Table of Contents
Definition / general | Essential features | Terminology | ICD coding | Epidemiology | Sites | Pathophysiology | Etiology | Diagrams / tables | Clinical features | Diagnosis | Laboratory | Prognostic factors | Case reports | Treatment | Gross description | Gross images | Microscopic (histologic) description | Microscopic (histologic) images | Sample pathology report | Differential diagnosis | Practice question #1 | Practice answer #1 | Practice question #2 | Practice answer #2Cite this page: Paulsen JD, Polydorides AD. Ulcerative proctitis. PathologyOutlines.com website. https://www.pathologyoutlines.com/topic/colonulcerativeproctitis.html. Accessed October 3rd, 2025.
Definition / general
- Subgroup of idiopathic ulcerative colitis wherein the extent of disease is anatomically limited to the rectum (i.e., distal to the rectosigmoid junction)
- Corresponds to E1 in the Montreal classification of ulcerative colitis by extent of disease (Can J Gastroenterol 2005;19:5A)
- Comprises approximately 30% of patients with ulcerative colitis (Clin Gastroenterol Hepatol 2018;16:343)
Essential features
- Diagnosis is based on constellation of symptoms, endoscopic findings, histopathology and exclusion of alternative diagnoses (Lancet 2017;389:1756)
- Symptoms: rectal bleeding, tenesmus, passage of mucus, urgency
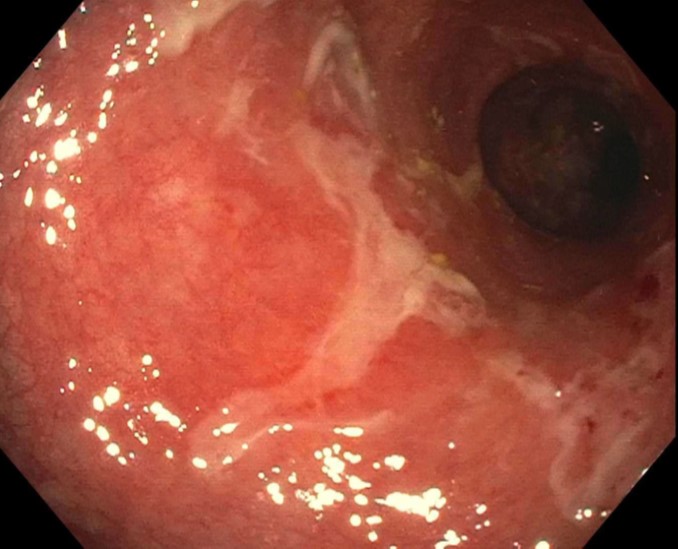
- Endoscopic findings: erythema, granularity, friability, erosions, ulcerations
- Histopathology:
- Active mucosal inflammation (cryptitis, crypt abscess, erosion, ulceration)
- Chronic mucosal inflammation (mucosal expansion, basal plasmacytosis)
- Evidence of chronic injury (crypt architecture distortion, Paneth cell metaplasia)
- Exclusion of alternative diagnoses:
- Infectious proctitis, including sexually transmitted (e.g., syphilis proctitis)
- Ischemic proctitis
- Therapy related proctitis, including drug induced (e.g., immune checkpoint inhibitor associated proctitis or NSAID injury) and radiation proctitis
- Segmental colitis associated with diverticulosis
- Fecal impaction (e.g., stercoral ulcer)
