Table of Contents
Definition / general | Essential features | Diagrams / tables | Metrics | Bethesda categories | Major updates in TBS 2023 | Cytology images | Videos | Additional references | Practice question #1 | Practice answer #1 | Practice question #2 | Practice answer #2Cite this page: Suzuki A, Bychkov A. Bethesda system diagnostic categories. PathologyOutlines.com website. https://www.pathologyoutlines.com/topic/thyroiddiagnostic.html. Accessed August 18th, 2025.
Definition / general
- The Bethesda System for Reporting Thyroid Cytopathology (TBS) is an international reporting system for thyroid cytology (Ali: The Bethesda System for Reporting Thyroid Cytopathology, 3rd Edition, 2023)
- Structure
- 6 categories with different propensity to malignancy, from benign to malignant (4 - 97%), each assigned management approach
- Indeterminate categories constitute categories III - V and may require molecular testing to tailor clinical decision (i.e., surgery versus conservative) (Molecular testing in FNA)
- Other national systems exist, including contemporary British, Japanese, Italian and outdated Papanicolaou, which could be translated into TBS terminology (Cancer Cytopathol 2016;124:457)
Essential features
- TBS is an international standard for reporting thyroid FNA
- Uniform terminology aimed to standardize the reporting of thyroid fine needle aspiration (FNA) cytology
- Understandable by various specialists in different countries
- In conjunction with the International Academy of Cytology, endorsed by the American Thyroid Association and other leading professional communities (Thyroid 2016;26:1)
- Currently widely adopted worldwide
- Similar to reporting systems in other organs (Adv Anat Pathol 2016;23:193, Cancer Cytopathol 2020;128:348)
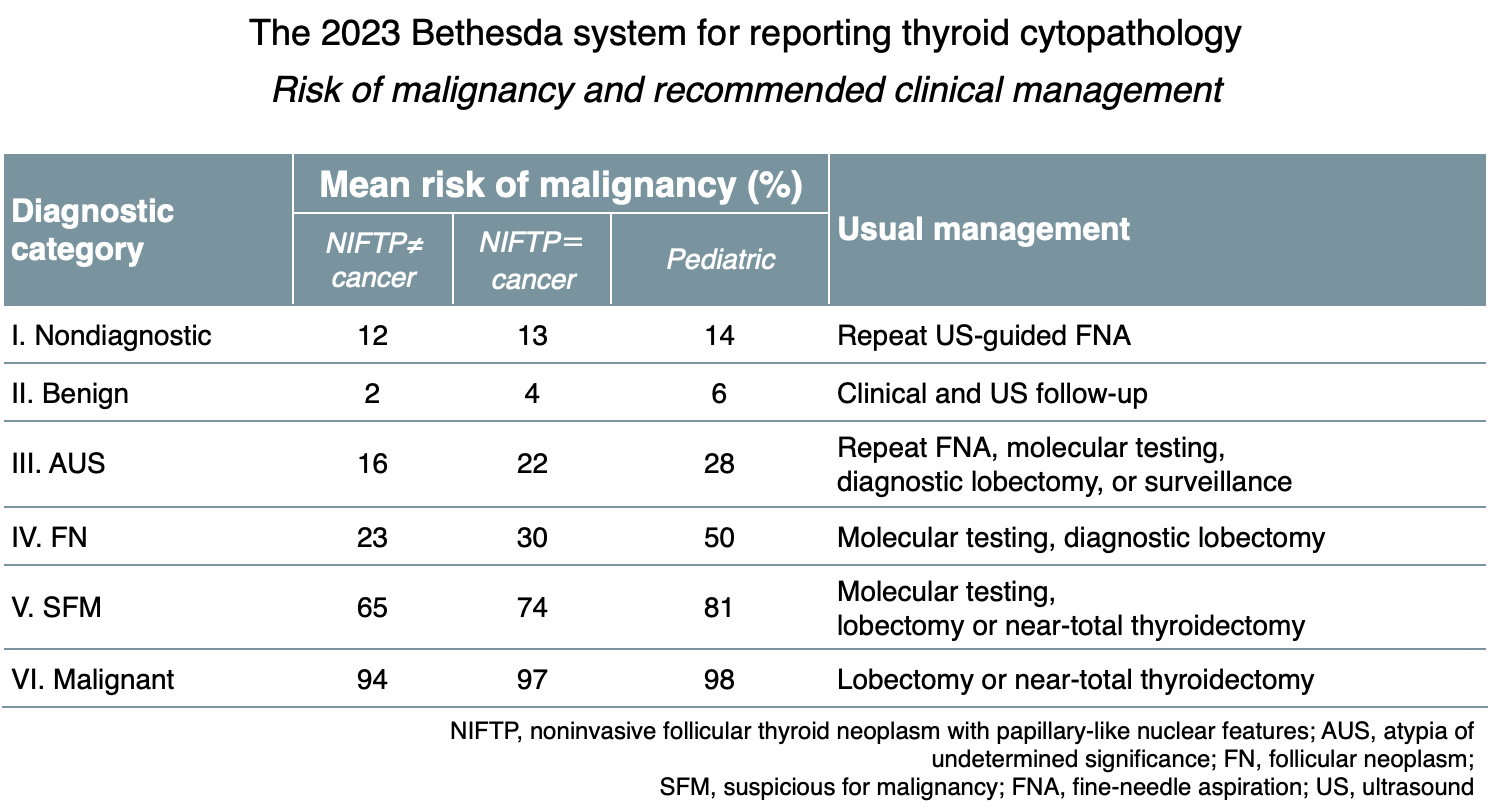
- Divided into 6 categories that are linked to malignancy risk and recommended clinical management
- Clinically significant statistical metrics of each category are frequency, resection rate and risk of malignancy
Diagrams / tables
Metrics
- TBS metrics / outputs include several statistical indicators
- Frequency: number of nodules in a given category out of all aspirated nodules
- Malignancy risk, also known as risk of malignancy (ROM): number of malignant nodules on surgery out of all resected in a given category
- Indicators not currently included in TBS
- Resection rate (RR): ratio of operated / resected nodules out of all aspirated nodules in a category
- Overall risk of malignancy (oROM): calculated out of all aspirated nodules, including resected and unresected (Cancer Cytopathol 2016;124:181)
- Risk of neoplasia (RON): number of neoplastic nodules (benign plus malignant) on surgery out of all resected in a given category (Cancer Cytopathol 2020;128:232)
- Utility
- Defines management of thyroid nodules: high ROM suggests surgical resection while low ROM implies conservative strategy (e.g., follow up)
- Quality control in lab / hospital
- High nondiagnostic rate (> 20%) requires revision of FNA sampling procedure and lab workflow (e.g., implementation of rapid onsite evaluation or ultrasound guided sampling)
- Abnormal measures, such as high rate of indeterminate diagnoses, low ROM in malignant nodules or high ROM in benign nodules may be due to limited expertise of FNA readers
- Variation in TBS outputs depends on different factors
- Institutions: reference center versus primary care; expertise of operator and reader
