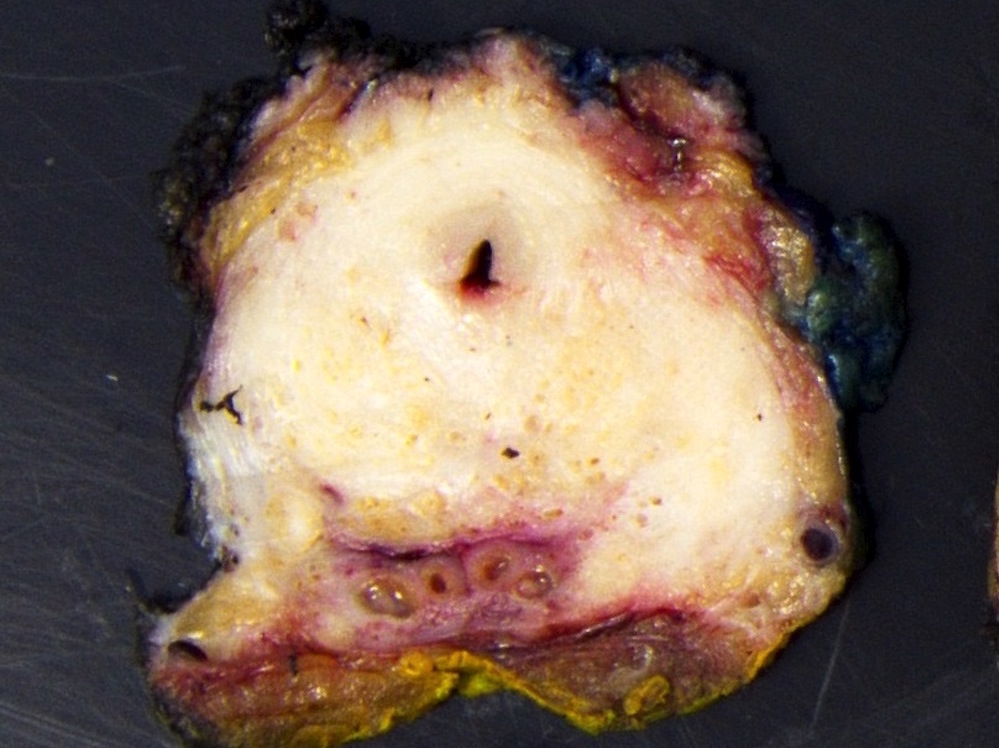
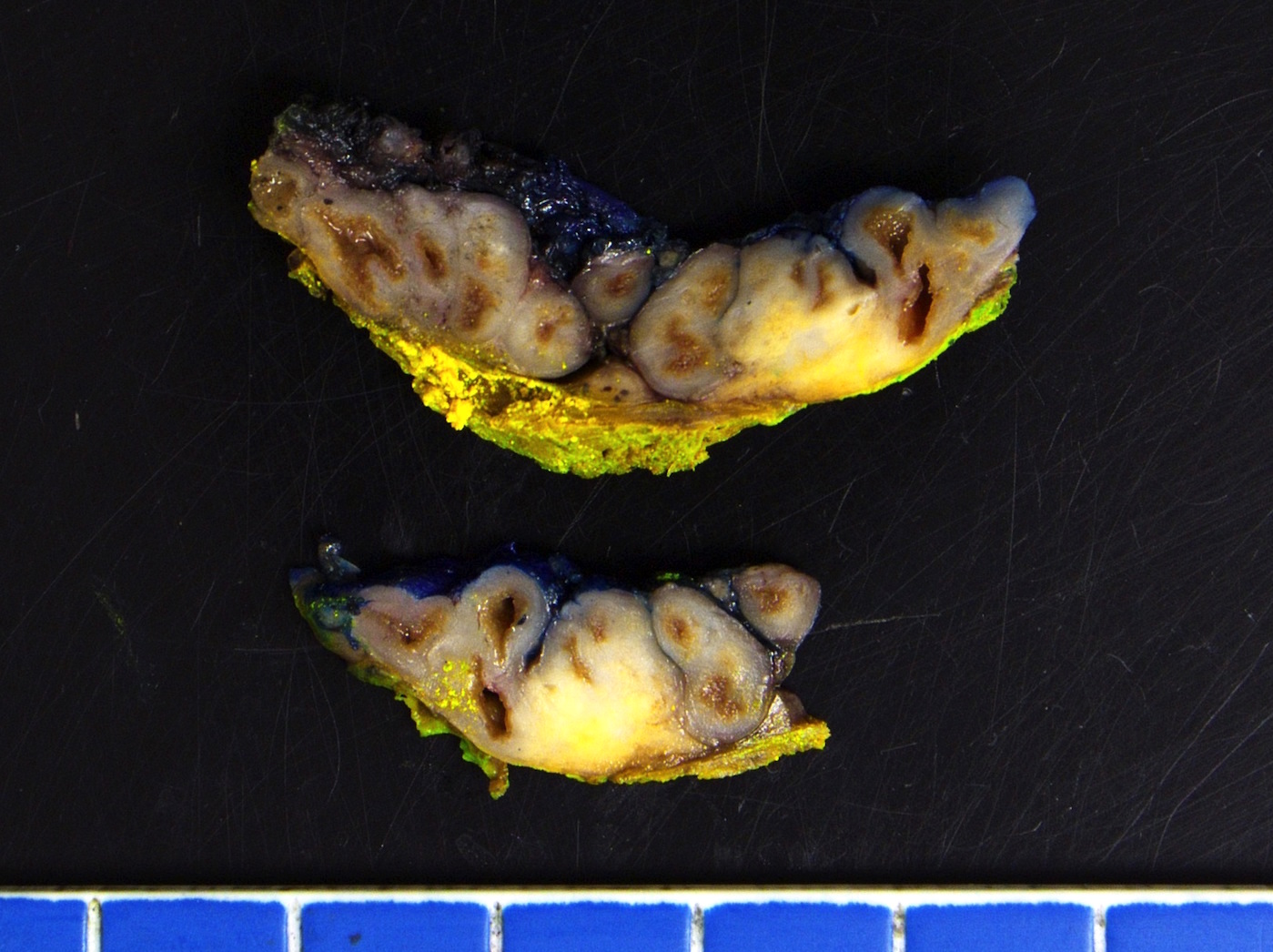
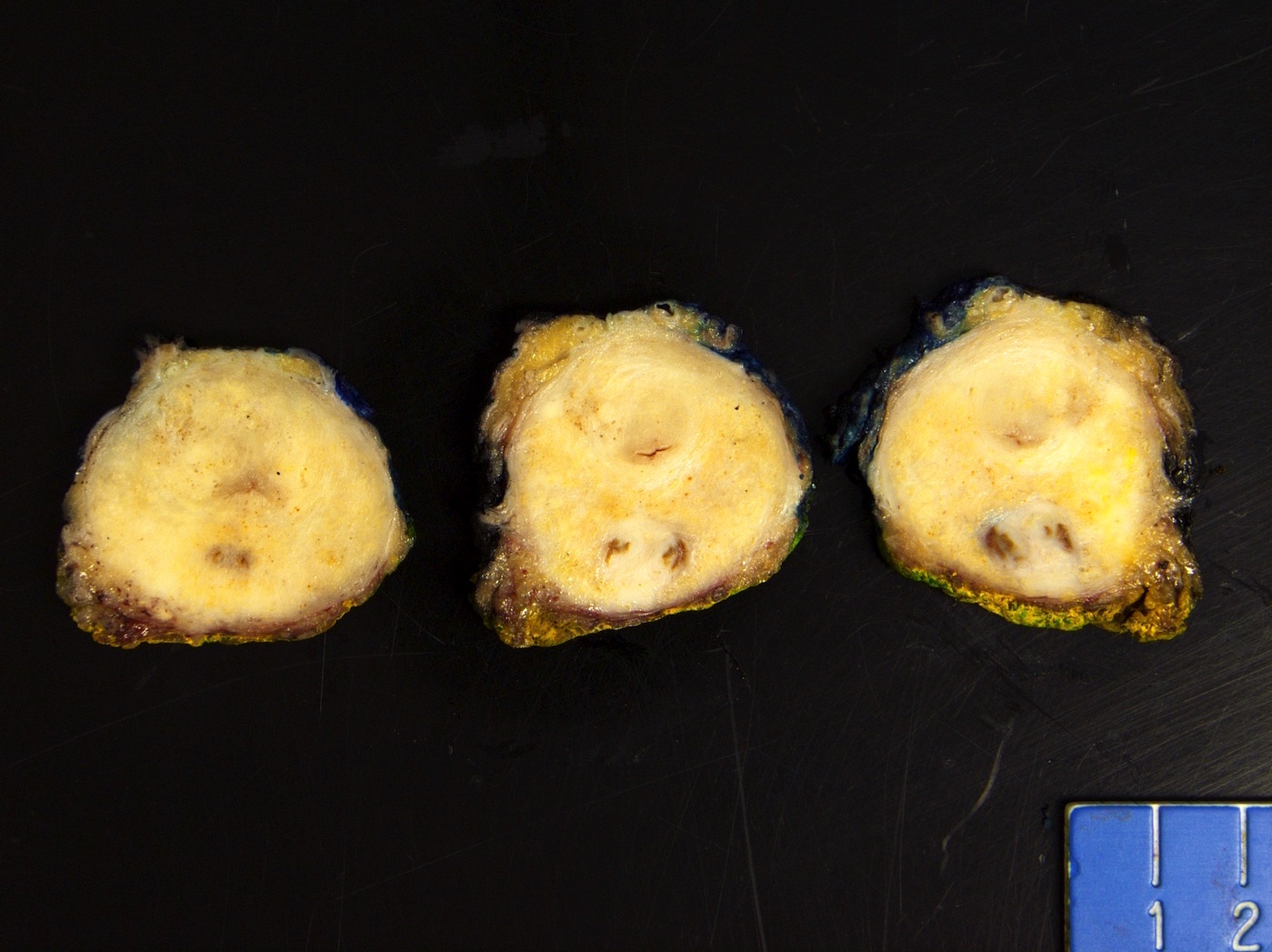
Gross description
- Often grossly inapparent
- May form a cream mass
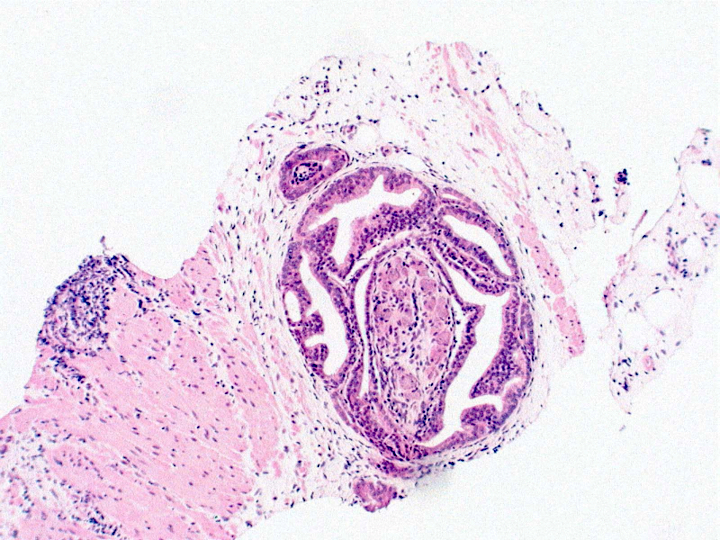
Gross images
