Table of Contents
Definition / general | Essential features | Terminology | ICD coding | Epidemiology | Sites | Pathophysiology | Autoantibodies | Ophthalmopathy | Clinical features | Diagnosis | Laboratory | Radiology description | Case reports | Treatment | Clinical images | Gross description | Gross images | Microscopic (histologic) description | Microscopic (histologic) images | Cytology description | Cytology images | Positive stains | Negative stains | Electron microscopy description | Videos | Differential diagnosis | Additional references | Practice question #1 | Practice answer #1Cite this page: Aly F, Satturwar S. Graves disease. PathologyOutlines.com website. https://www.pathologyoutlines.com/topic/thyroidgraves.html. Accessed September 14th, 2025.
Definition / general
- Named after Robert J. Graves (1796-1853)
- Commonly seen in middle aged women
- Autoimmune disease characterized by hyperthyroidism due to circulating autoantibodies against thyrotropin (TSH receptor) that activates the receptor, leading to increased thyroid hormone synthesis and secretion and growth of the thyroid gland
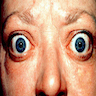
- Associated with diffuse goiter, infiltrative ophthalmopathy and less commonly infiltrative dermopathy, including pretibial myxedema and thyroid acropachy (extremity swelling, clubbing of fingers and toes due to periosteal new bone formation)