- Prolapse related (e.g., solitary rectal ulcer syndrome)
Terminology
- Idiopathic ulcerative proctitis
ICD coding
- ICD-10:
Epidemiology
- Similar to ulcerative colitis:
- No sex predilection (M = F)
- Incidence and prevalence is increasing worldwide (Gastroenterology 2012;142:46)
- Highest prevalence in Europe, Canada and United States
Sites
- Rectum
Pathophysiology
- Same as ulcerative colitis (results from abnormalities in immune regulation, epithelial barrier functions and host - microbiome interactions)
Etiology
- Same as ulcerative colitis
Clinical features
- Symptoms:
- Rectal bleeding, tenesmus, passage of mucus, urgency
- Up to 10% show paradoxical constipation (Lancet 2017;389:1756)
- Ulcerative proctitis less likely to show constitutional symptoms (such as fatigue and fever) when compared to extensive ulcerative colitis
- Endoscopic findings (limited to rectum):
- Erythema, granularity, friability, erosions, ulcerations
Diagnosis
- Diagnosis is based on constellation of symptoms, endoscopic findings, histopathology and exclusion of alternative diagnoses
Laboratory
- Same as ulcerative colitis
Prognostic factors
- Factors associated with poor outcomes (i.e., complicated or aggressive disease requiring immunosuppressants, biologics [e.g., antibody based drugs]) in adults include (Clin Gastroenterol Hepatol 2015;13:635):
- Young age of onset (< 40 years)
- Nonsmokers and ex smokers
- Deep ulcerations
- Extent of disease (ulcerative proctitis is more associated with lower risk of colectomy than left sided and extensive colitis):
- Disease can extend proximally in up to 19% of patients after 5 years and up to 28% of patients after 10 years (Inflamm Bowel Dis 2012;18:573)
- Progression to left sided colitis in 28 - 30% of patients and to extensive colitis in 14 - 16% (Clin Gastroenterol Hepatol 2018;16:343)
- Colitis related dysplasia and carcinoma:
- When disease is limited to the rectum (i.e., ulcerative proctitis), patients have not been shown to be at increased risk for colorectal neoplasia (N Engl J Med 1990;323:1228, Gastroenterology 2006;130:1039)
Case reports