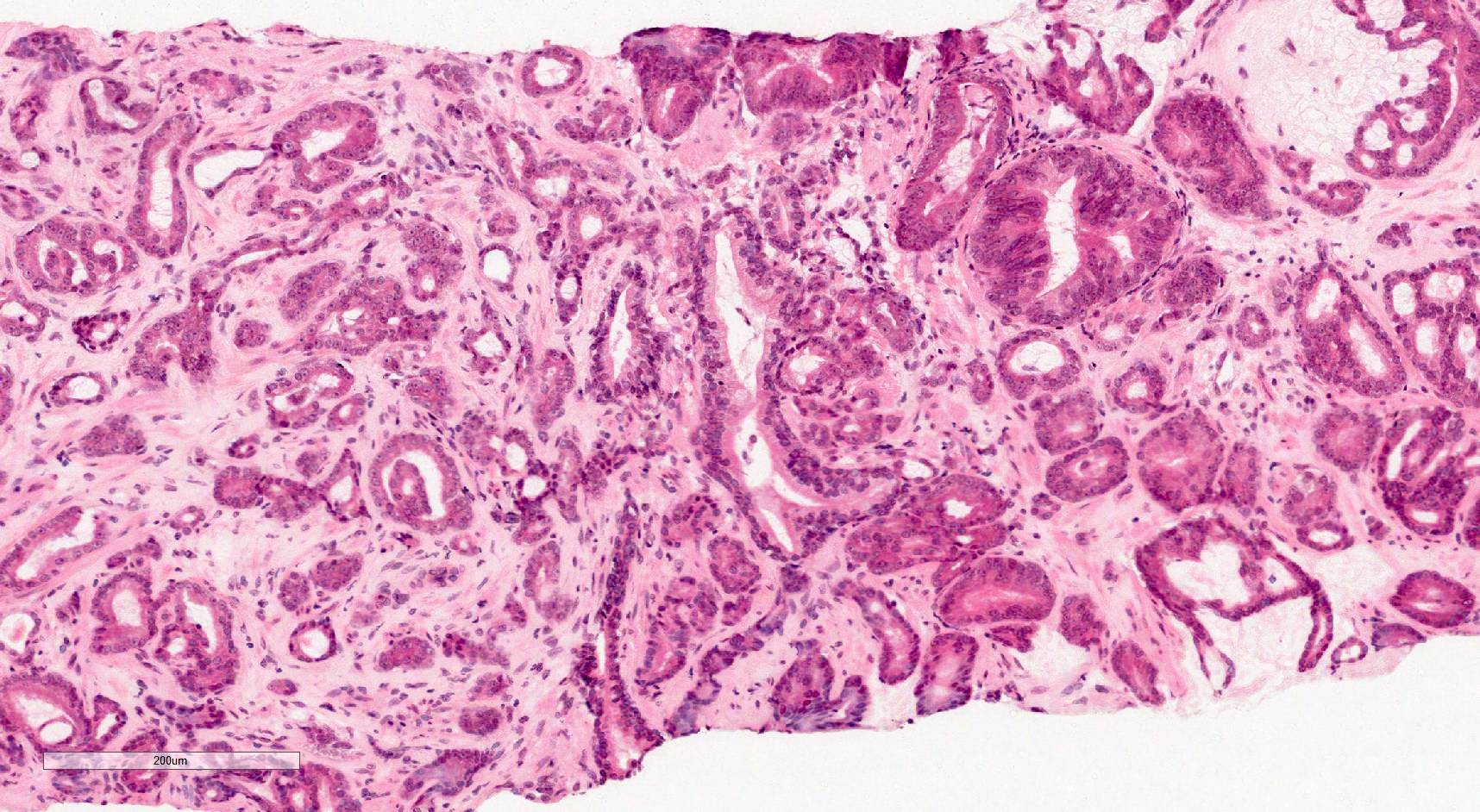
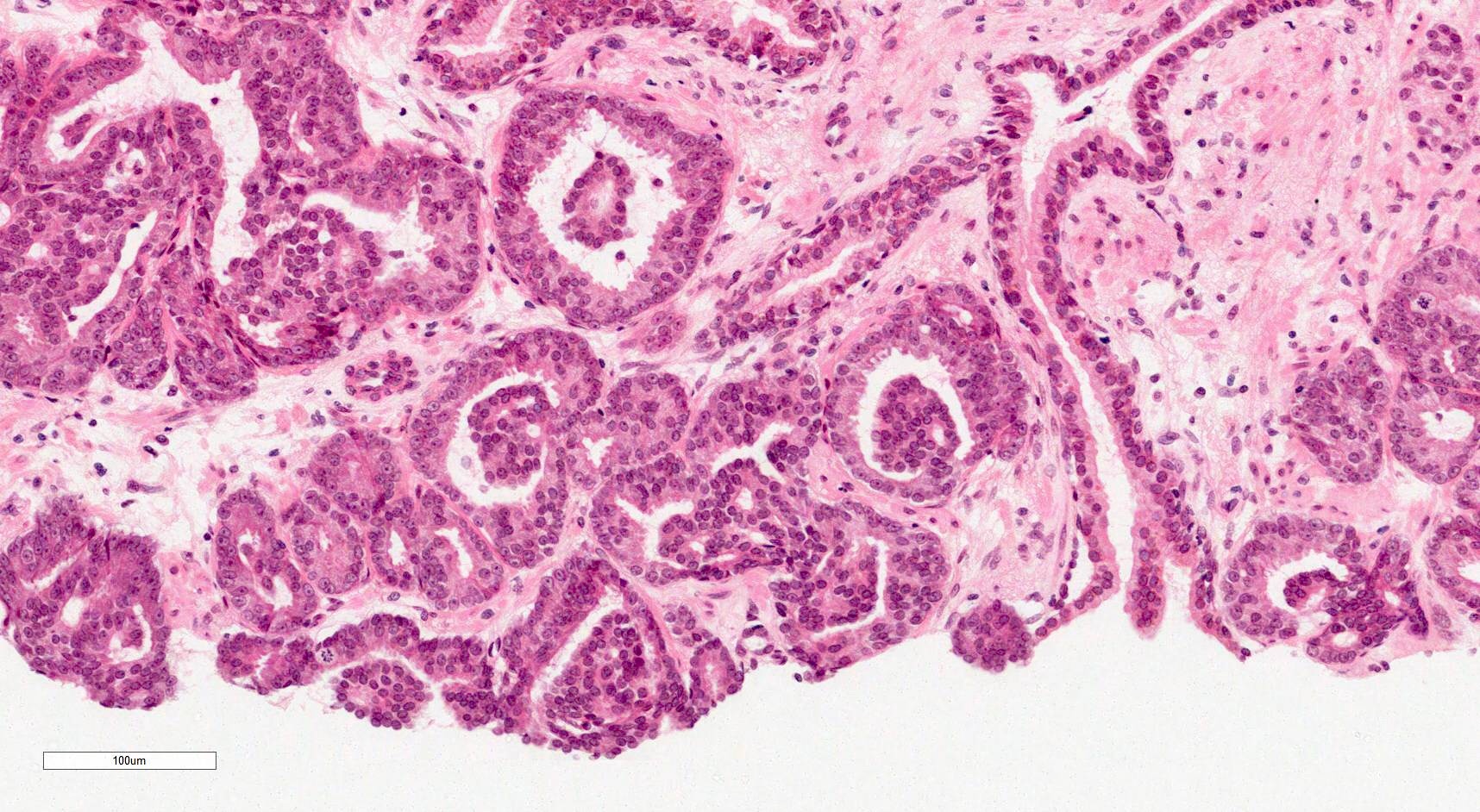
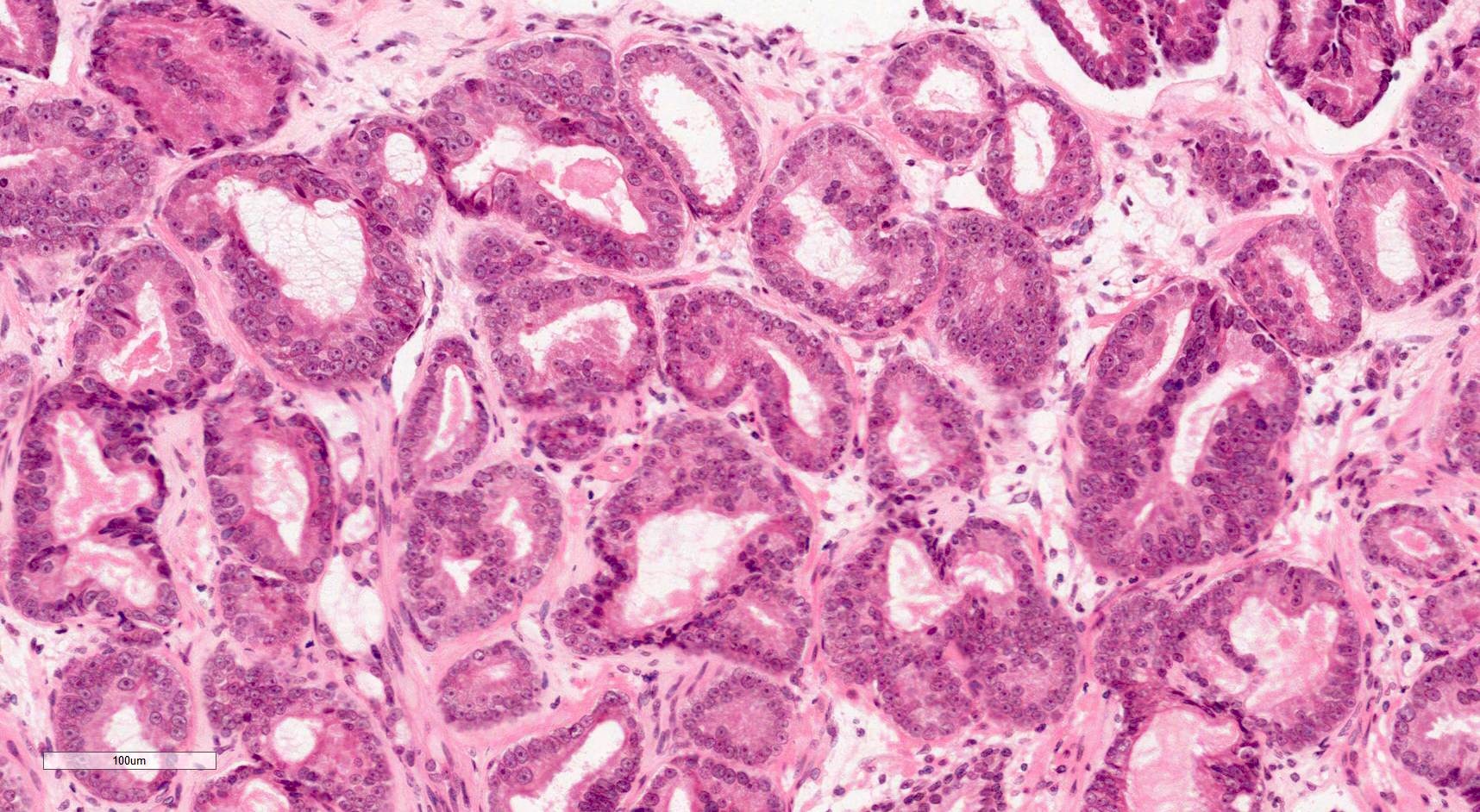
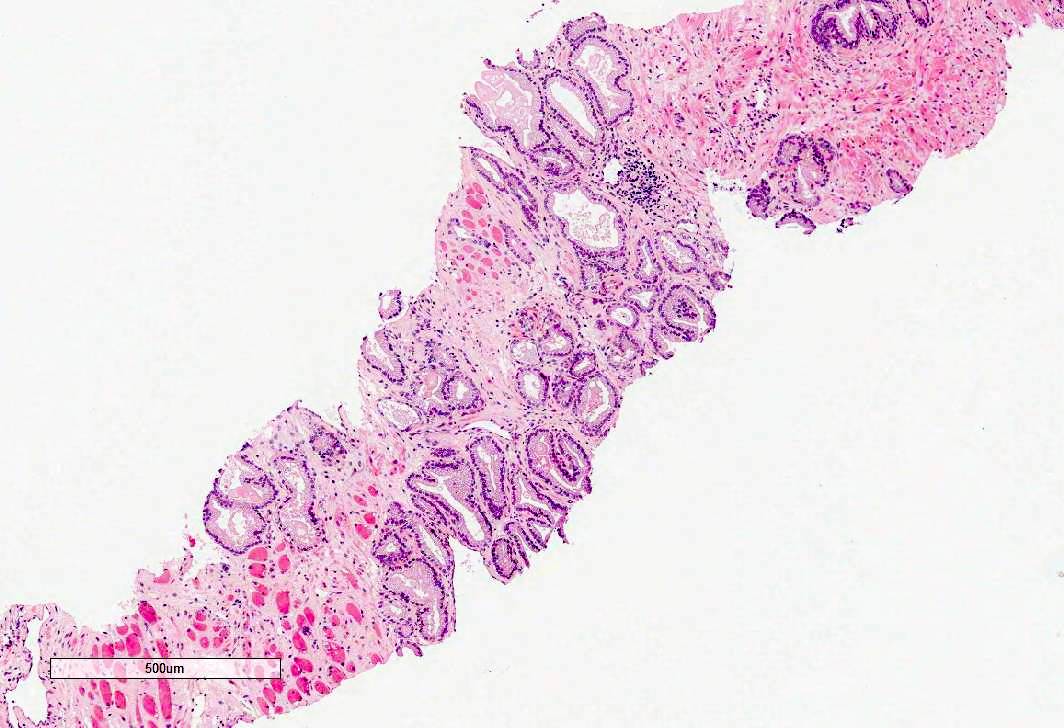
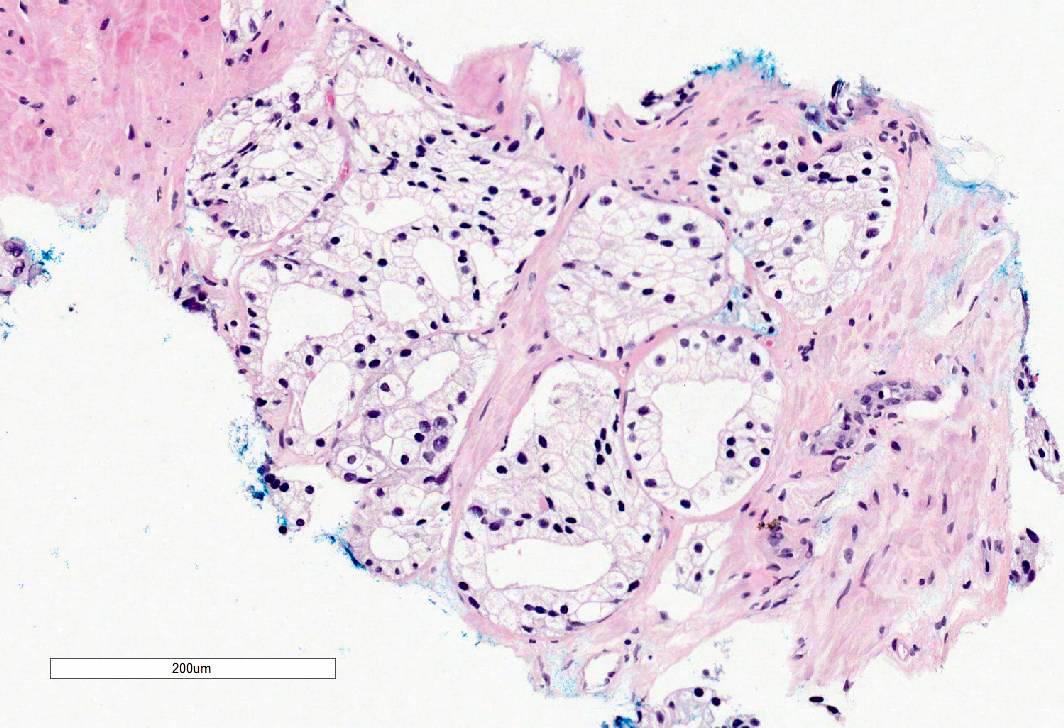
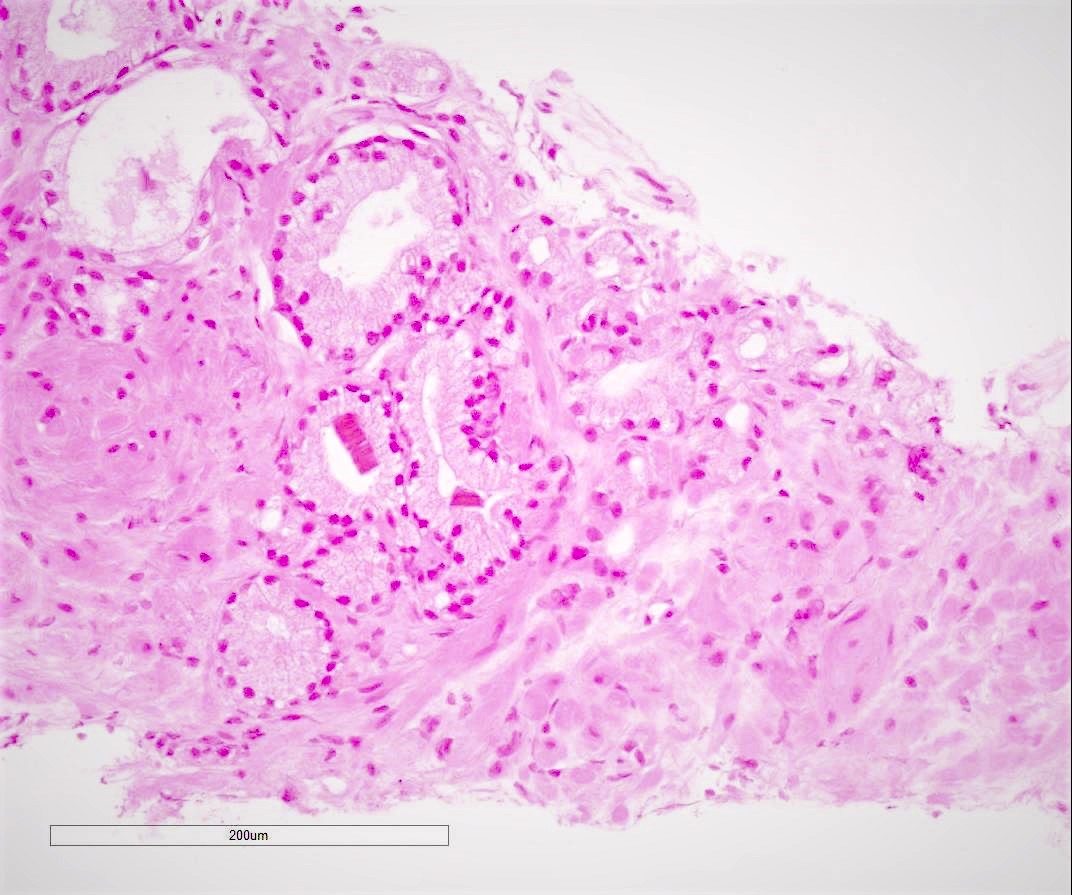