- Presence of thyrotropin receptor antibody in the serum and orbitopathy on clinical examination distinguishes Graves disease from other causes of hyperthyroidism
- Maternal Graves disease may lead to neonatal thyroidism in 1 - 5% of children due to transplacental transfer of antibodies
- People with other autoimmune diseases such as type 1 diabetes and rheumatoid arthritis are more likely to be affected
Essential features
- Most common cause of hyperthyroidism in United States
- Autoimmune disease characterized by hyperthyroidism due to circulating autoantibodies against thyrotropin (TSH receptor) that activates the receptor leading to increased thyroid hormone synthesis and secretion and growth of the thyroid gland
- Presence of thyrotropin receptor antibody in the serum and ophthalmopathy on clinical examination distinguishes Graves disease from other causes of hyperthyroidism
- Most commonly affects middle aged women, with female to male ratio of 4:1
- Associated with HLA class II molecule HLA-DR (HLA-DRB1*08 and DRB3*0202)
Terminology
- Also called diffuse toxic goiter, autoimmune hyperthyroidism, Basedow disease (in Europe)
ICD coding
Epidemiology
- Most common cause of hyperthyroidism in United States, affecting 2% of women and 0.3% of men
- 85% of patients are women (female to male ratio is 4:1), usually ages 20 - 40 years; men are usually older
- 60% concordance in identical twins; associated with HLA-B8 and HLA-DR3
Sites
- Global involvement of thyroid gland
Pathophysiology
- Exact cause is unclear