- Geography: North America versus Asia, due to different practice patterns (Cancer Cytopathol 2020;128:238)
- Population: adults versus children (Thyroid 2021;31:1203)
- Classification of noninvasive follicular thyroid neoplasm with papillary-like nuclear features (NIFTP) either as benign or malignant tumor (Endocr Pract 2019;25:491)
- All of the above explain differences between the model outputs implied by TBS and real world data reported in meta analysis studies
- Ideally, local institutional metrics (especially ROM) should be calculated to adjust clinical decisions in a given hospital (Cancer Cytopathol 2020;128:917)
Bethesda categories
- TBS I: nondiagnostic
- Includes inadequate by cellularity, unsatisfactory by quality and cyst fluid only specimens
- TBS criteria for adequacy of thyroid FNA specimens is ≥ 6 groups of well visualized follicular cells (≥ 10 per cluster)
- Frequency 3 - 34%, resection rate 7 - 15%, ROM 10 - 13% of all nodules and up to 30% of resected nodules
- Management: reaspiration, except for pure cyst
- TBS II: benign
- Cytology sample that is adequate for evaluation and consists of colloid and benign appearing follicular cells
- Frequency 60 - 70%, resection rate 10%, ROM < 3%
- Follicular nodular disease is the most common lesion
- Management: follow up based on ultrasound pattern
- TBS III: atypia of undetermined significance (AUS)
- Aspirates with few cells that have distinct but mild nuclear atypia or with more extensive cell numbers but subtle nuclear atypia
- Frequency 1 - 20%, resection rate 43 - 65%, ROM 13 - 30% (NIFTP = malignant) or 6 - 20% (NIFTP ≠ malignant) and up to 40% of resected TBS III nodules
- AUS is subdivided into AUS with nuclear atypia and other atypia
- ROM: AUS with nuclear atypia 36 - 44%, AUS with other atypia 15 - 23%
- On resection / histopathology diagnosed as follicular nodular disease, follicular adenoma and papillary thyroid carcinoma (PTC)
- Management: reaspiration or molecular testing (Thyroid 2016;26:1)
- TBS IV: follicular neoplasm (FN)
- Cases with most of the follicular cells arranged in cell crowding or microfollicle formation
- Frequency 7%, resection rate 42 - 45%, ROM 23 - 34% (NIFTP = malignant) or 23% (NIFTP ≠ malignant)
- Histopathology: follicular adenoma, follicular nodular disease, NIFTP, follicular variant of papillary thyroid carcinoma and follicular carcinoma
- Management: diagnostic thyroid lobectomy or molecular testing
- TBS IV: follicular neoplasm (oncocytic follicular neoplasm [FN-OFN])
- Cases with most of the follicular cells showing abundant fine granular cytoplasm (oncocytes)
- Frequency 1.2 - 9%, resection rate 30%, ROM 25 - 50%
- Histopathology: oncocytic adenoma and carcinoma
- Management: diagnostic thyroid lobectomy or molecular testing
- TBS V: suspicious for malignancy
- Used when cytology strongly suggests malignancy but is not sufficient for a conclusive diagnosis
- Frequency 3%, resection rate 70%, ROM 74% (NIFTP = malignant) or 65% (NIFTP ≠ malignant)
- Histopathology: usually PTC
- Management: surgery (usually)
- TBS VI: malignant
- Used when cytology strongly suggests malignancy
- Frequency 5%, resection rate 65 - 80%, ROM 97 - 100% (NIFTP = malignant) or 94% (NIFTP ≠ malignant)
- Histopathology: wide spectrum of thyroid malignancies, from PTC (most common) to medullary thyroid carcinoma, anaplastic thyroid carcinoma, lymphoma, etc.