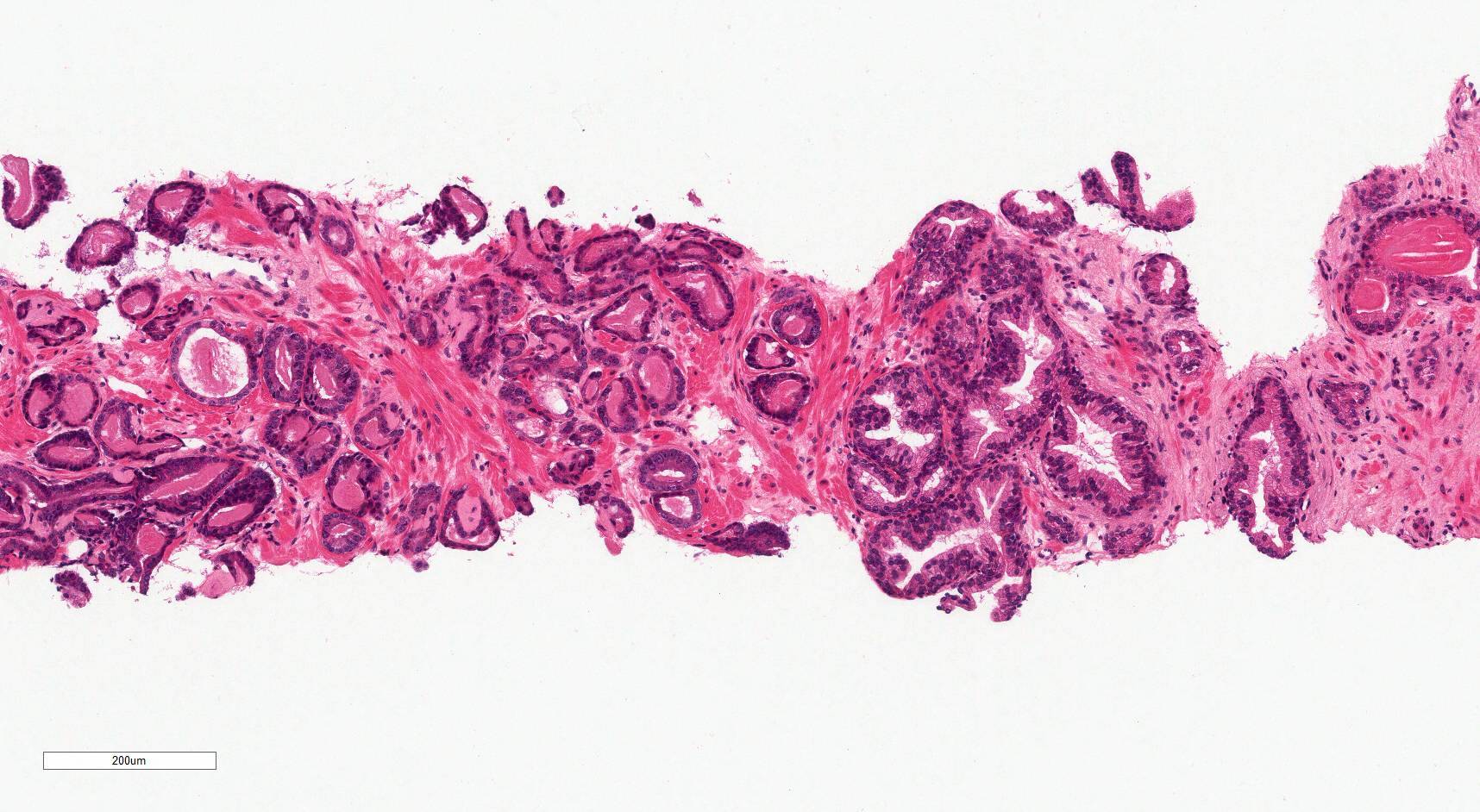
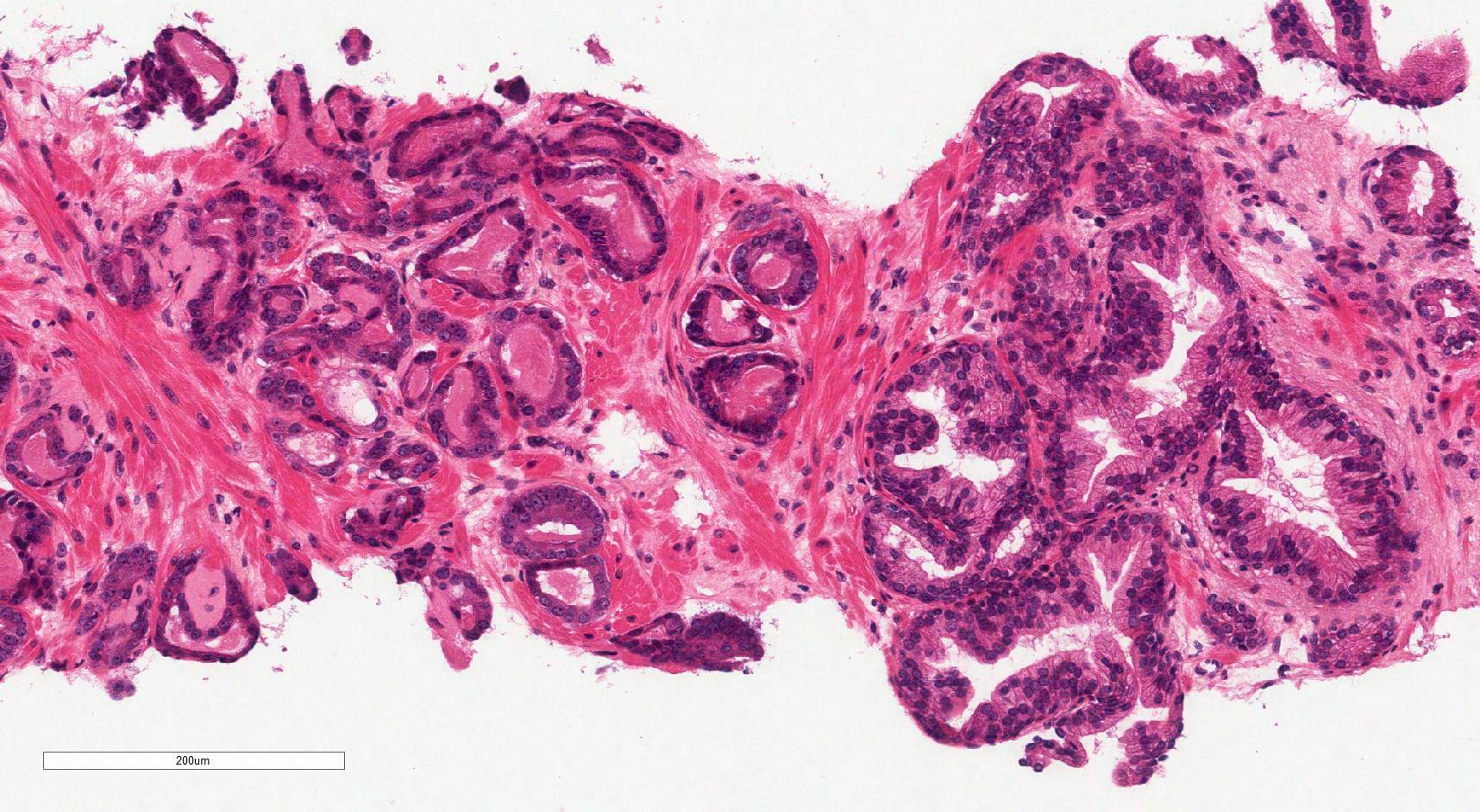
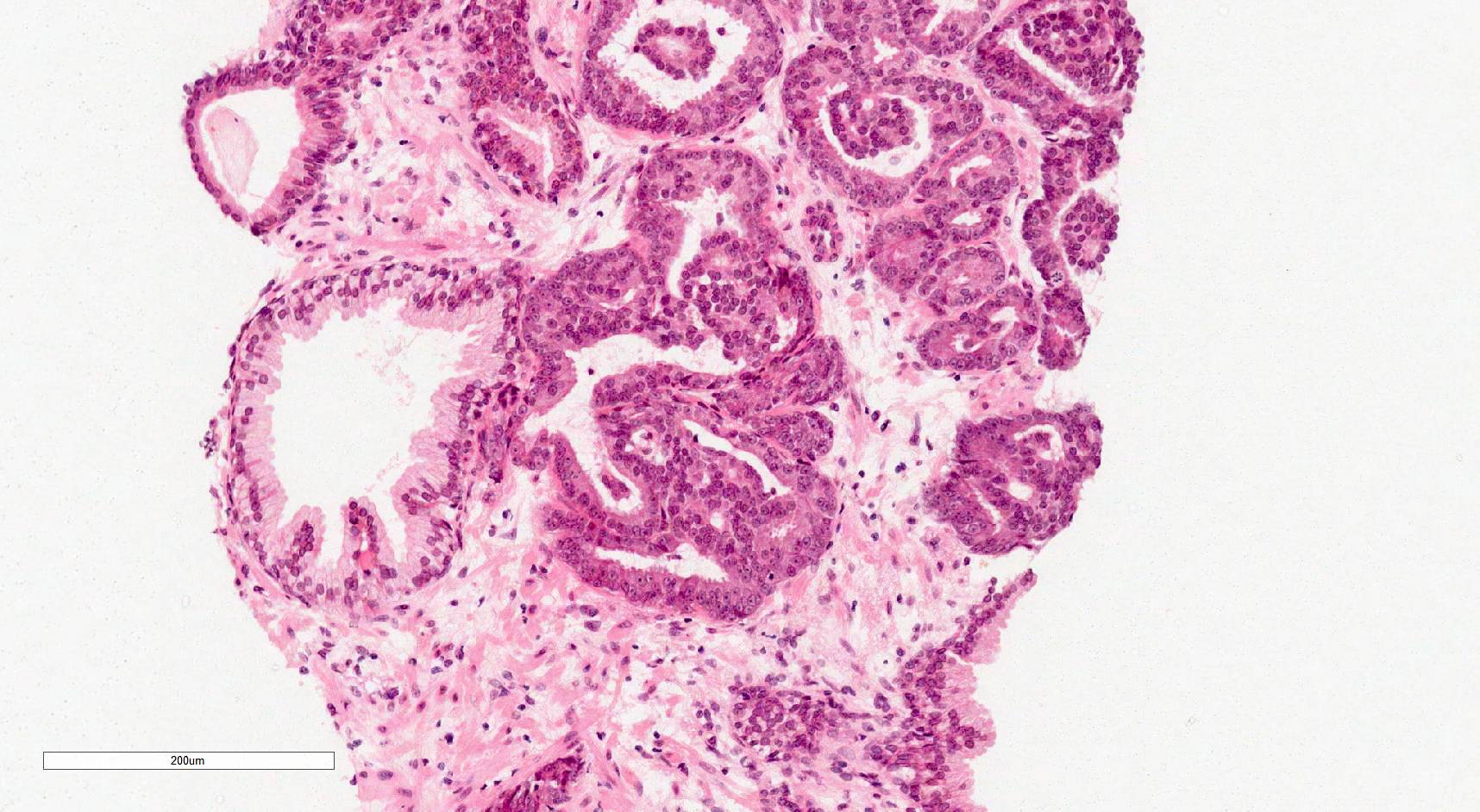
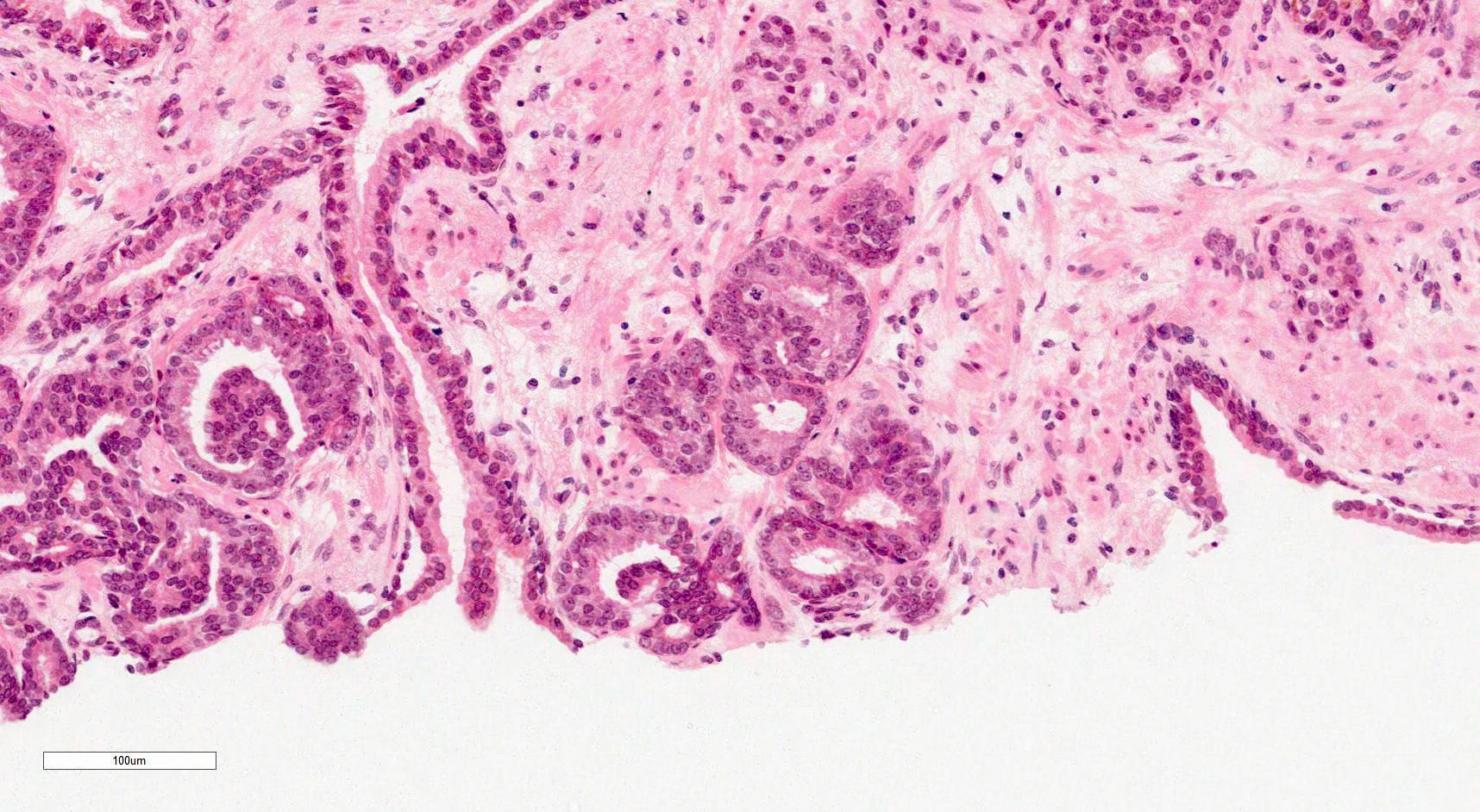
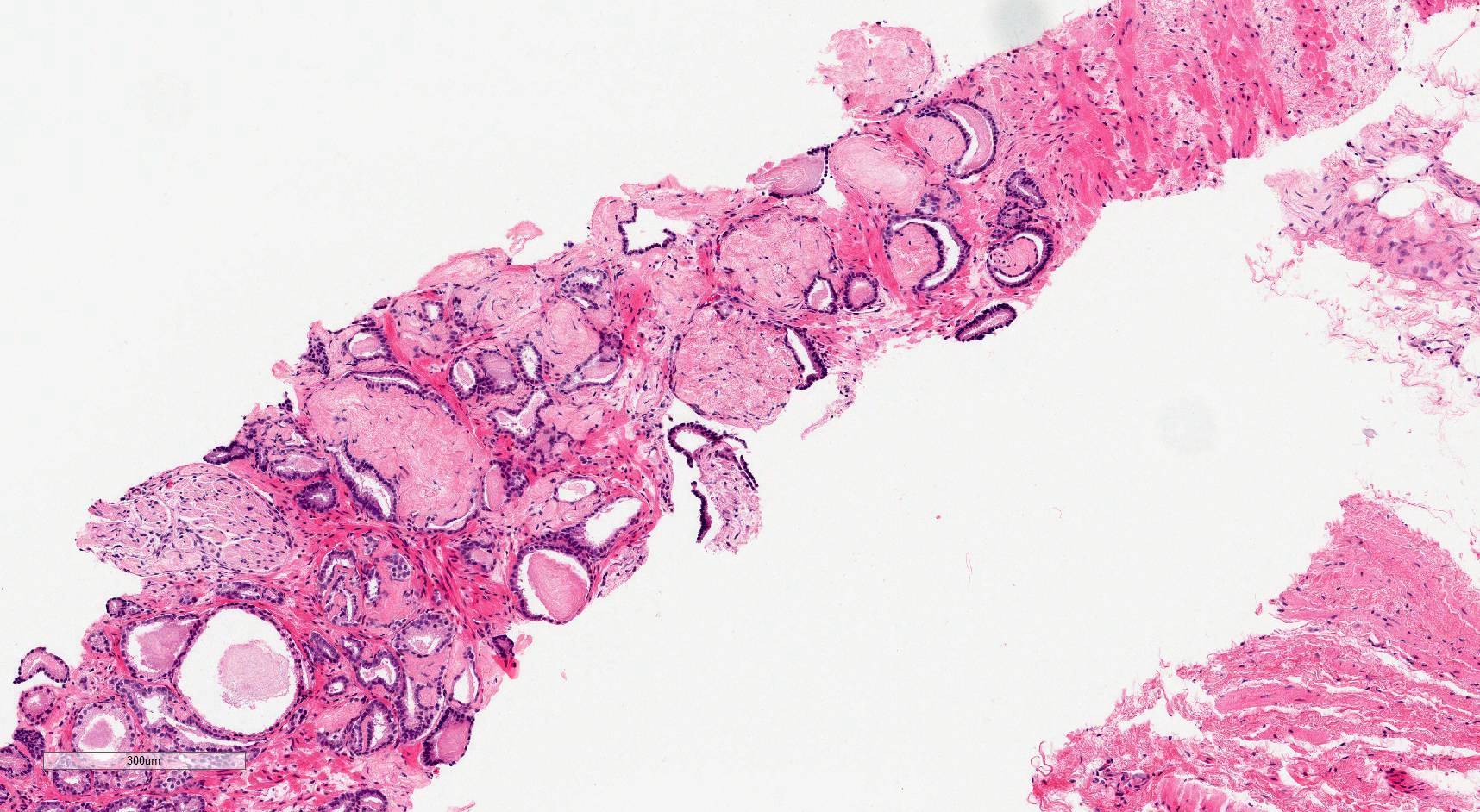
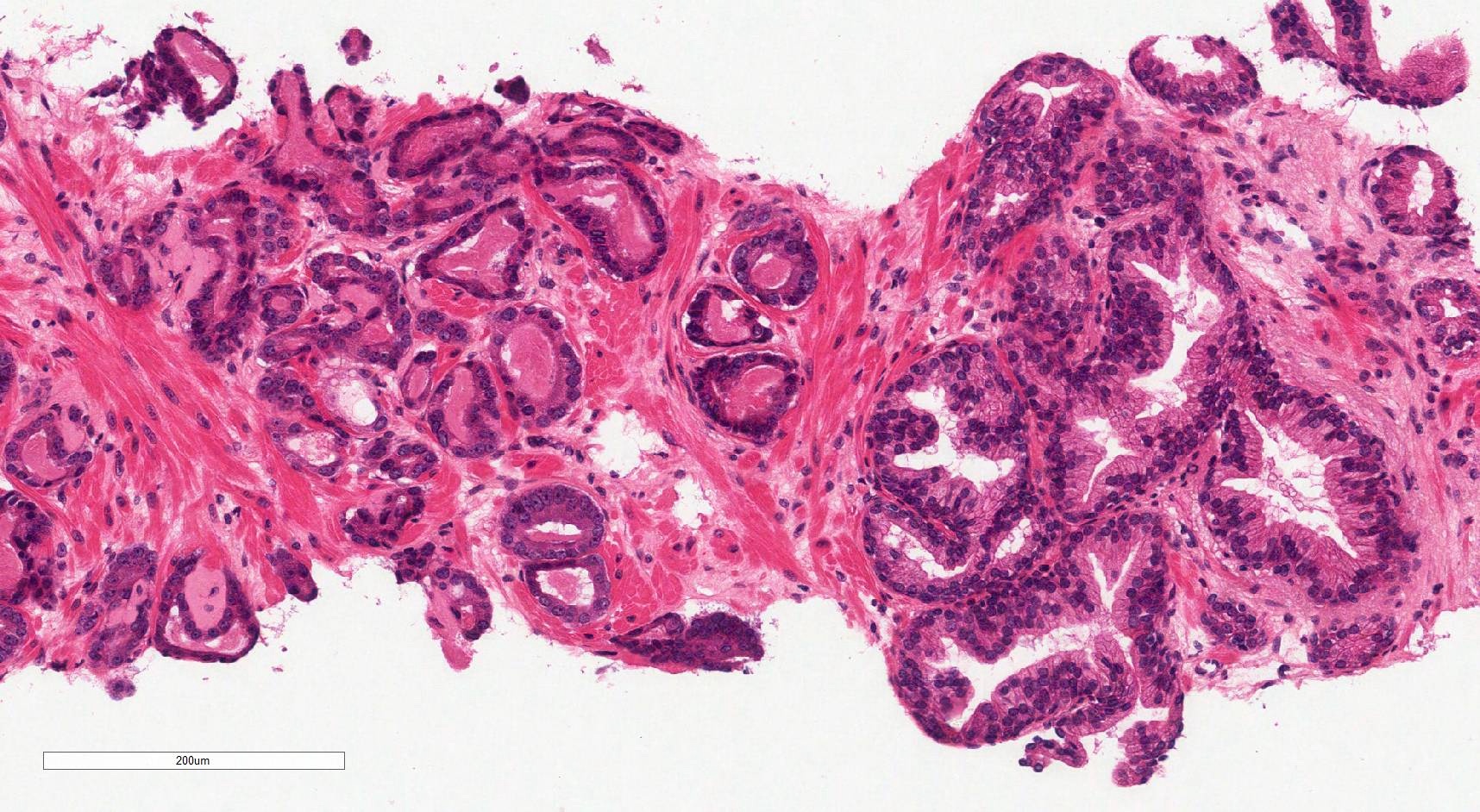