- It is believed to involve a combination of genetic and environmental factors
- A person is more likely to be affected if they have a family member with the disease
- Onset of disease may be triggered by stress, infection or giving birth
- Smoking increases the risk of disease and may worsen eye problems
- Caused by B and T cell mediated immune responses leading to production of autoantibodies to thyrotropin / TSH receptor
- These autoantibodies are IgG1 subclass, mimic the effects of TSH, cause thyroid hormone synthesis and secretion and cause thyroid growth resulting in a diffuse goiter
- Stimulatory antibodies increase the synthesis and activity of sodium iodide symporter leading to increased iodide uptake in Graves disease in the absence of TSH and stimulate protein C kinase pathway leading to cell proliferation
- Pituitary secretion of TSH is suppressed due to negative feedback of increased thyroid hormones
- A variety of immune mechanisms may be involved in the pathogenesis
- Major mechanisms are thyroid cell expression of human leukocyte antigen (HLA) associated molecules associated with bystander activation
Autoantibodies
- There are four thyroid antigens: thyroglobulin, thyroid peroxidase, sodium iodide symporter and thyrotropin / TSH receptor
- Anti-thyrotropin antibodies are specific for Graves disease
- Previously detected long acting thyroid stimulators have now been identified as the auto antibodies
- Antibodies can be either stimulatory / inhibitory or neutral, leading to various clinical presentations of hyperthyroidism or hypothyroidism
- Main autoantigen is the thyrotropin / thyroid stimulating hormone (TSH) receptor which is expressed primarily in the thyroid but also in adipocytes, fibroblasts, bone cells and a variety of additional sites
- Genes associated with autoimmune thyroid disease are: HLA, CD40, CTLA-4, thyroglobulin, TSH receptor and PTPN22
- Antibodies to thyroid peroxidase (microsomal antigen) and thyroglobulin are also seen
Ophthalmopathy
- Thyroid stimulating antibodies and activated T cell cytokines such as tumor necrosis factor (TNF) alpha and interferon gamma stimulate adipocytes to proliferate and orbital fibroblasts to secrete glycosaminoglycans
- Accumulation of hydrophilic glycosaminoglycan causes a change in osmotic pressure, which in turn leads to a fluid accumulation, muscle swelling and an increase in pressure within the orbit
- Together with retroorbital adipogenesis, the eyeball is displaced resulting in malfunction of the extraocular muscles as well as the venous drainage
Clinical features
- Features of hyperthyroidism: goiter / enlarged thyroid, muscle weakness, tremors, sweating, heat intolerance, oligomenorrhea, weight loss, exophthalmus (ophthalmopathy), tachycardia (atrial flutter or fibrillation), anxiety, congestive heart failure, pretibial nonpitting edema and dermopathy and acropachy (extremity swelling, clubbing of fingers and toes due to periosteal new bone formation)