- Management: surgery (usually)
- TBS 2023 continues to advise using the category names and optionally, their corresponding category numbers (e.g., benign [Bethesda II], follicular neoplasm [Bethesda IV])
Major updates in TBS 2023
- Single distinct name for each category
- Each category is associated with updated ROM based on new data since the 2017 edition; clinical management algorithms for each category have also been revised
- Subdivision of AUS category into AUS - nuclear atypia and AUS - other, with different implied ROM (higher in AUS - nuclear) and molecular profile
- New discussions on pediatric thyroid disease have been included, supplied with pediatric ROMs and management algorithms
- Revised terminology in accordance with the 2022 WHO classification of thyroid tumors
- Reference: Thyroid 2023;33:1039
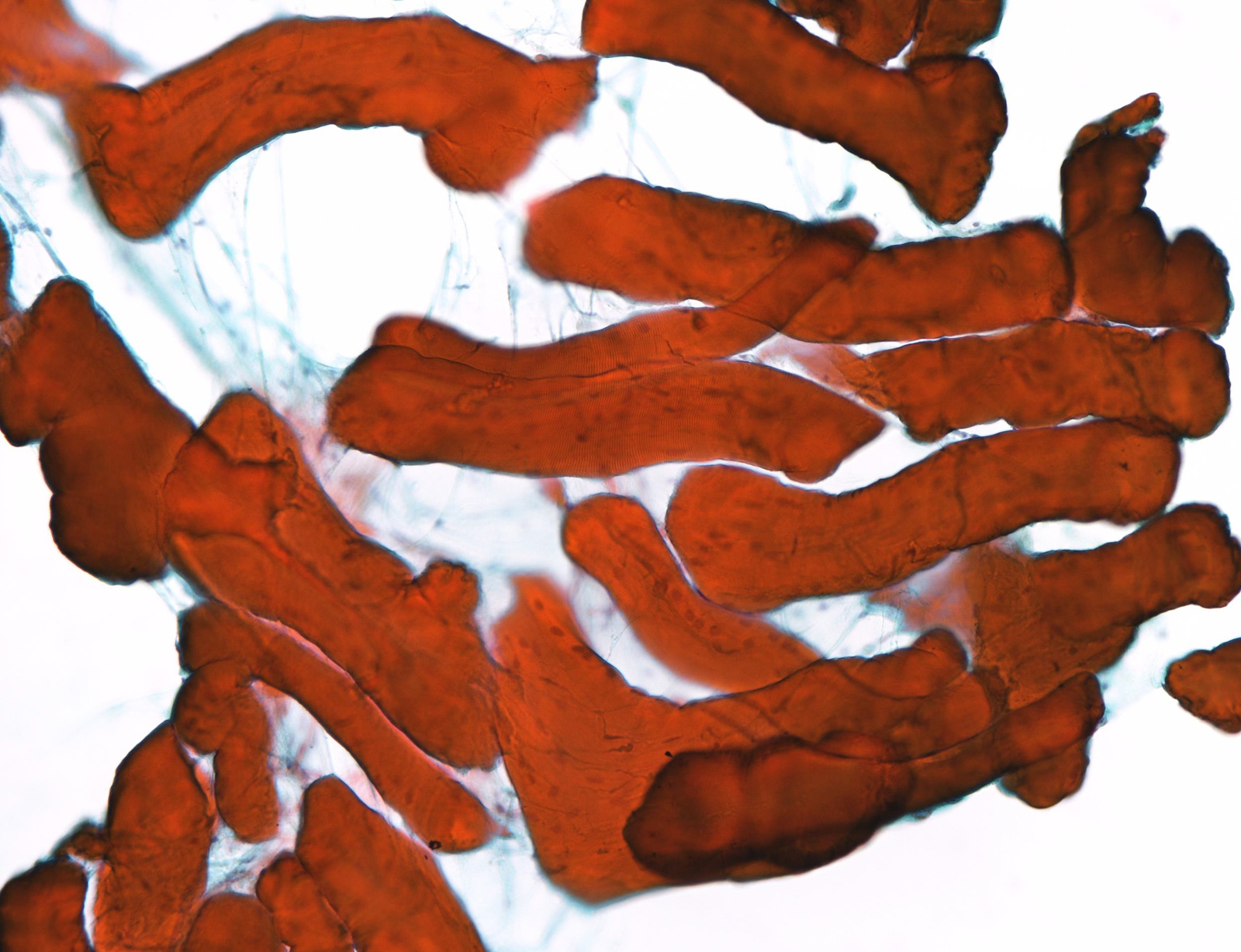
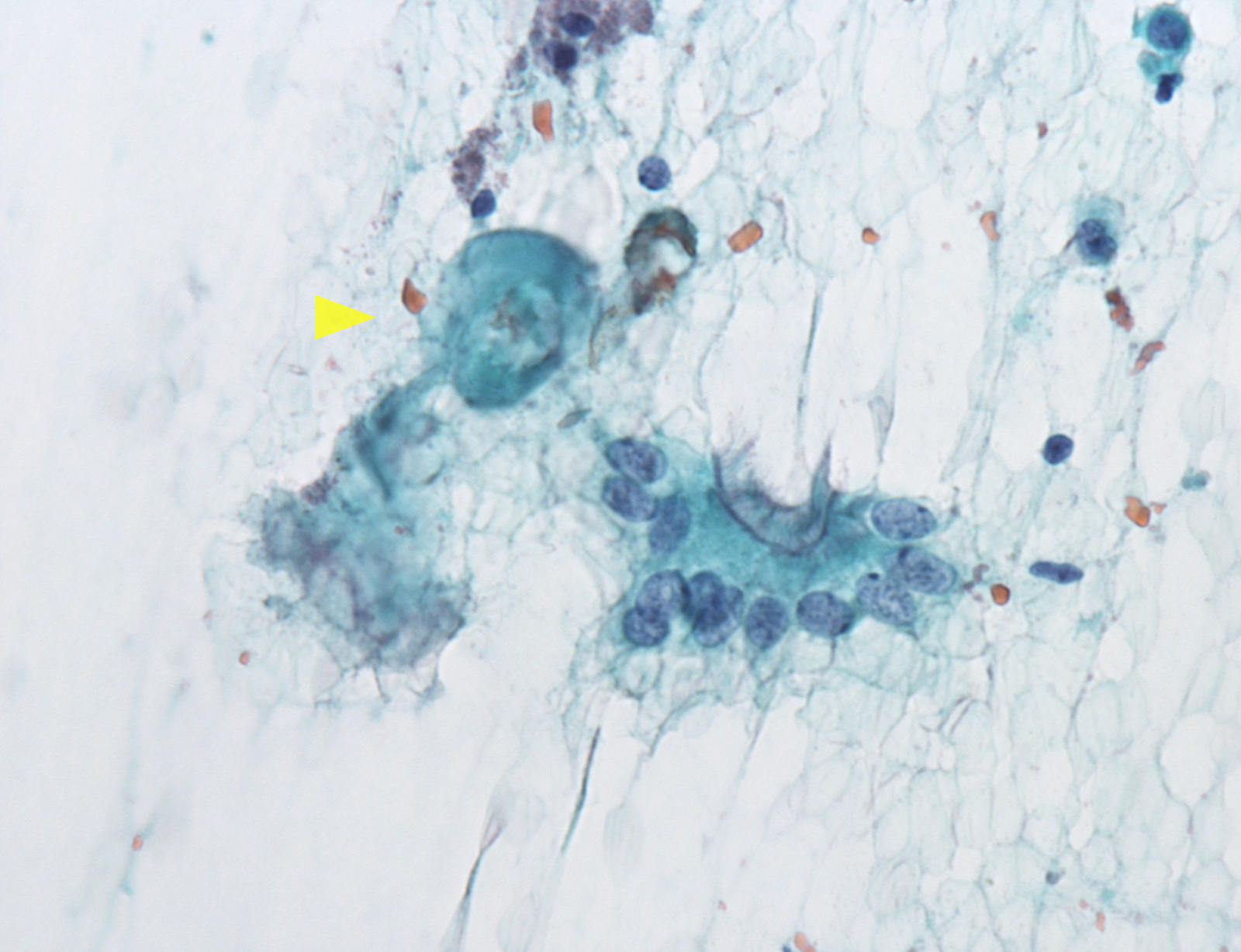
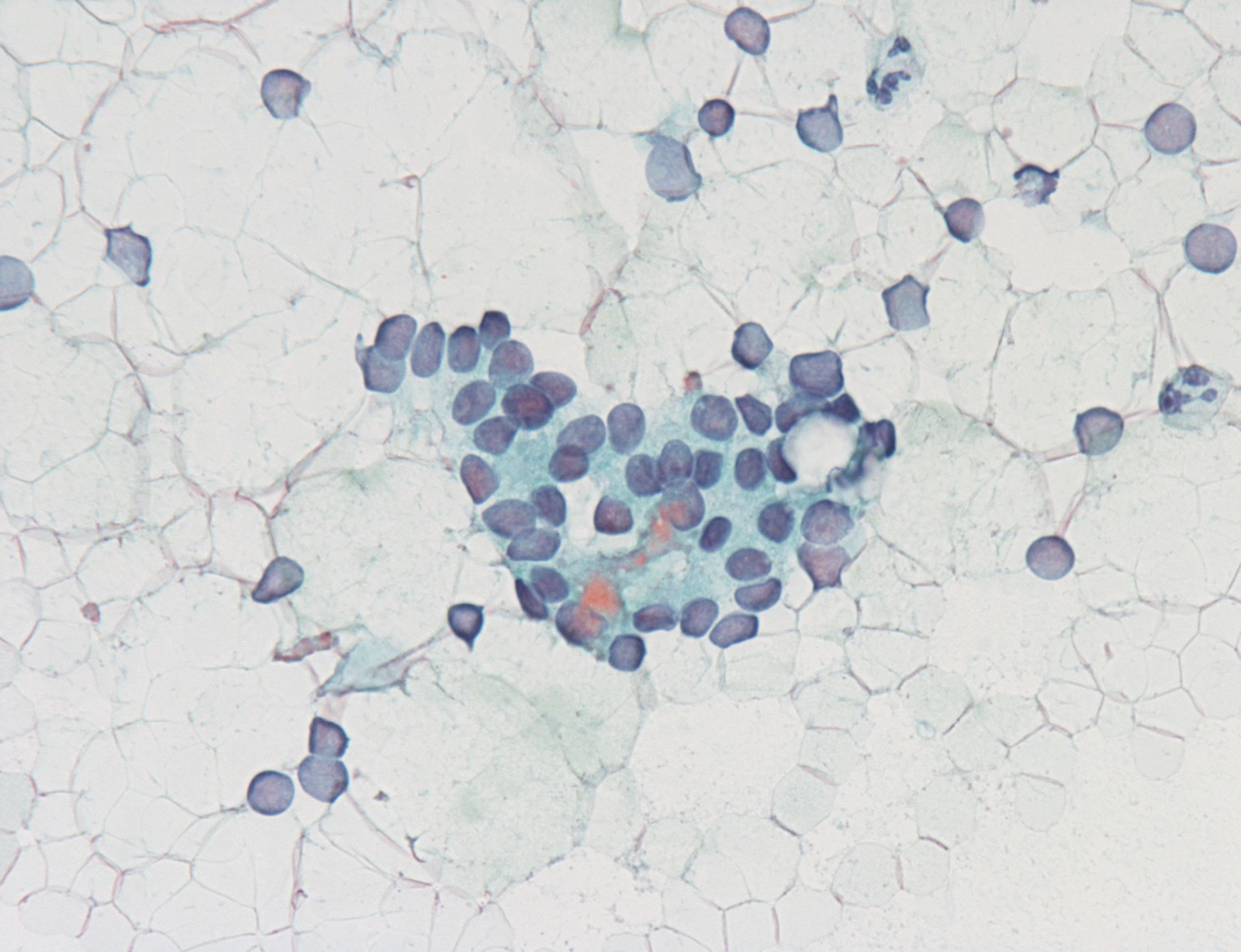
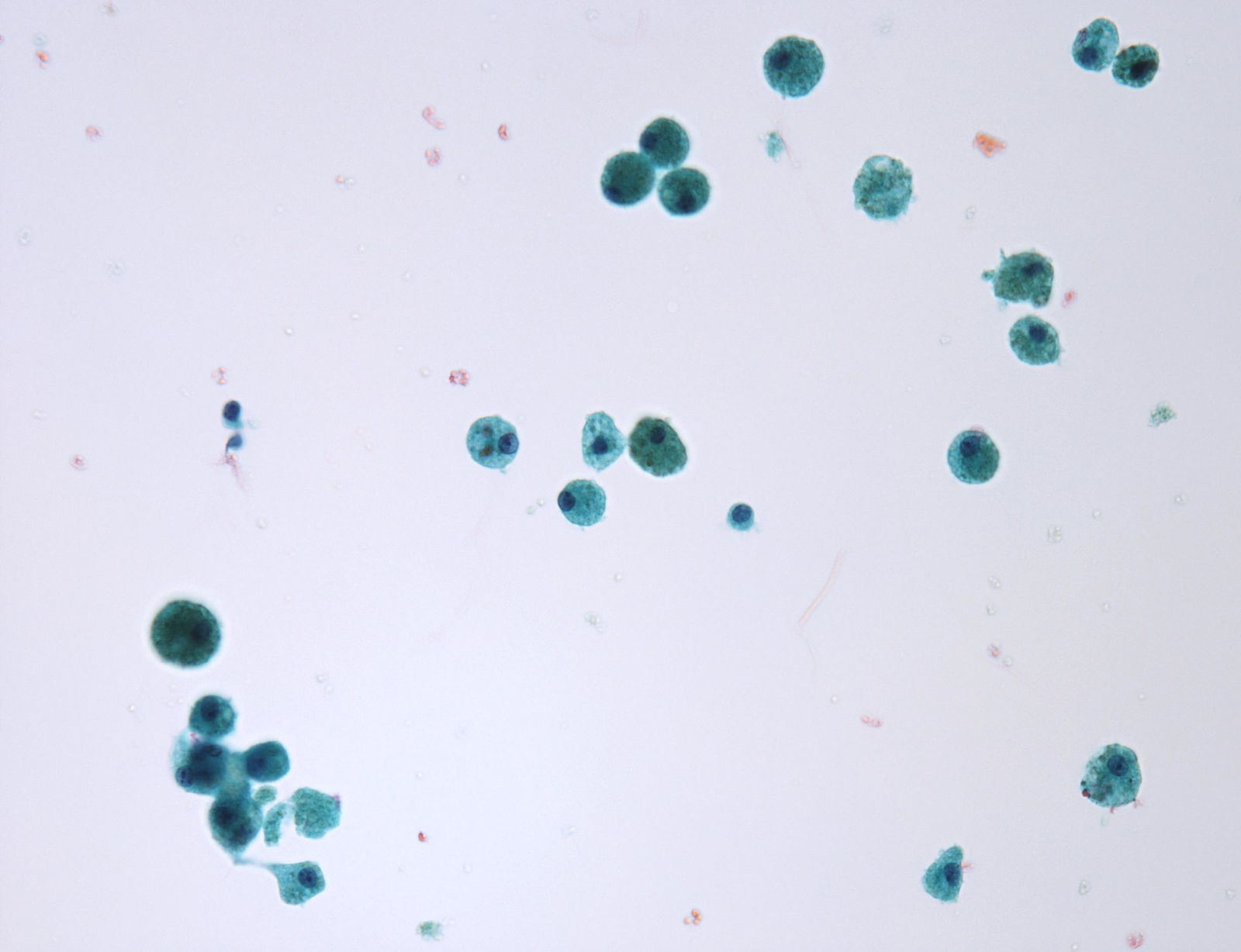