Microscopic (histologic) description
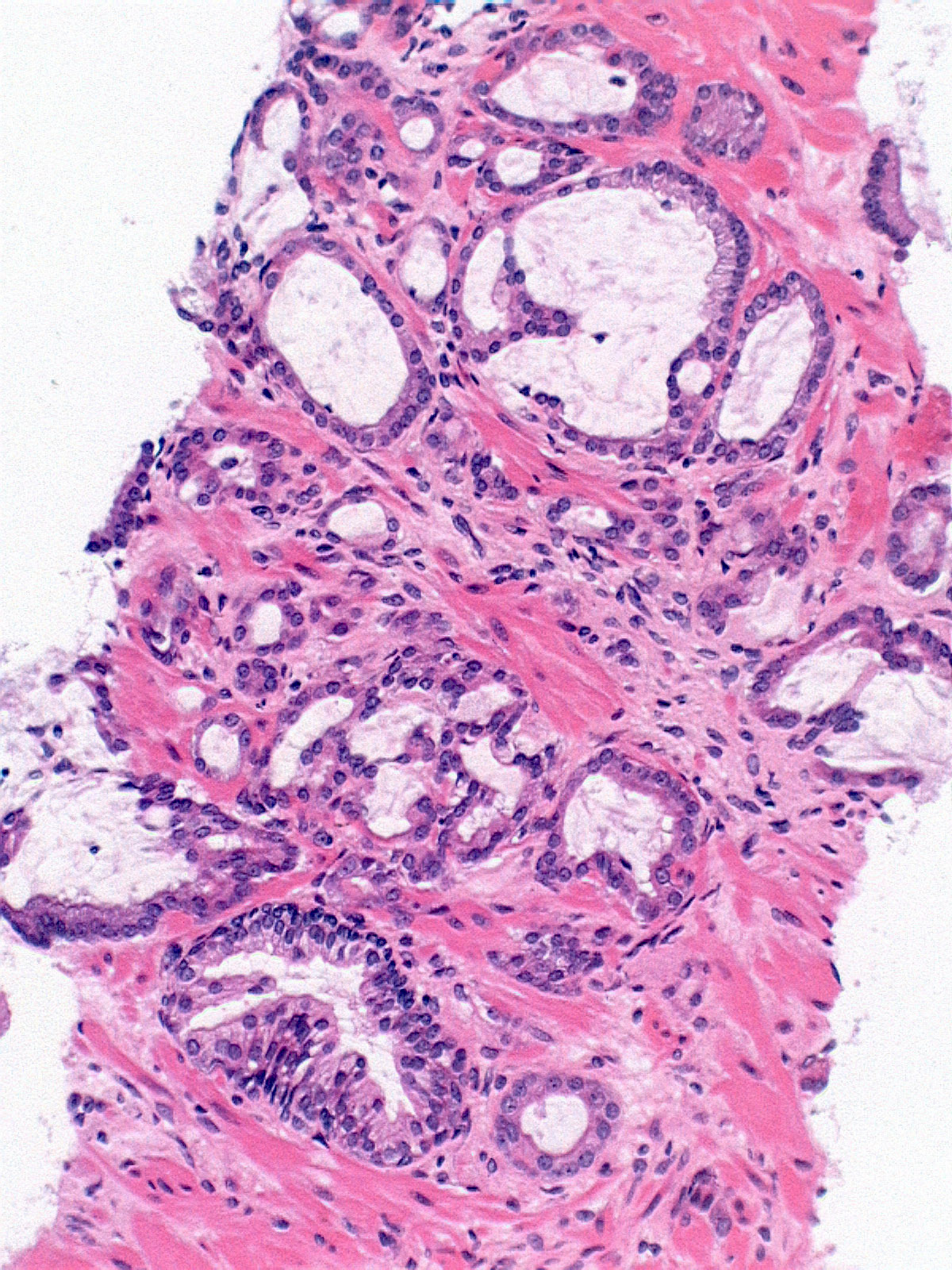
- Gleason grading is based on the architecture of the tumor
- Gleason grades represent a morphological spectrum from well formed glands (pattern 3) to increasingly