- 46 year old man with bloody diarrhea and mucus discharge (Acta Gastroenterol Belg 2021;84:509)
- 63 year old man with new onset abdominal pain and diarrhea (ACG Case Rep J 2020;7:e00364)
- 65 year old man with bloody diarrhea and tenesmus (Rev Esp Enferm Dig 2019;111:889)
Treatment
- Same as ulcerative colitis
- Because disease in ulcerative proctitis is limited to within 15 - 20 cm of the anal verge, medical therapies (such as mesalamine) may be administered topically (i.e., by suppository or enema)
- Reference: Am J Gastroenterol 2019;114:384
Gross description
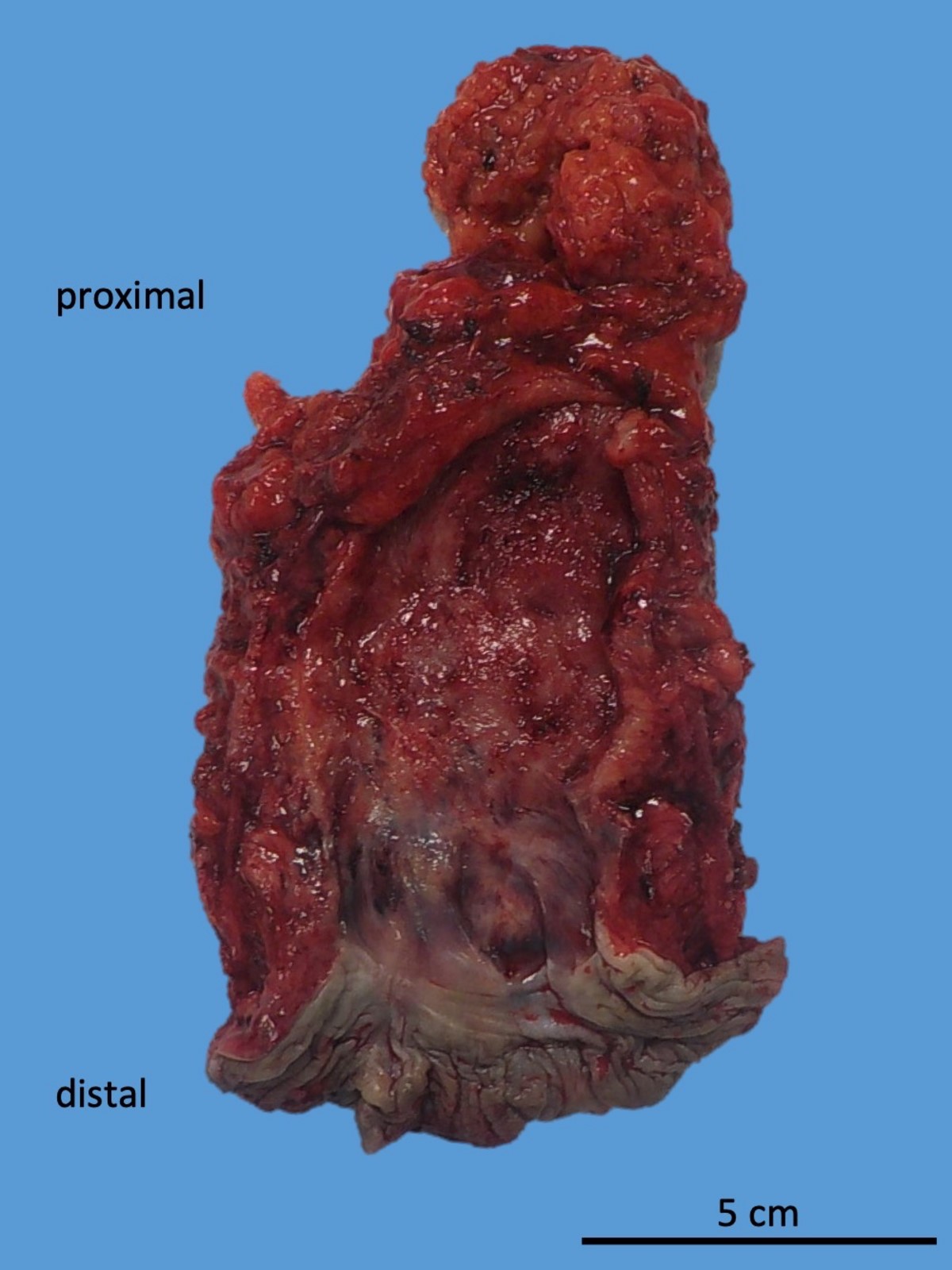
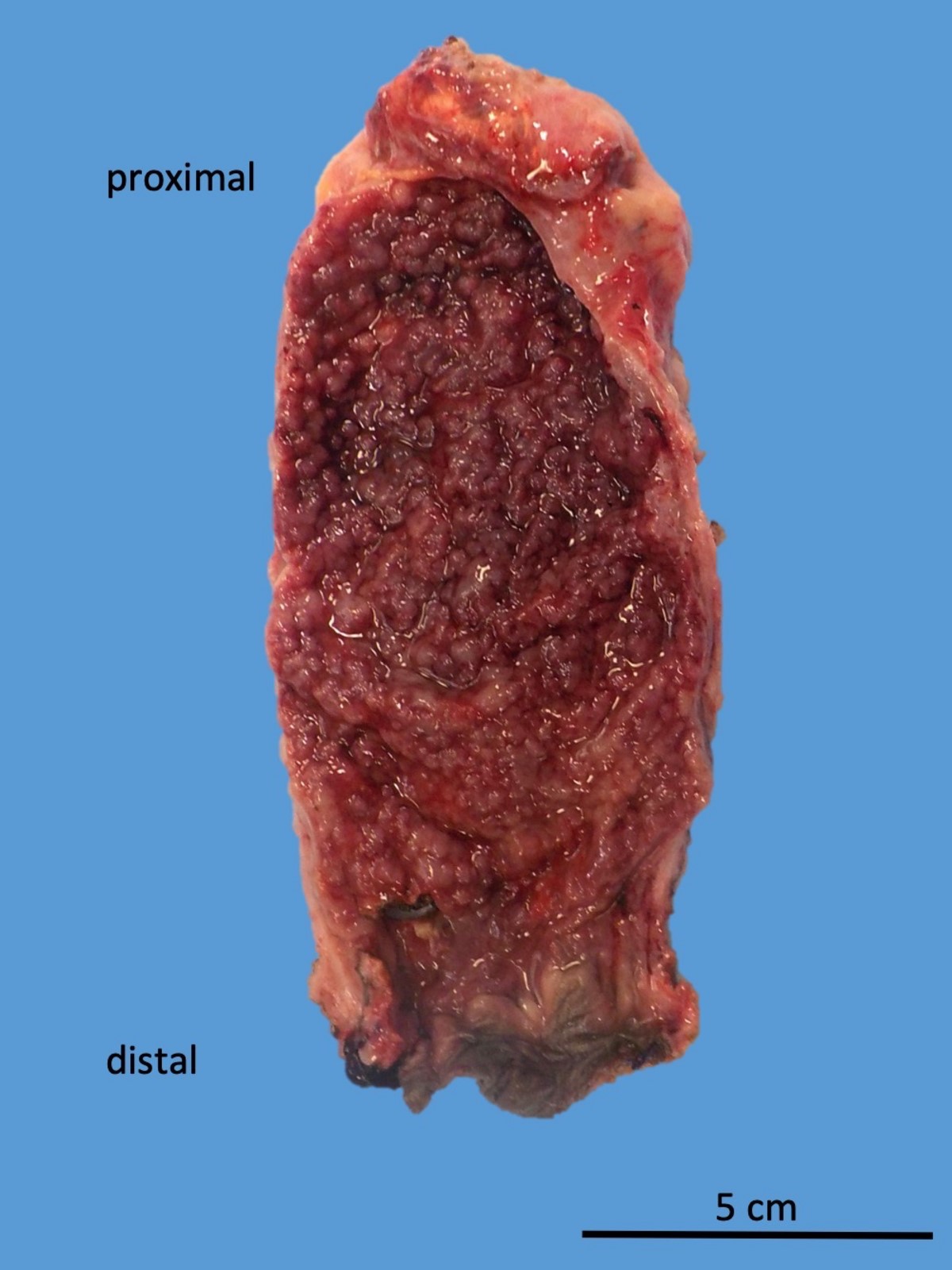
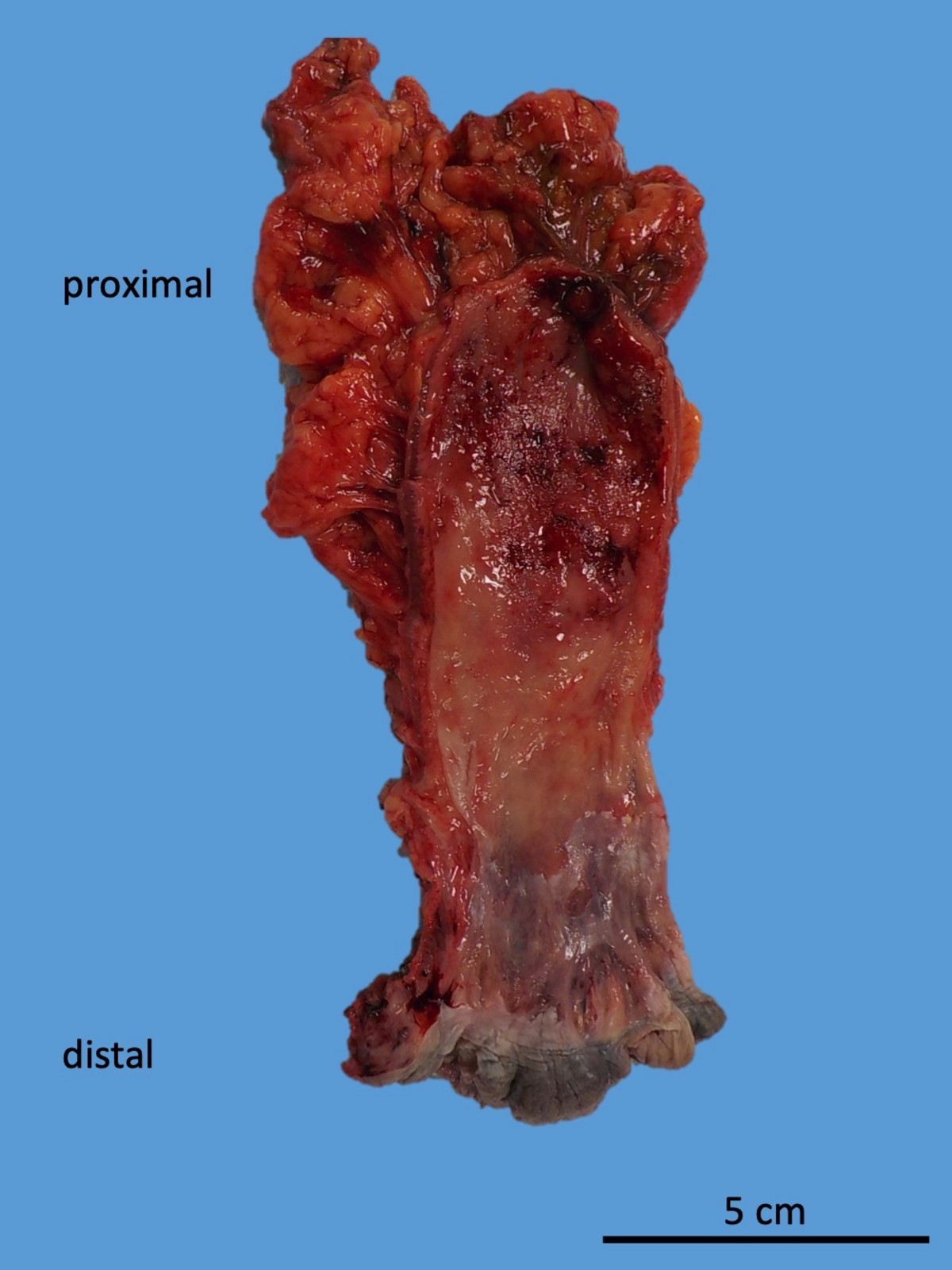
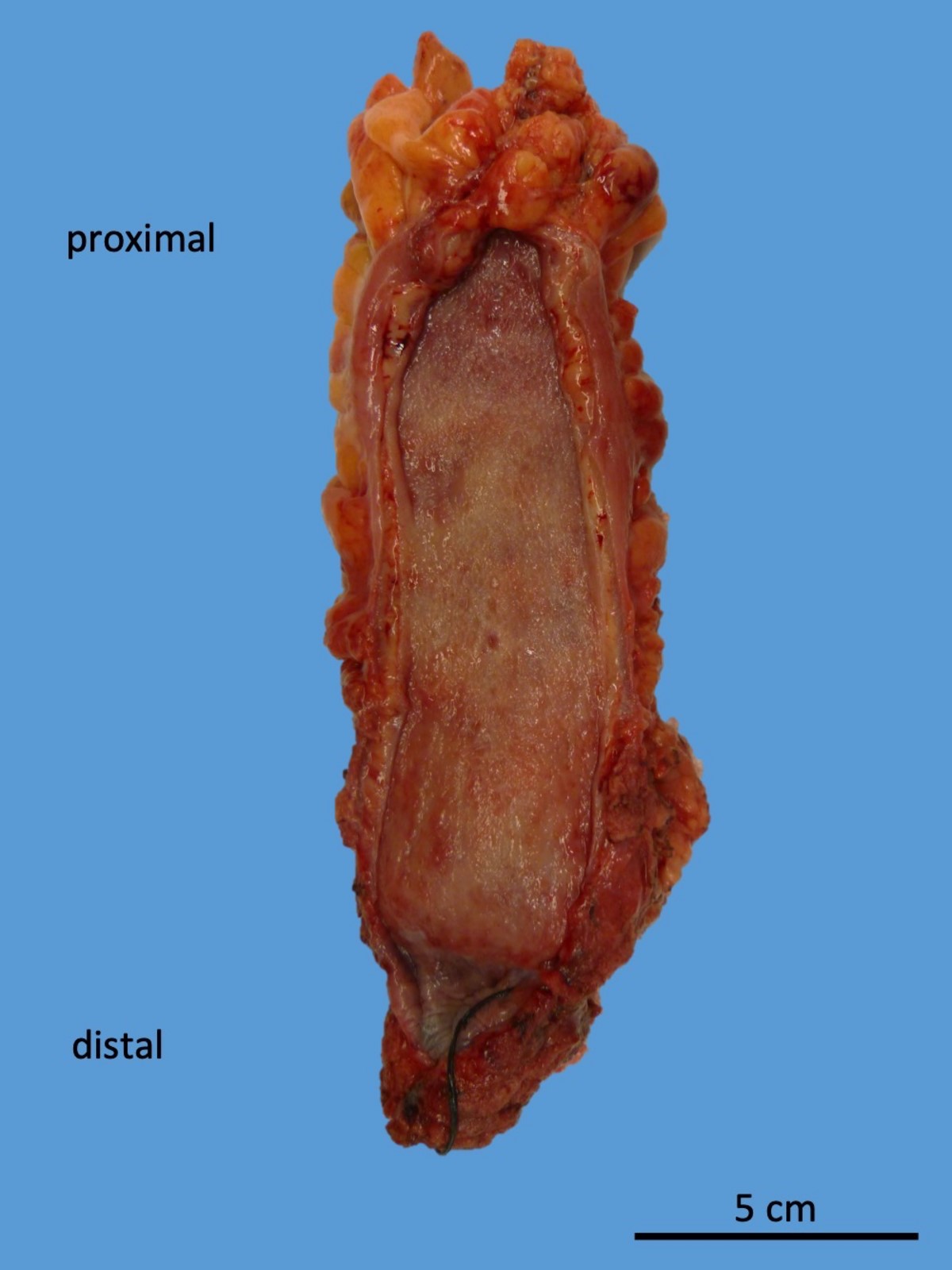
- Diffuse, continuous involvement of the rectal mucosa by:
- Congestion, granularity and edema
- Shallow, broad based ulcerations
- Inflammatory pseudopolyps (islands of regenerating mucosa amid shallow ulcers)
- Mucosal bridges (fused inflammatory pseudopolyps)
- Mucosal disease may be patchy after medical treatment (J Clin Pathol 2004;57:1233)