- Progression of ophthalmopathy can lead to compromised vision and blindness
- Long standing thyrotoxicosis causes severe weight loss with osteoporosis and muscle protein breakdown
- Thyroid storm is associated with mortality rate of 20% even with treatment
Diagnosis
- Diagnosed clinically by symptoms, presence of laboratory markers of hyperthyroidism, ophthalmopathy and presence of serum anti thyrotropin antibodies
- Most patients have diffuse thyroid enlargement; large or cold nodules should prompt evaluation by fine needle aspiration cytology
Laboratory
- Increased T3 / T4, increased uptake of radioactive iodine, decreased TSH and positive thyroid receptor antibodies
Radiology description
-
Ultrasound
- Thyroid gland is often enlarged and can be hyperechoic
- Heterogeneous thyroid echotexture
- Relative absence of nodularity in uncomplicated cases
- Hypervascular; may demonstrate a thyroid inferno pattern on color Doppler
- Iodine-123: imaging performed at around 2 - 6 days; classically demonstrates homogeneously increased activity in an enlarged gland
- Tc-99m pertechnetate: homogeneously increased activity in an enlarged thyroid gland
Case reports
- 46 year old woman with case occurring after excision of multinodular goiter (Exp Clin Endocrinol Diabetes 2009;117:95)
- Woman with peripartum cardiomyopathy (Int J Cardiol 2010;145:e23)
Treatment