complicated glandular proliferations (pattern 4) to almost no glandular differentiation (pattern 5) (Diagnostic Histopathology 2019;25:371)
- Glandular crowding and infiltrative growth pattern
- Nuclear enlargement, nucleolar prominence
- Round generally monomorphic nuclei
- Amphophilic cytoplasm
- Mitoses
- Apoptotic bodies
- Stromal desmoplasia
- Intraluminal contents: crystalloids, pink amorphous secretions, blue mucin
- Glomerulations, collagenous micronodules (mucinous fibroplasia)
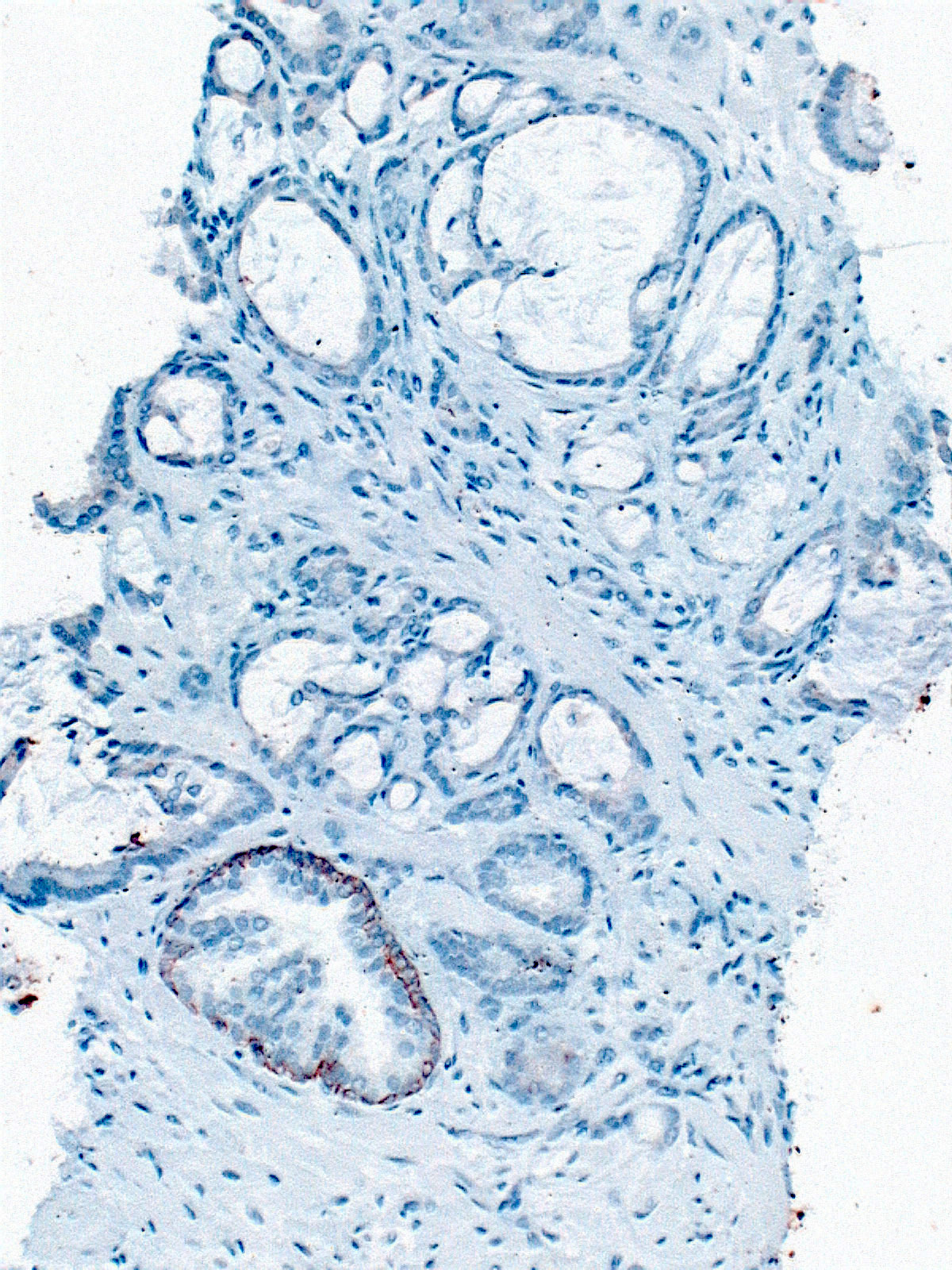
- Absence of basal cell layer (generally requires immunohistochemical confirmation)
- Reference: Cold Spring Harb Perspect Med 2017;7:a030411
Microscopic (histologic) images
Contributed by Murali Varma, M.B.B.S.