- Longstanding chronic disease may show flattened, featureless and atrophic mucosa (burned out proctitis)
Gross images
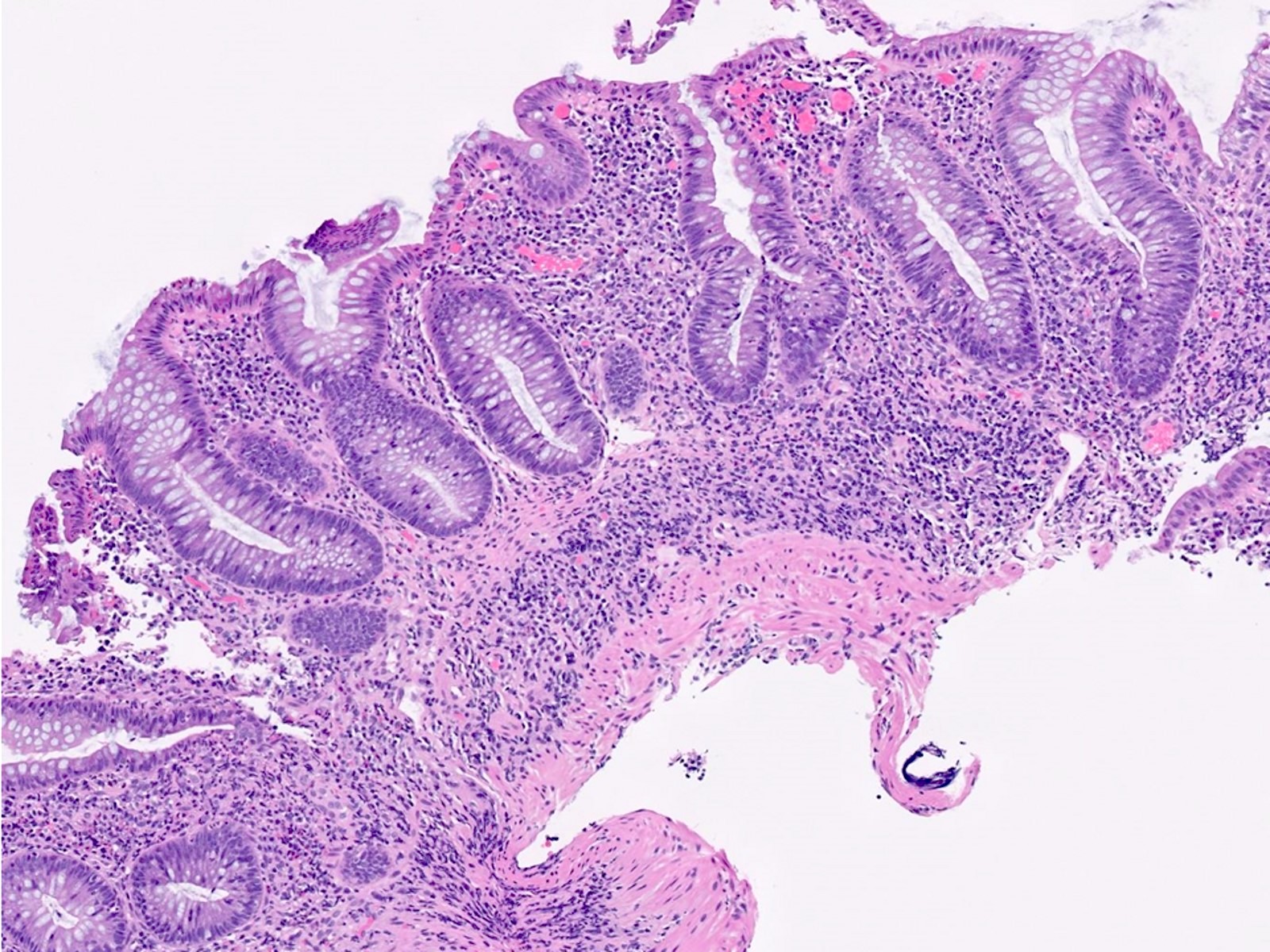
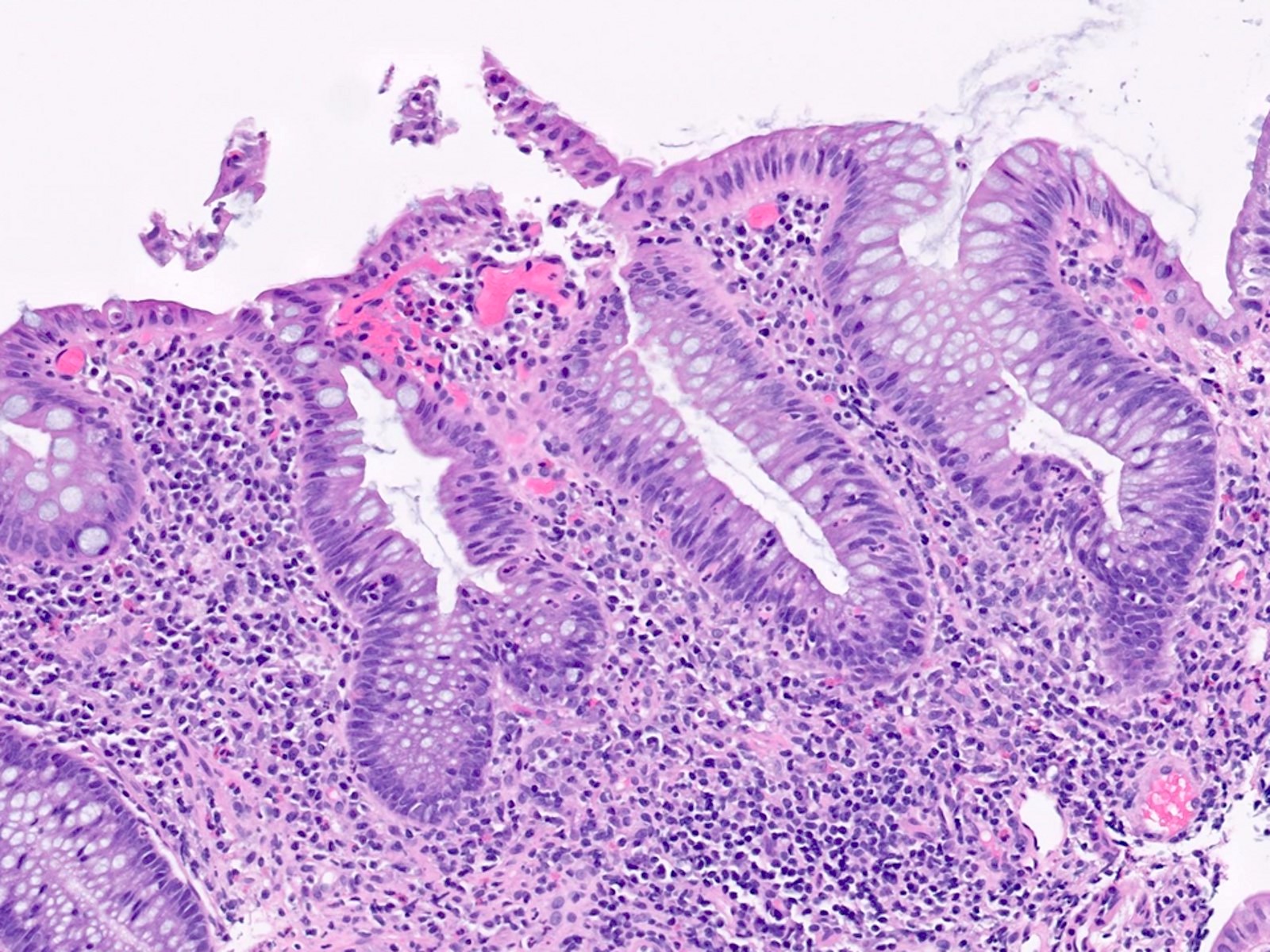
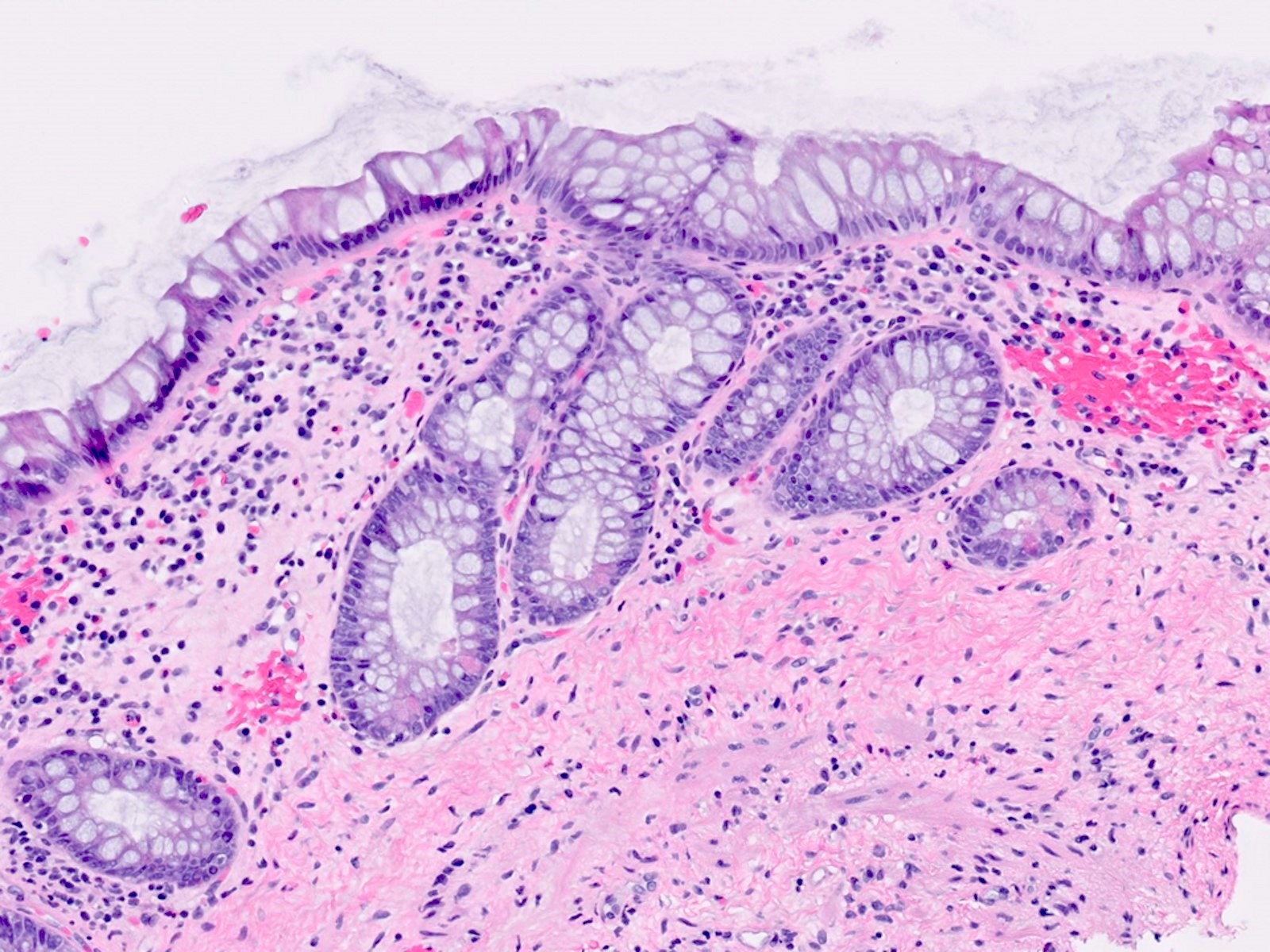
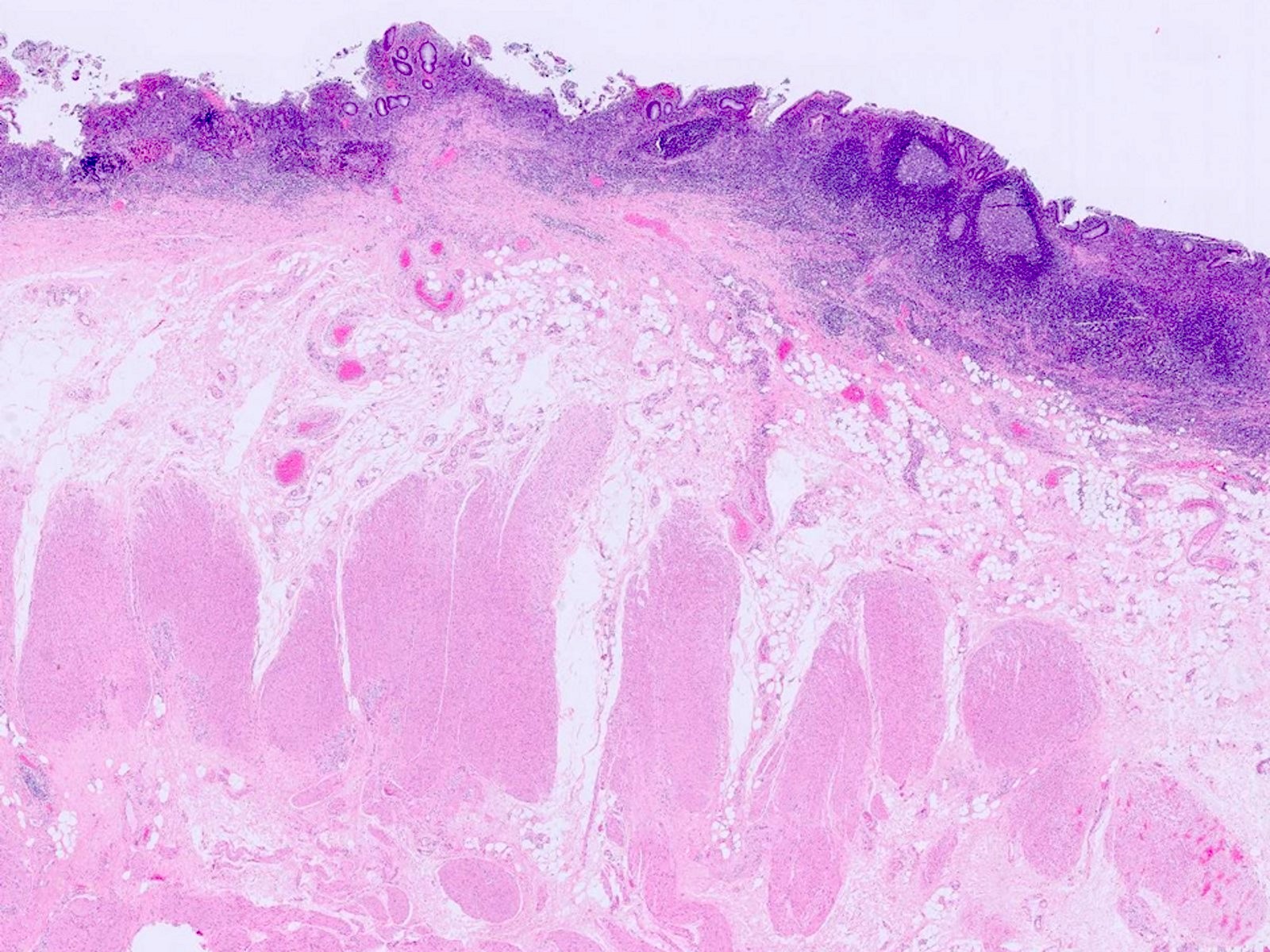
Microscopic (histologic) description
- Same as ulcerative colitis; however, only rectum is involved
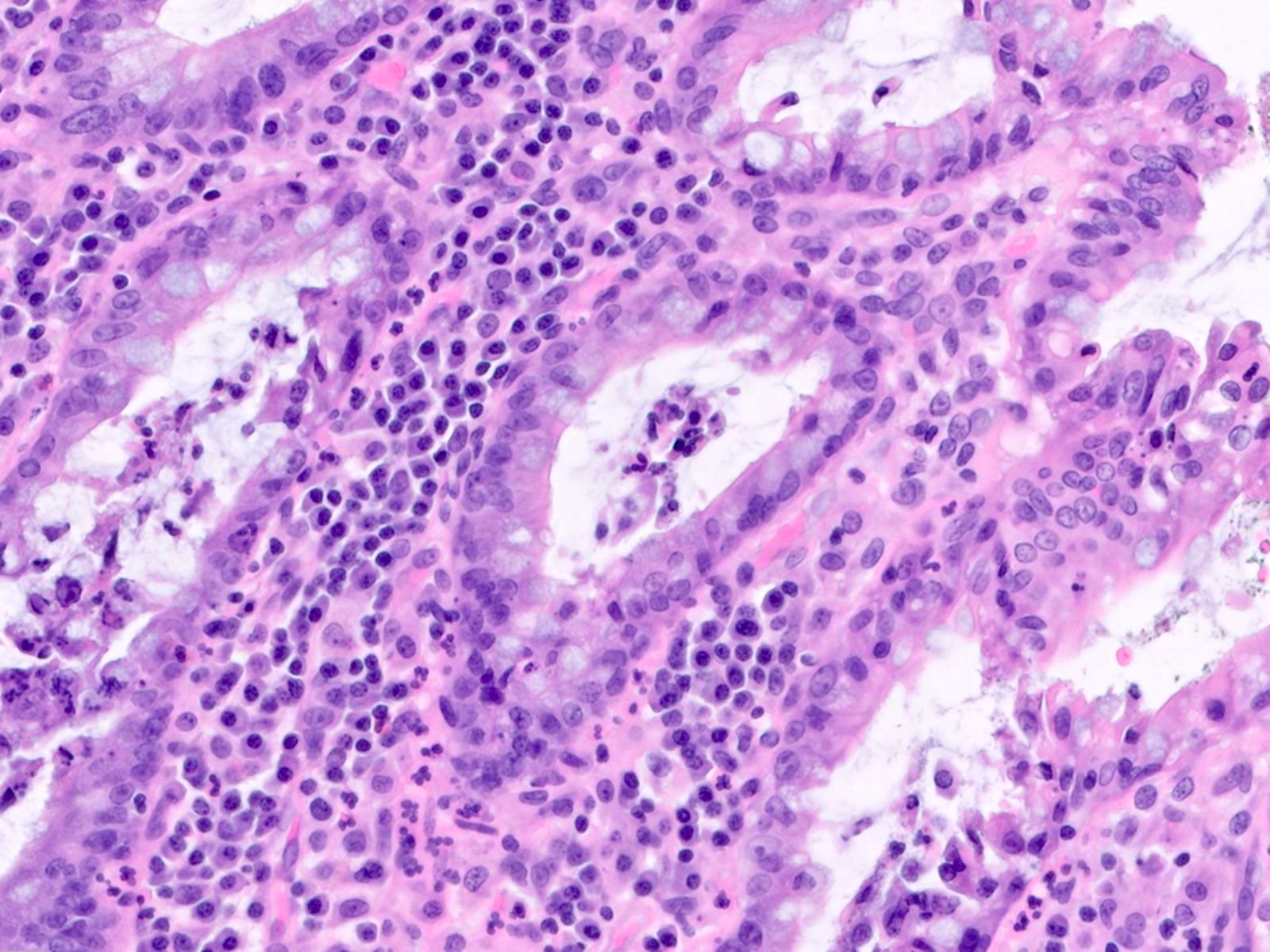
- Active mucosal inflammation (J Clin Pathol 1995;48:749):
- Cryptitis (neutrophilic infiltration of crypt epithelium)
- Crypt abscess (crypt with intraluminal aggregates of neutrophils)
- Erosions
- Ulceration
- Chronic mucosal inflammation (J Clin Pathol 1995;48:749):
- Mucosal expansion: increased lymphoplasmacytic inflammation in the lamina propria with loss of gradient (i.e., lack of increased infiltrates near lumen)
- Basal plasmacytosis (so called crypt shortfall)
- Evidence of chronic injury:
- Paneth cell metaplasia (Paneth cells are normally absent distal to the mid transverse colon)
- Crypt architectural distortion (branched, dilated and disorganized crypts)
- Cryptolytic granulomas (granulomas in association with ruptured crypts) may be seen (Histopathology 2002;41:50)
- Inactive (quiescent) proctitis in treated ulcerative proctitis may show subtle signs of chronicity (mild architecture distortion, focal Paneth cell metaplasia) or appear with near normal histology
- Dysplasia / carcinoma
- Active mucosal inflammation (J Clin Pathol 1995;48:749):
Microscopic (histologic) images
Sample pathology report
- Rectum, endoscopic biopsy:
- Moderately active chronic proctitis; negative for dysplasia (see comment)
- Comment: The proctitis is characterized by cryptitis and crypt abscesses, mucosal expansion by full thickness lymphoplasmacytosis, which is most prominent basally, crypt distortion, including cystic dilation and crypt shortfall and evidence of chronic mucosal injury, including atrophy and Paneth cell metaplasia. In the appropriate clinical setting, these findings are suggestive of idiopathic inflammatory bowel disease.