- Beta blockers, propylthiouracil or other drugs, radioiodine ablation, rituximab, surgery (subtotal thyroidectomy, Endocr J 2008;55:161, World J Surg 2008;32:1269)
- Surgery may improve exophthalmos (Chirurgia (Bucur) 2008;103:291)
- Myxedema may persist in spite of treatment
Clinical images
Gross description
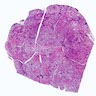
- Diffuse and symmetrically enlarged thyroid gland with beefy red cut surface, weight 50 - 150 grams
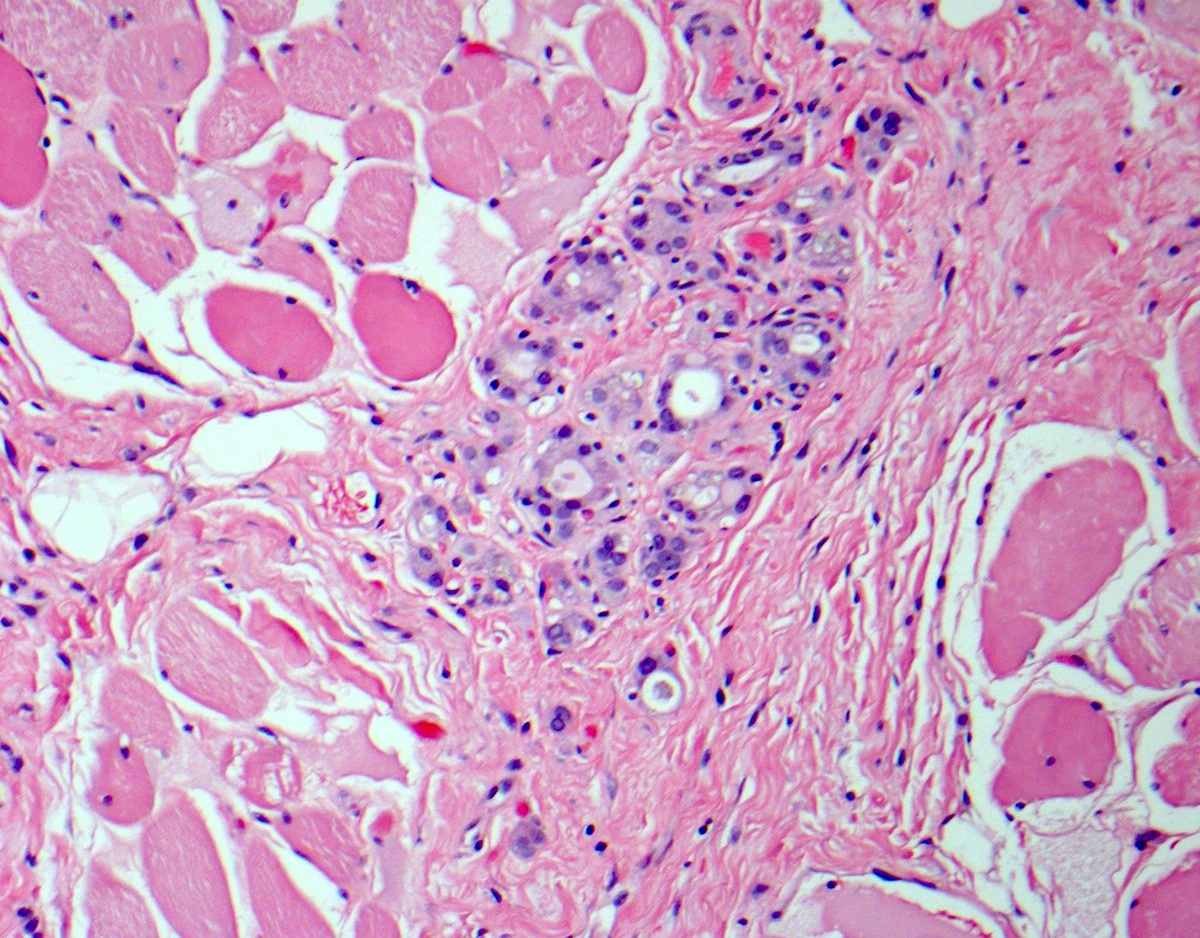
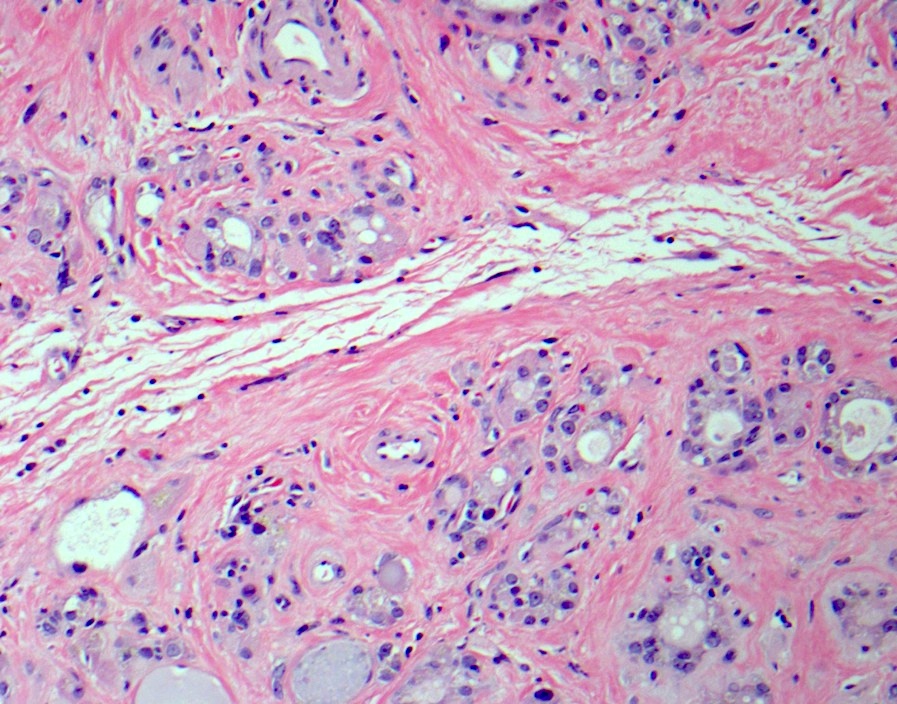
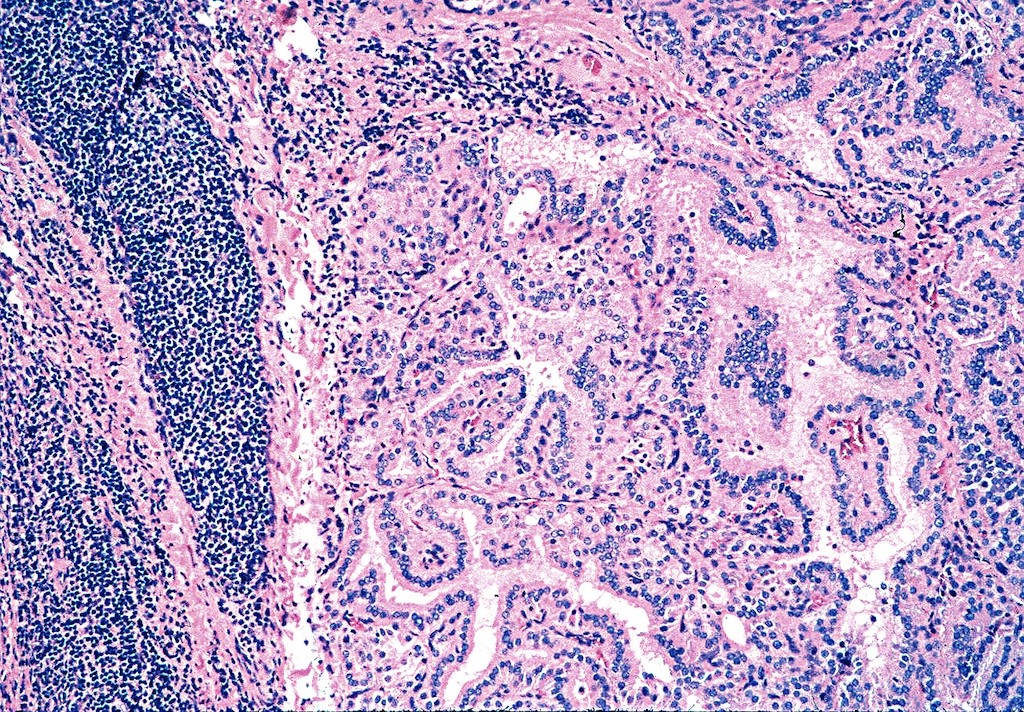
Microscopic (histologic) description
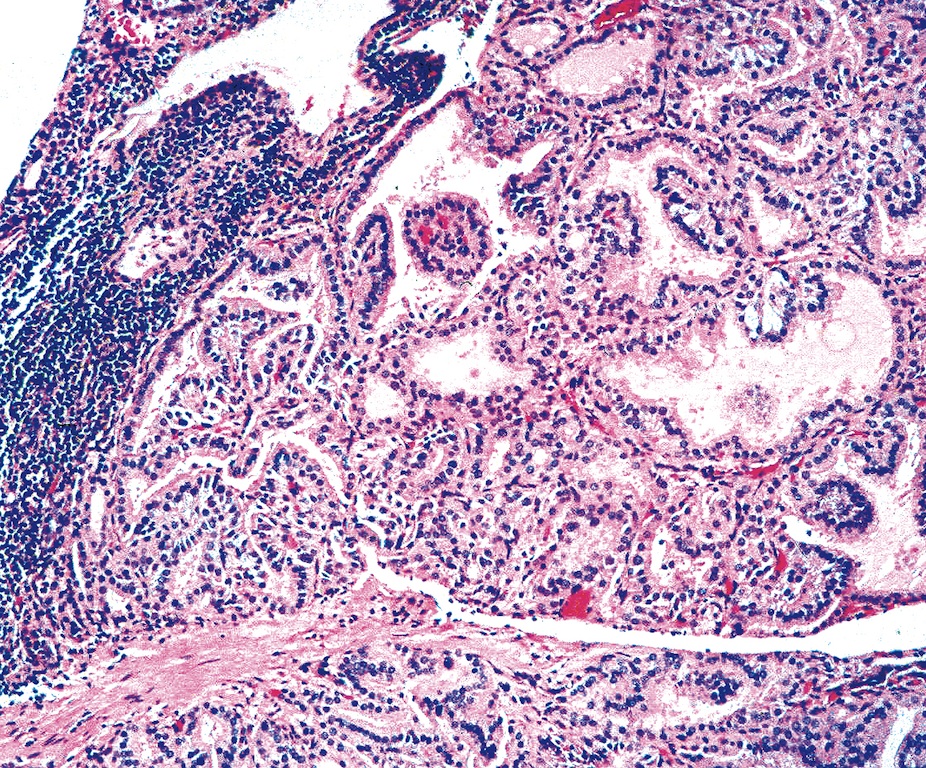
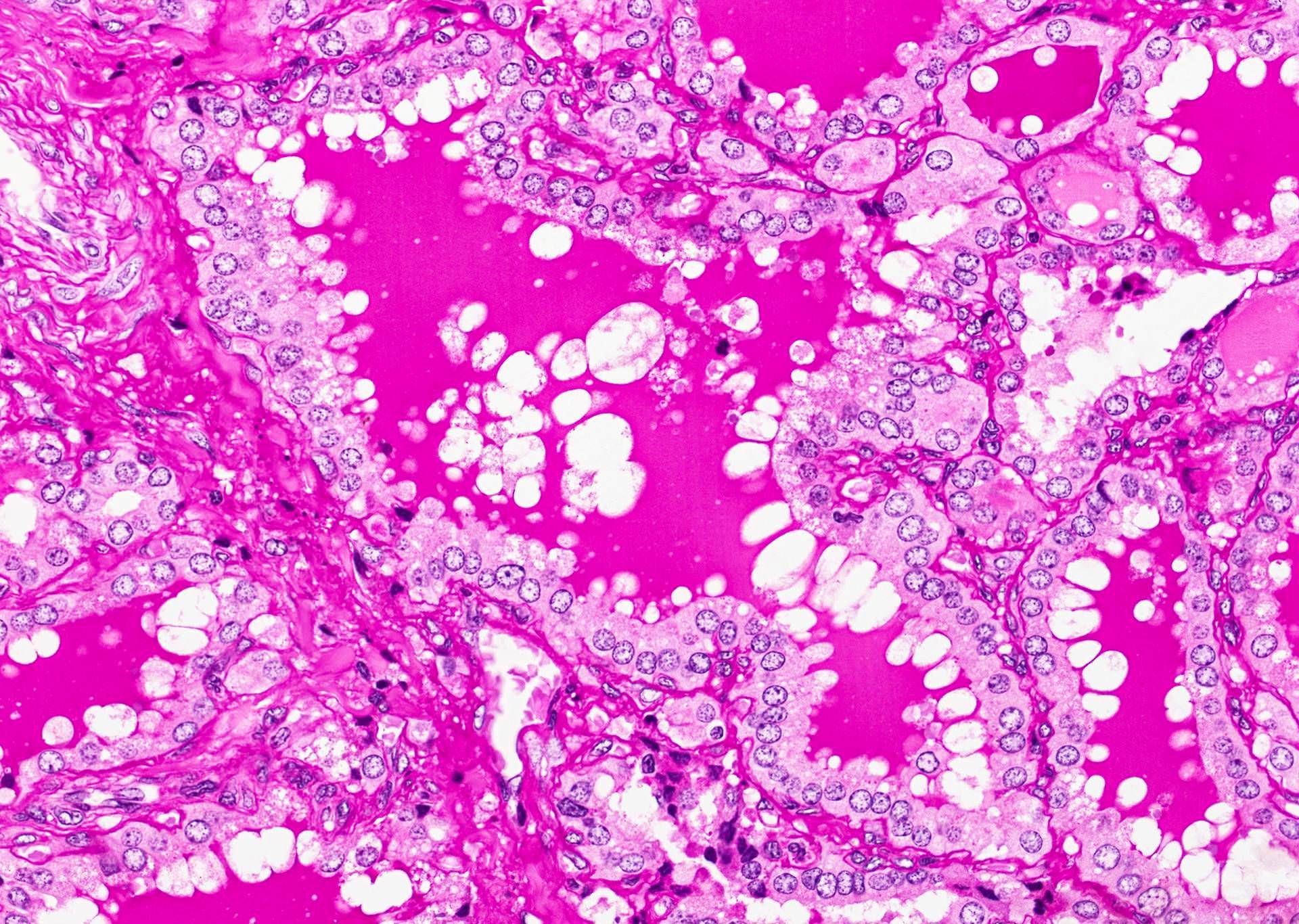
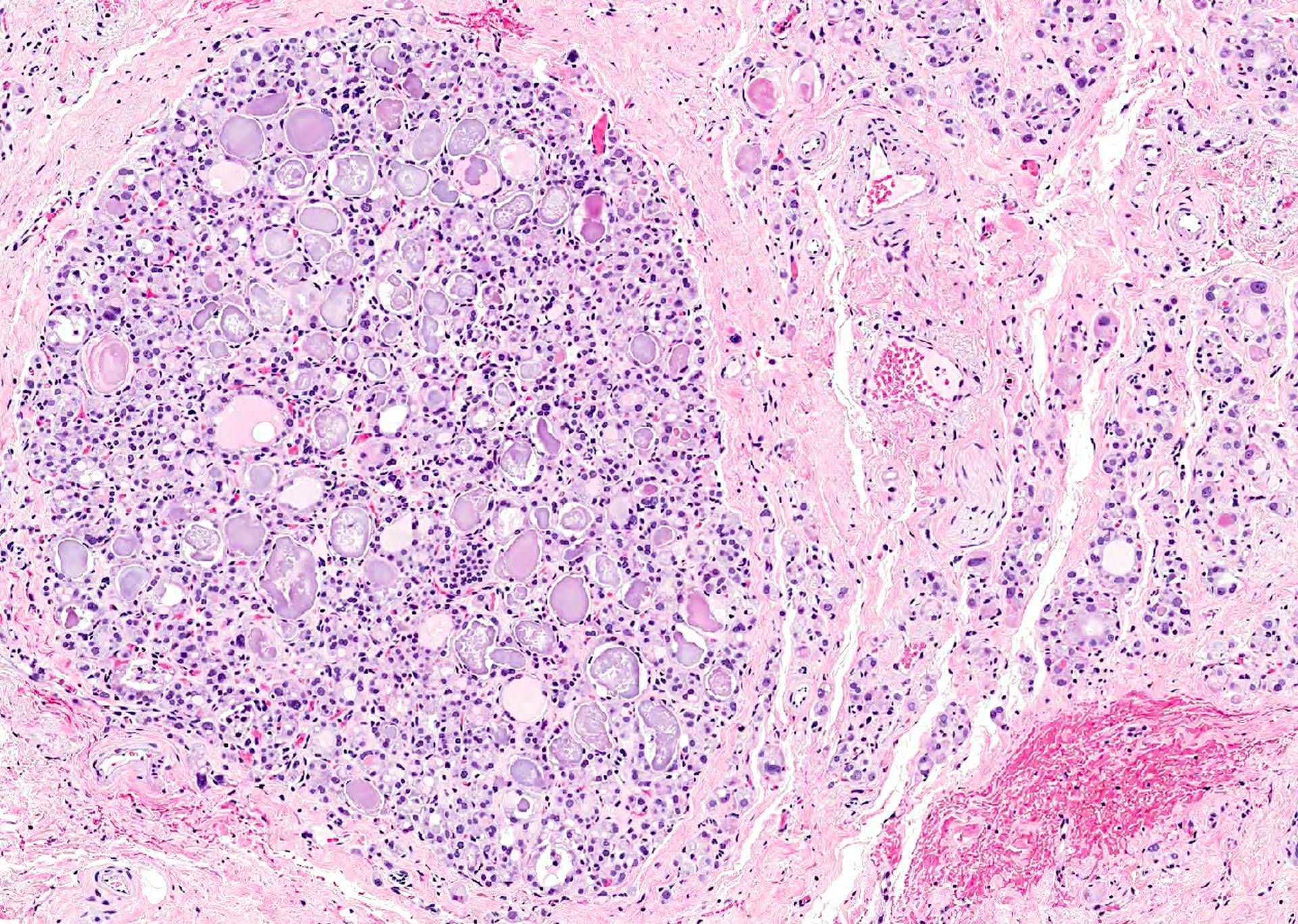
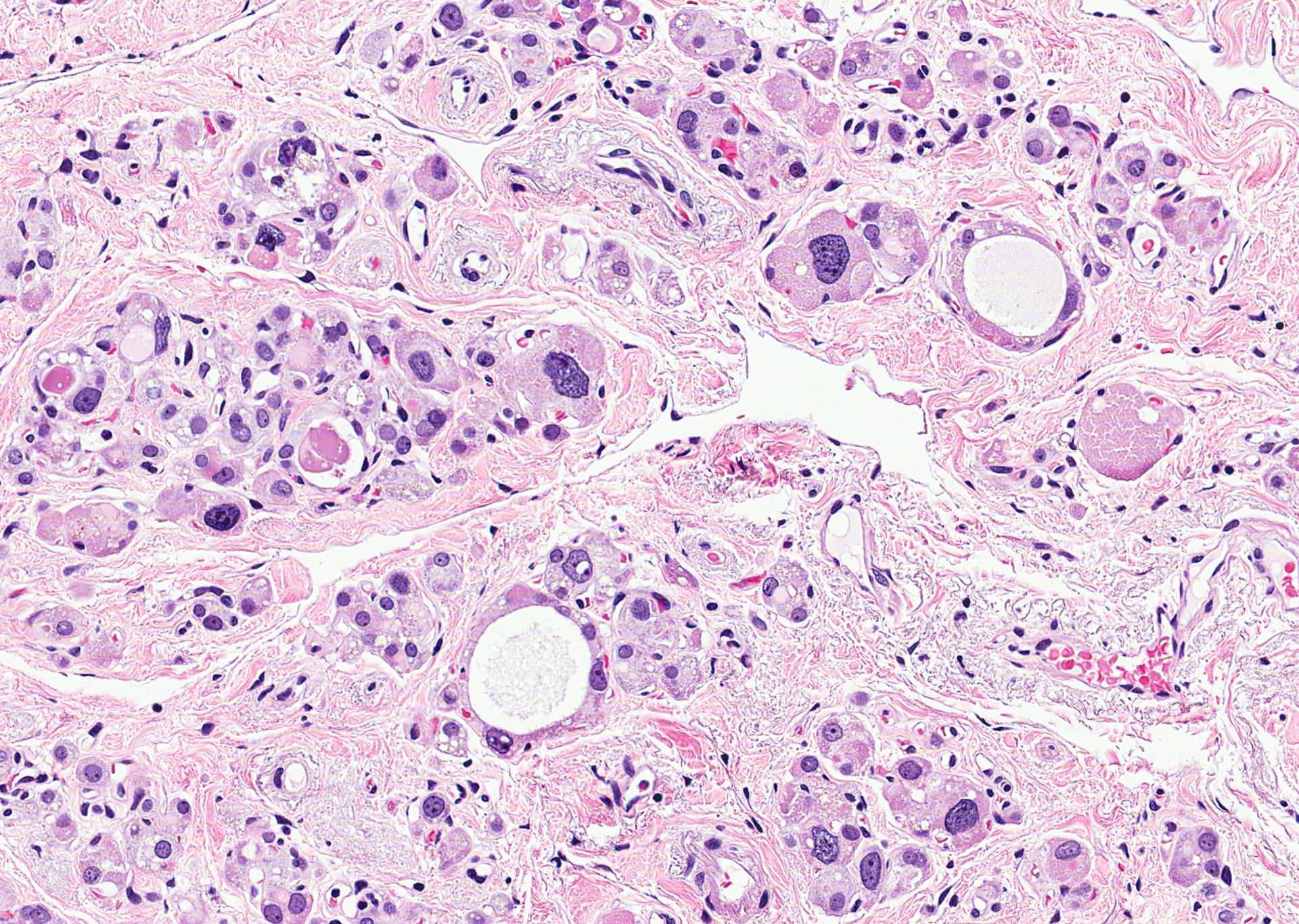
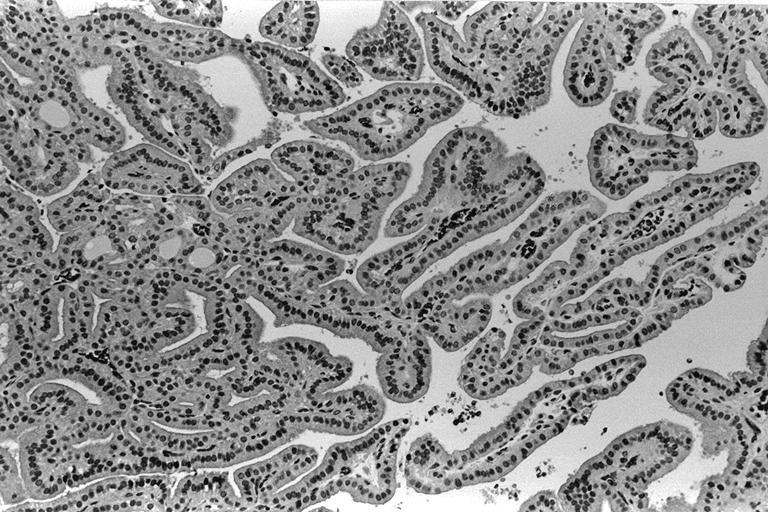
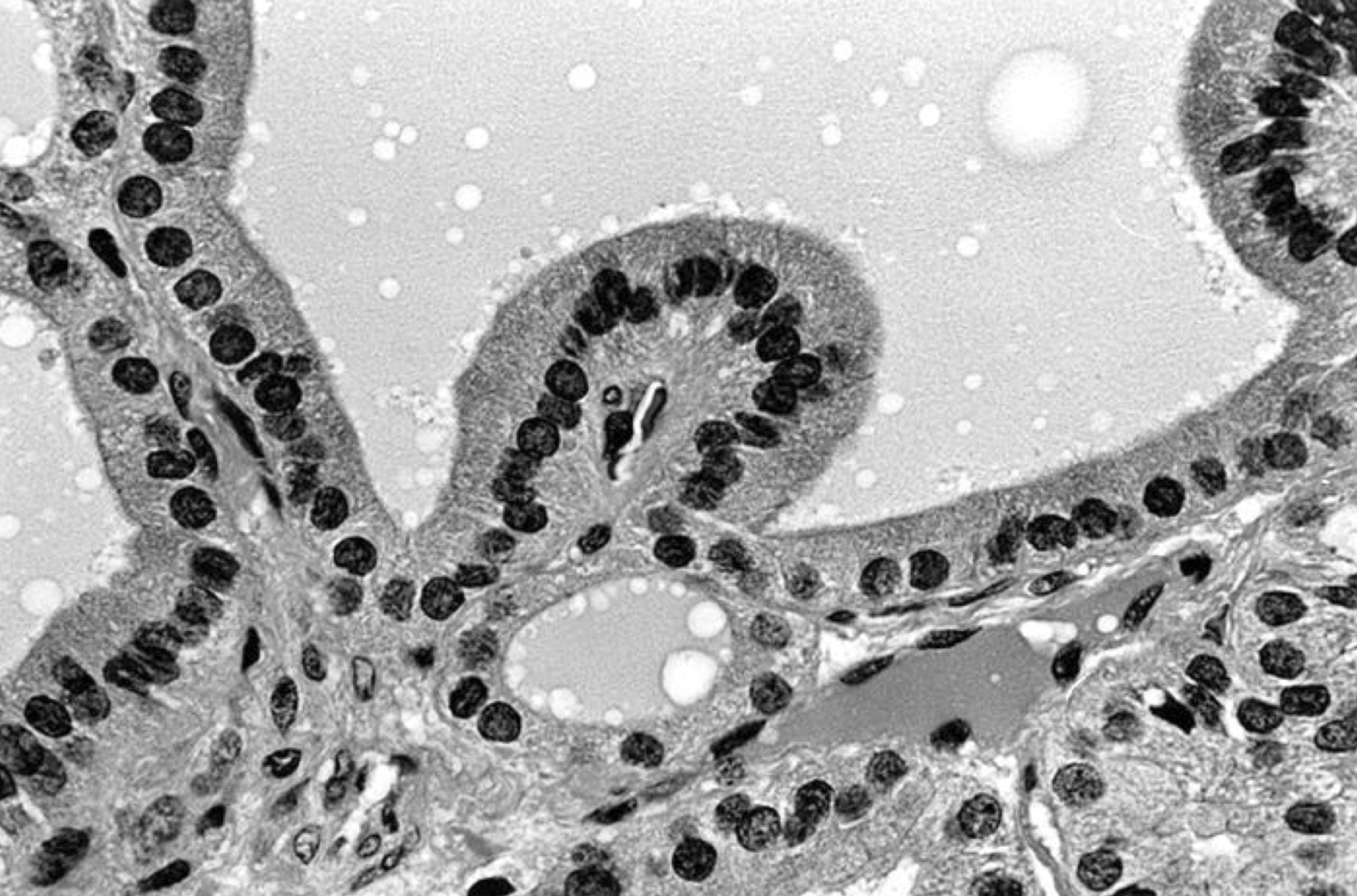
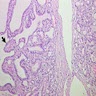
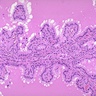
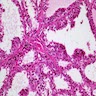
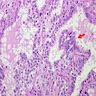
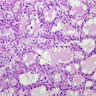
- Hyperplastic thyroid follicles with papillary infoldings
- Diffuse hyperplasia and hypertrophy of follicular cells with retention of lobular architecture and prominent vascular congestion
- Tall follicular cells with papillae usually lacking fibrovascular cores
- Nuclei are round, often basally located, rarely overlap
- Colloid is typically decreased, when present shows peripheral scalloping
- Colloid may increase after treatment