Virtual slides
Cytology description
- Urine cytology for detecting prostate cancer has a very low sensitivity (Prostate Cancer Prostatic Dis 2019;22:362)
- Urine cytology is not used clinically in the diagnosis of prostate cancer
- FNA of metastatic prostate cancer to a lymph node may show microacinar complexes / cell clusters / single cells with fragile cytoplasm and prominent nucleoli (Diagn Cytopathol 2007;35:565)
Positive stains
- PSA
- NKX3.1
- AMACR (P504S, racemase)
- Prostein (P501S)
- PSMA
- Rare tumors may have aberrant expression of p63 (Mod Pathol 2015;28:446)
- Reference: Am J Surg Pathol 2014;38:e6
Negative stains
- CK7
- CK20
- High molecular weight cytokeratins (34 beta E12, CK5, CK5/6)
- p63
- CDX2
- GATA3
- TTF1
- Reference: Am J Surg Pathol 2014;38:e6
Molecular / cytogenetics description
- Prostate cancer is a heritable disease
- Family history of a first degree relative with prostate cancer increases the risk of developing prostate cancer by 2 fold (Nat Rev Urol 2014;11:18)
- 30 - 40% of familial risk is due to genetic factors (Adv Anat Pathol 2020;27:11)
- Genetic factors include highly penetrable rare variants and more common low to moderate risk variants (Adv Anat Pathol 2020;27:11)
- Highly penetrant variants occur in BRCA2 and HOXB13
- Over 280 SNPs have been identified as prostate cancer risk factors (Adv Anat Pathol 2020;27:11)
- For most SNPs, the molecular mechanism of cancer association is generally unknown, as they occur in noncoding regions of the genome (Adv Anat Pathol 2020;27:11)
- Somatic mutations occur in genes such as ERG, ETV1/4, FLI1, SPOP, FOXA1, IDH1, PTEN, TP53, MYC, CDH1 (Cell 2015;163:1011, Adv Anat Pathol 2020;27:11)
- Most common somatic genomic rearrangement is fusion of the androgen regulated gene TMPRSS2 with a member of the ETS transcription family (Adv Anat Pathol 2020;27:11)
- Somatic mutation profiles of prostate cancer are associated with clinical and pathological outcomes
- There are 7 major subtypes, which are defined by either specific gene fusions of ETS transcription family members (ERG, ETV1, ETV4 and FLI1) or mutations (SPOP, FOXA1, IDH1) (Oncotarget 2018;9:14723)
- Different subtypes have different molecular profiles, for example (Oncotarget 2018;9:14723):
- ETS subset (59% of cases) are enriched in PTEN mutations
- SPOP mutant subset (11%) of cases have distinct somatic copy number alteration profiles, including deletions of CHD1, 6q and 2q
Sample pathology report
- Prostate core biopsies:
- Acinar adenocarcinoma (see comment)
- Comment:
- Number of cores involved:
- Right 1/6 cores
- Location(s): right apex
- Left 0/6 cores
- Total number of cores involved: 1/12
- Greatest length of cancer in a core: 5 mm (40%)
- No evidence of perineural invasion or extraprostatic extension
- Gleason score: 3+4=7 (10% pattern 4, no cribriform morphology)
- Grade group 2
- Number of cores involved:
- Radical prostatectomy:
- Histological tumor type: acinar adenocarcinoma
- Gleason score:
- Primary Gleason grade: 3
- Secondary Gleason grade: 3
- Tertiary Gleason grade (< 5%): not applicable
- Gleason score: 3+3=6
- Grade group: 1
- Location of dominant tumor: right apex
- Extraprostatic extension: not identified
- Bladder neck: not involved
- Seminal vessels: not involved
- Margin status: not involved
- Lymphovascular invasion: not identified
- Regional lymph node status:
- Number of nodes examined: 9
- Number of positive lymph nodes: 0
- Primary tumor: pT2 pN0
Differential diagnosis
- Benign prostate tissue:
- Pale cytoplasm
- Corpora amylacea
- No other intraluminal contents
- Basal cell marker immunoreactivity
- Prostatic atrophy:
- Lobular architecture
- Scant cytoplasm
- Basal cell marker immunoreactivity
- Adenosis:
- Lobular architecture
- Basal cell marker immunoreactivity (often scattered)
- Atypical small acinar proliferation (ASAP):
- Small size
- Lack of significant cytological atypia, including a lack of macronucleoli
- High grade prostatic intraepithelial neoplasia (HGPIN):
- Less architectural atypia
- Maintained basal cells
- Postatrophic hyperplasia:
- Some glands atrophic
- Basal cell marker immunoreactivity (often scattered)
- Partial atrophy:
- Atrophic glands with abundant lateral pale cytoplasm
- Irregularly distributed nuclei
- Basal cell marker immunoreactivity (often scattered)
- Radiation atypia:
- Glandular atrophy
- Nuclear irregularity and pleomorphism
- Atypical stromal cells
- Basal cell marker immunoreactivity
- Urothelial carcinoma:
Board review style question #1
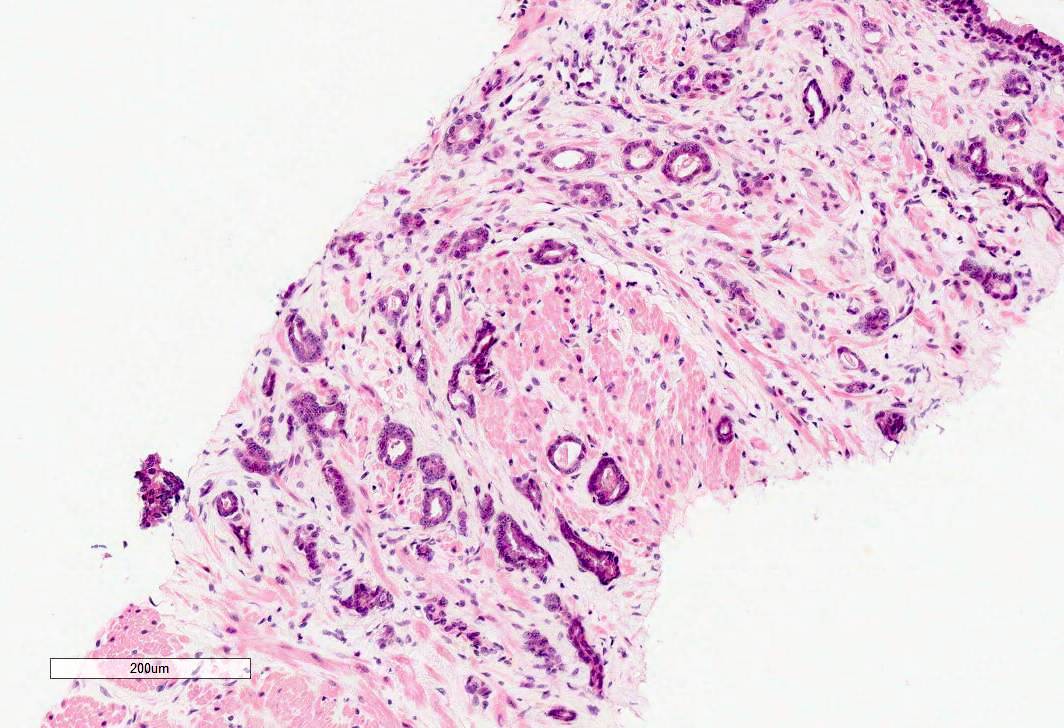
A 74 year old man with urinary hesitancy was found to have a PSA of 8 ng/mL. He had a transrectal ultrasound (TRUS) and prostate biopsy. A representative image is above. Which of the following is the typical immunoprofile of such a tumor?
- CK7- CK20- NKX3.1-
- CK7- CK20- NKX3.1+
- CK7- CK20+ NKX3.1-
- CK7+ CK20- NKX3.1-
- CK7+ CK20+ NKX3.1+
Board review style answer #1
Board review style question #2
Board review style answer #2