- Rectum, completion proctectomy:
- Segment of rectum with mildly active chronic idiopathic ulcerative proctitis; negative for dysplasia (see comment)
- Reactive lymph nodes
- Comment: There is also prominent follicular lymphoid hyperplasia, possibly representing a (superimposed) component of diversion proctitis.
Differential diagnosis
- Crohn's disease:
- Nonnecrotizing epithelioid cell granulomas (not associated with ruptured crypts)
- Transmural lymphoid aggregates in areas of intact (nonulcerated) mucosa
- Fistulas or strictures
- Perianal disease
- Sexually transmitted proctitis (i.e., syphilis and lymphogranuloma venereum):
- Mucosal expansion by plasma cells and histiocytes
- Granulomas (rare)
- Relatively less crypt architecture distortion and Paneth cell metaplasia when compared to ulcerative proctitis (Am J Clin Pathol 2015;144:771)
- Positive spirochete immunohistochemical stain in cases of syphilis proctitis
- Correlation with gold standard clinical tests (rapid plasma reagin [RPR], Treponema specific serology, nucleic acid amplification [NAAT] using rectal swabs)
- Clinical history of receptive anal intercourse
- Acute infectious (self limited) proctitis:
- Neutrophils predominate in lamina propria
- Lack of crypt architecture distortion
- Lack of basal plasmacytosis
- Lack of Paneth cell metaplasia
- Correlation with clinical tests on stool samples (such as multiplex PCR panels)
- CMV proctitis:
- May complicate ulcerative proctitis in patients on immunosuppressive treatment
- Endothelial cells, mesenchymal stromal cells or glandular epithelial cells may show viral cytopathic effect, including enlargement (cytomegaly) and inclusions (owl eye intranuclear inclusions or pink cytoplasmic inclusions)
- CMV immunohistochemical stain is an extremely useful adjunct
- Diversion proctitis:
- Prominent mucosal / submucosal lymphoid hyperplasia
- History of fecal stream diversion (i.e., ileostomy or colostomy proximal to the rectum)
- Often seen in completion proctectomy specimens in patients with idiopathic inflammatory bowel disease undergoing multistage ileal pouch anal anastomosis (IPAA) procedures
- Immune checkpoint inhibitor associated proctitis:
- Several patterns of injury have been described, including active colitis, lymphocytic colitis, collagenous colitis and apoptosis with increased intraepithelial apoptosis, apoptotic crypt abscesses and multifocal crypt injury (withered / shrunken crypts) (Am J Surg Pathol 2022;46:e15)
- Mucosal expansion, crypt distortion, cryptitis and crypt abscesses may also be seen, mimicking idiopathic ulcerative proctitis
- Clinical history of immune checkpoint inhibitor use
- Ischemic proctitis:
- Uncommon in rectum due to dual blood supply from inferior mesenteric and iliac arteries
- Surface active inflammation / necrosis
- Hyalinized lamina propria
- Withered / shrunken crypts
- Ischemic type changes may be seen in stercoral ulceration, trauma and solitary rectal ulcer syndrome
- Solitary rectal ulcer syndrome (SRUS):
- Microscopic changes imparted by rectal prolapse: fibromuscular hyperplasia of lamina propria splaying basal crypts (resulting in angulated or diamond shaped crypts), ectatic and congested subepithelial capillaries, surface ulceration, pseudomembranes
- Stercoral ulcer:
- Ulceration secondary to impacted fecal material
- Microscopically shows ulcer and focal ischemic changes variable embedded fecal material
- Background mucosa should be relatively uninflamed
Practice question #1
A 63 year old woman presents to her primary care physician with a 3 month history of bloody diarrhea and tenesmus. She is referred for a colonoscopy, which demonstrates an erythematous friable mucosa with erosions in the rectum, as demonstrated in the figure above. Biopsies are performed. Which of the following histologic findings would most suggest idiopathic ulcerative proctitis in this case?
- Follicular lymphoid hyperplasia and cryptitis
- Increased intraepithelial apoptosis and multifocal crypt injury
- Lamina propria hyalinization and withered / shrunken crypts
- Mucosal expansion, crypt architecture distortion and crypt abscesses
- Prominent lamina neutrophils and preserved crypt architecture
Practice answer #1
D. Mucosal expansion, crypt architecture distortion and crypt abscesses are all features characteristic of idiopathic ulcerative proctitis. Answer A describes diversion proctitis. Answer B describes histology that can be seen in immune checkpoint inhibitor associated colitis. Answer C describes ischemic proctitis. Answer E describes acute infectious (self limited) proctitis.
Comment Here
Reference: Ulcerative proctitis
Comment Here
Reference: Ulcerative proctitis
Practice question #2
A 22 year old man with a history of ulcerative proctitis undergoes surveillance colonoscopy. The rectum appears erythematous and granular. Biopsies are taken of the rectal mucosa and are sent for histopathologic evaluation. Which of the following, if found in the biopsy, would provide the best histologic evidence of active disease?
- Basal plasmacytosis (crypt shortfall)
- Crypt architecture distortion
- Cryptitis
- Mucosal expansion
- Paneth cell metaplasia
Practice answer #2
C. Cryptitis (neutrophilic infiltration of the crypt epithelium) is a form of active mucosal inflammation and is evidence of active disease. The other forms of injury listed (basal plasmacytosis, crypt architecture distortion, mucosal expansion and Paneth cell metaplasia) are evidence of chronicity in ulcerative proctitis.
Comment Here
Reference: Ulcerative proctitis
Comment Here
Reference: Ulcerative proctitis