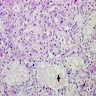
- Variable patchy lymphoid infiltrate in the stroma
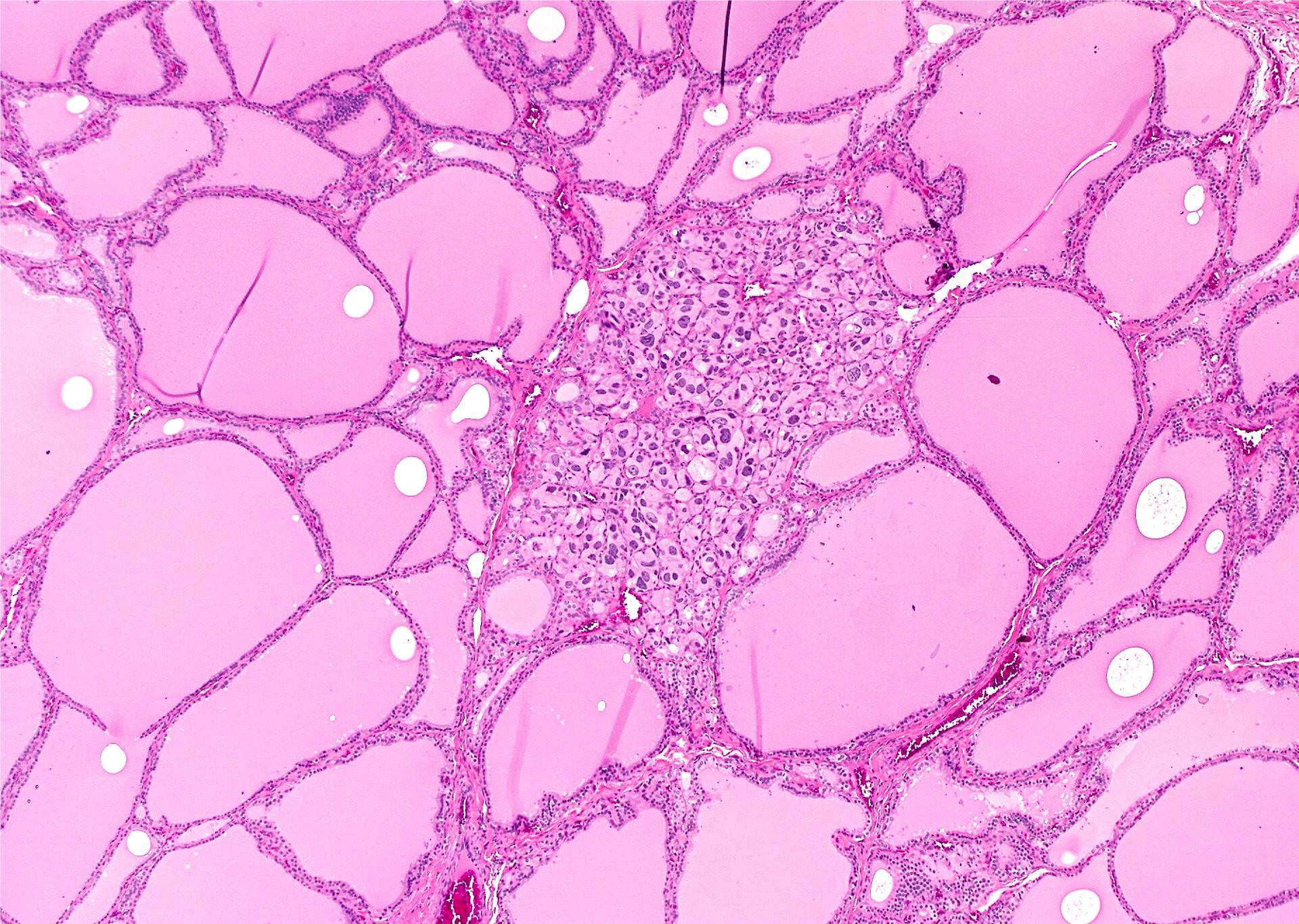
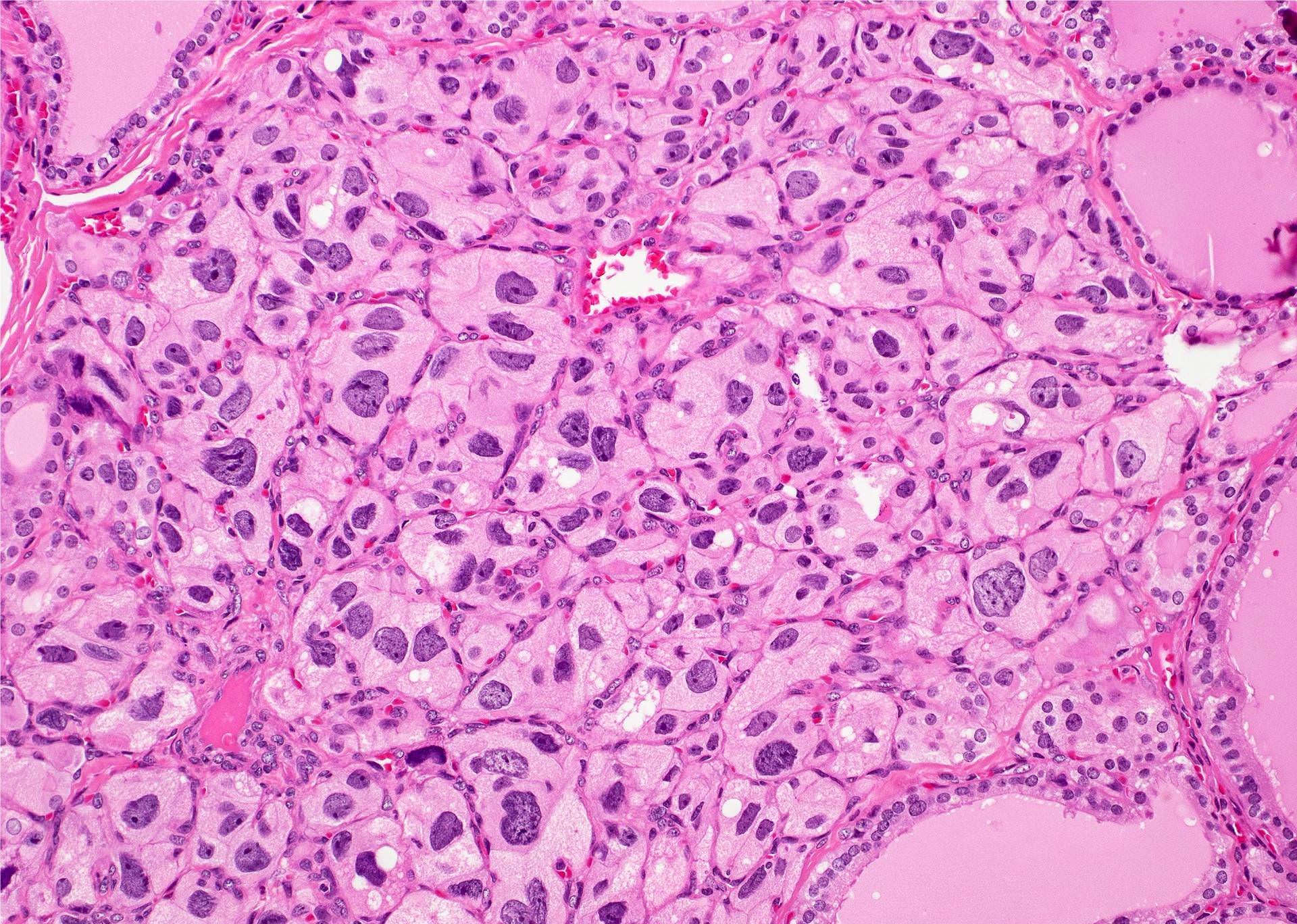
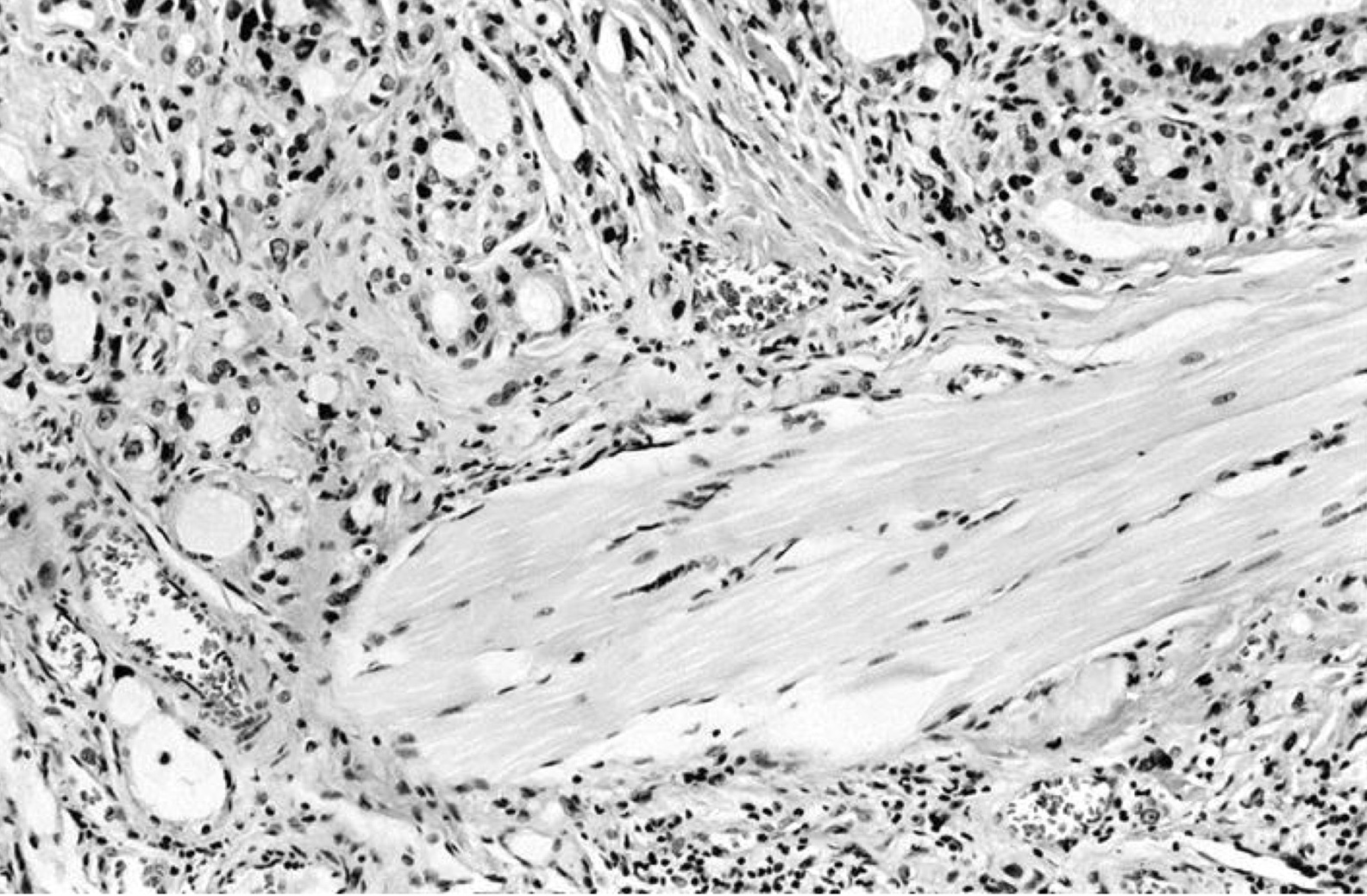
- Nuclear clearing (15%), florid papillary hyperplasia (13%, may resemble papillary thyroid carcinoma), nuclear grooves or pseudonuclear inclusions (8%), nuclear enlargement, multinucleation, pleomorphism or prominent nucleoli (7%), mitotic figures (6%), psammoma bodies (1%), hyperplastic follicles may extend into adjacent skeletal muscle (1%)
- Rarely small clusters of normal thyroid follicles in adjacent lymph node sinuses (Hum Pathol 2008;39:1080)
- Note: preoperative potassium iodide to suppress vascularity causes epithelial involution and colloid accumulation
- Gland may look normal after 3 weeks of treatment
- Preoperative PTU exaggerates the hyperplasia and hypertrophy
- Radioactive iodine initially causes dissolution of some follicles, vascular changes, nuclear atypia and stromal fibrosis
- Late changes are follicular atrophy, fibrosis, nodularity and oncocytic changes
- Periorbital tissue: lymphoplasmacytic infiltrate present in periorbital soft tissue and extraorbital skeletal muscles
- Skin hyperkeratosis: deposition of acid mucopolysaccharides in dermis
Microscopic (histologic) images
Scroll to see all images:
Contributed by Swati Satturwar, M.D., Mark R. Wick, M.D. and Andrey Bychkov, M.D., Ph.D.
AFIP images
Images hosted on other servers:
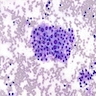
Cytology description
- Cytology features are nonspecific and similar to benign follicular lesions such as nodular goiter, adenomatoid nodules or colloid nodules
- Cellular smears with follicular cells in flat sheets and loosely cohesive clusters
- Cells are tall with finely granular cytoplasm, marginal vacuoles and basal nuclei
- Nuclei are enlarged, vesicular and show prominent nucleoli
- Background may show lymphocytes and oncocytes
- Flame cells represented by marginal cytoplasmic vacuoles with pink red frayed edges may be prominent, however not specific for Graves disease
- After radioactive therapy: prominent microfollicular architecture with significant nuclear atypia, overlapping and crowding
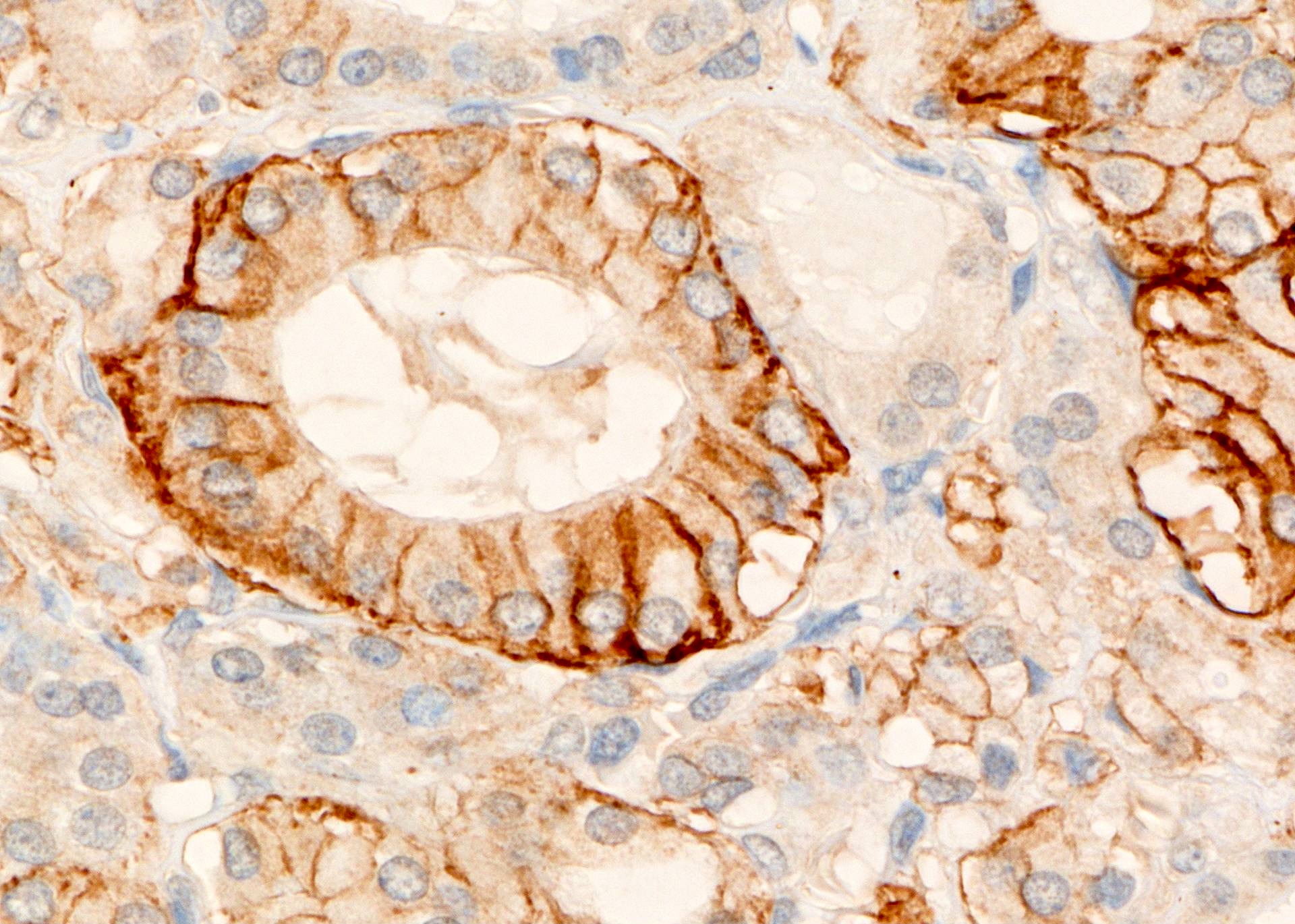
Positive stains
- p27 (Mod Pathol 2000;13:1014) may distinguish it from papillary thyroid carcinoma
- HLA-DR in cytoplasm of thyrocytes and lymphocytes
Negative stains
Electron microscopy description
- Prominent rough endoplasmic reticulum and golgi, well developed nucleoli in enlarged nuclei
Videos
Graves gross and micro
Thyroid: compare and contrast
Histopathology thyroid: Graves disease
Differential diagnosis
- DD of thyrotoxicosis (other than Graves disease):
- Amiodarone induced
- Hashitoxicosis
- Struma ovarii
- Toxic follicular adenoma
- Toxic sporadic goiter
- Trophoblastic tumor
- Papillary thyroid carcinoma: enlarged overlapping nuclei, nuclear grooves, nuclear inclusions
Additional references
Practice question #1
All of the following statements are true about Graves disease except:
- Graves disease is caused by anti-TPO autoantibodies
- Associated with HLA-DR
- Caused due to anti-thyrotropin antibodies
- Most commonly affects middle aged women
Practice answer #1
A. Graves disease may be associated with anti-TPO anti-bodies but it is caused by anti-thyrotropin / TSH receptor auto-antibody that mimic TSH and stimulate hormone synthesis, secretion and thyroid growth. Most commonly affects middle aged women with female to male ratio of 4:1. It is associated with HLA class II molecule HLA-DR (HLA-DRB1*08 and DRB3*0202).
Comment Here
Reference: Graves disease
Comment Here
Reference: Graves disease