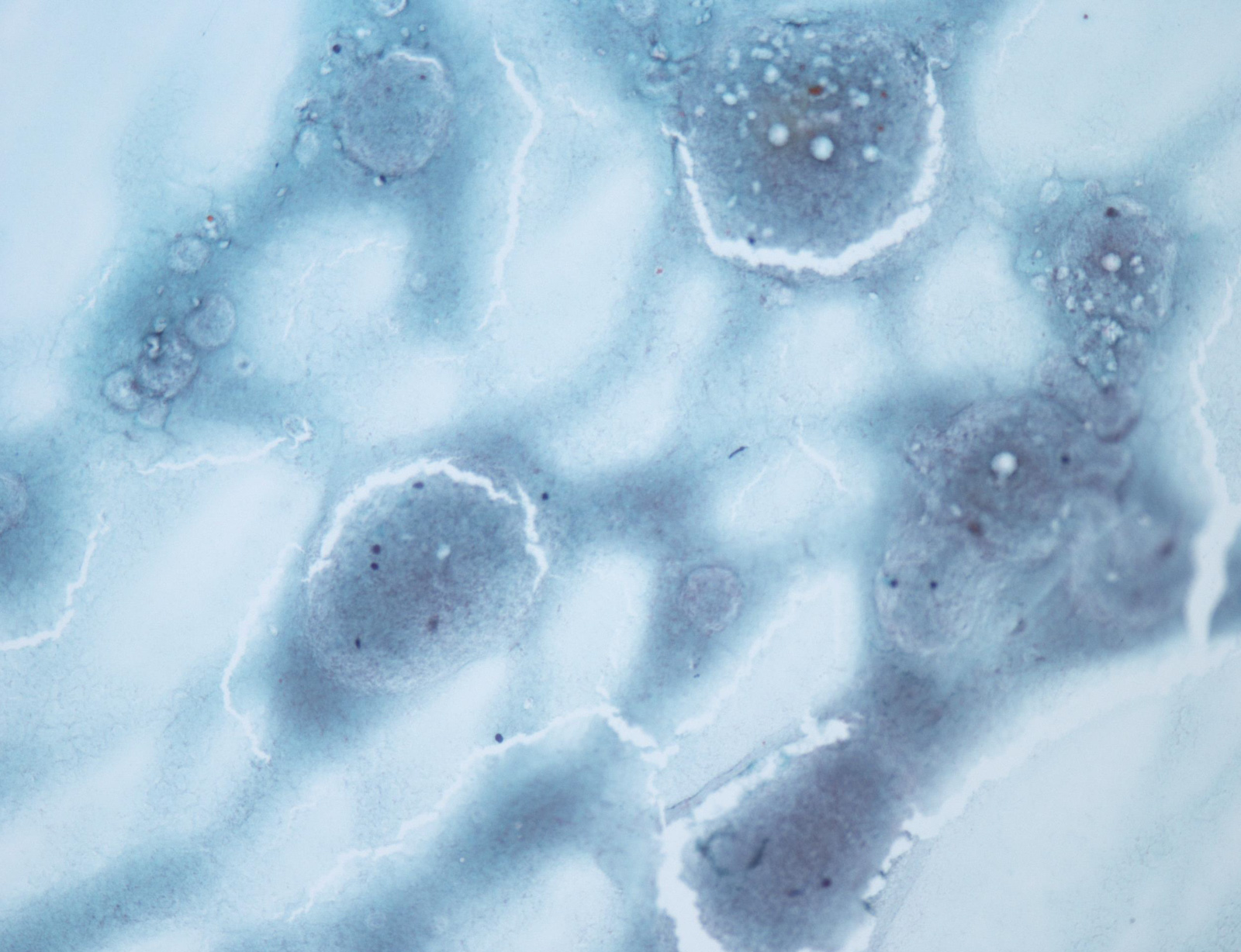
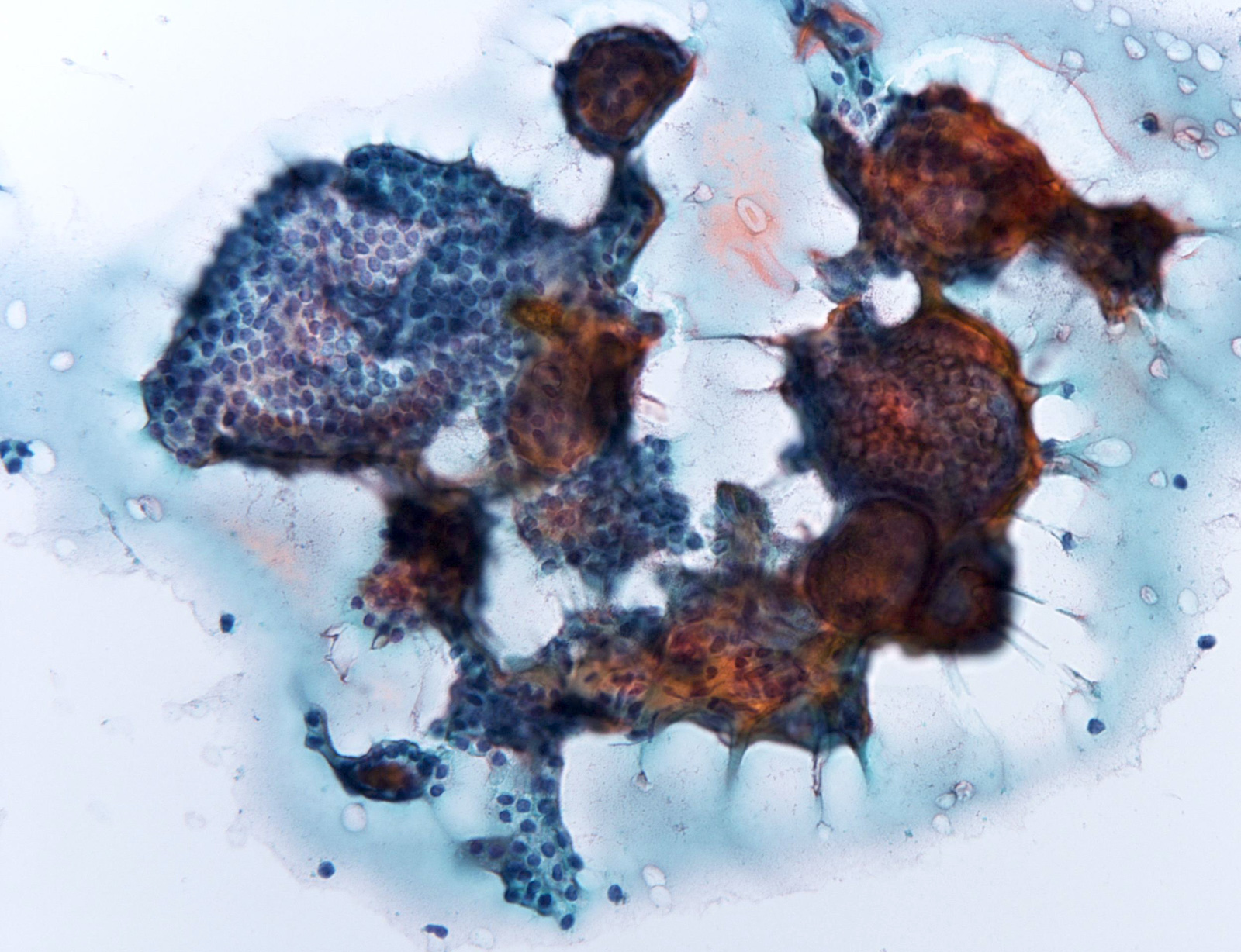
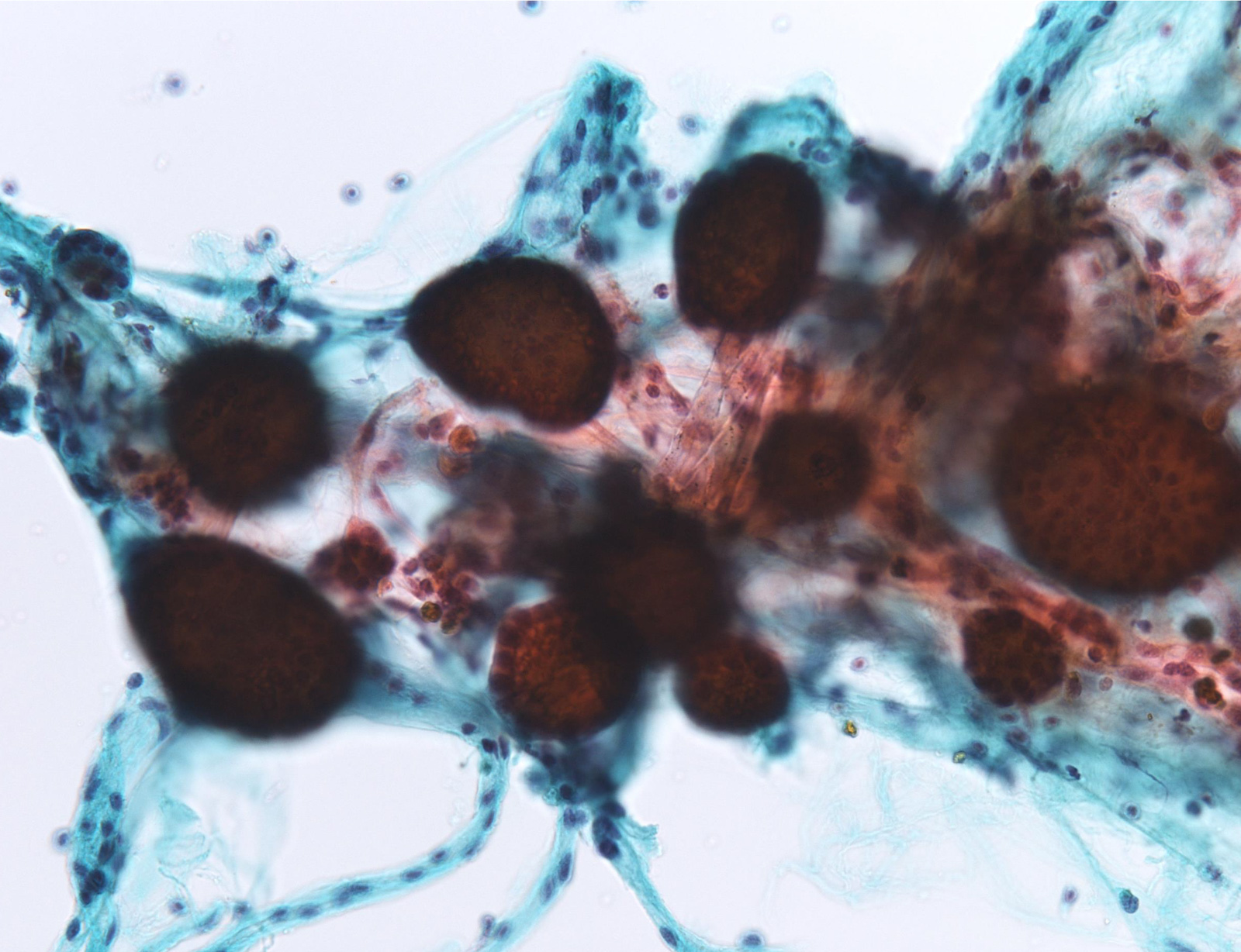
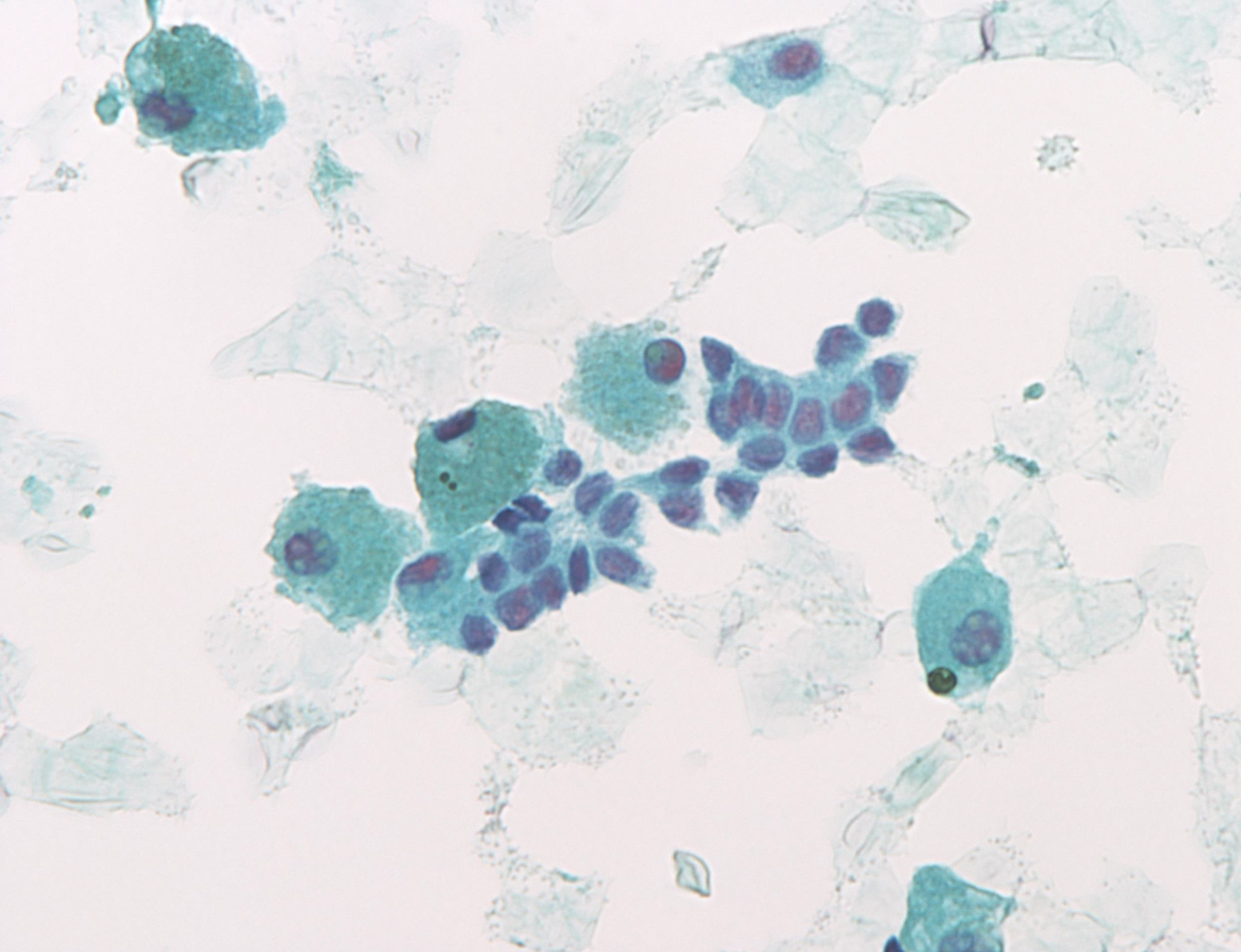
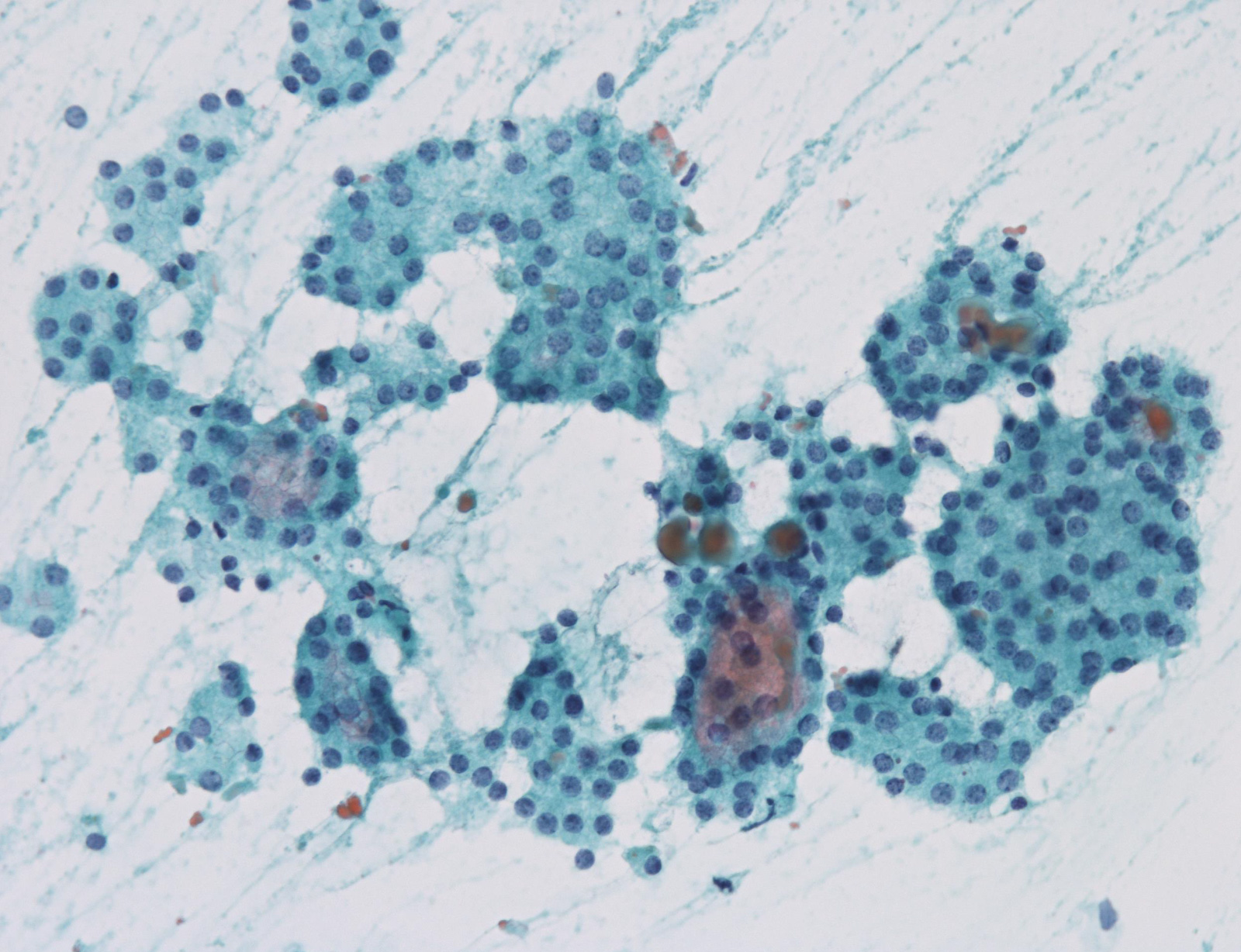
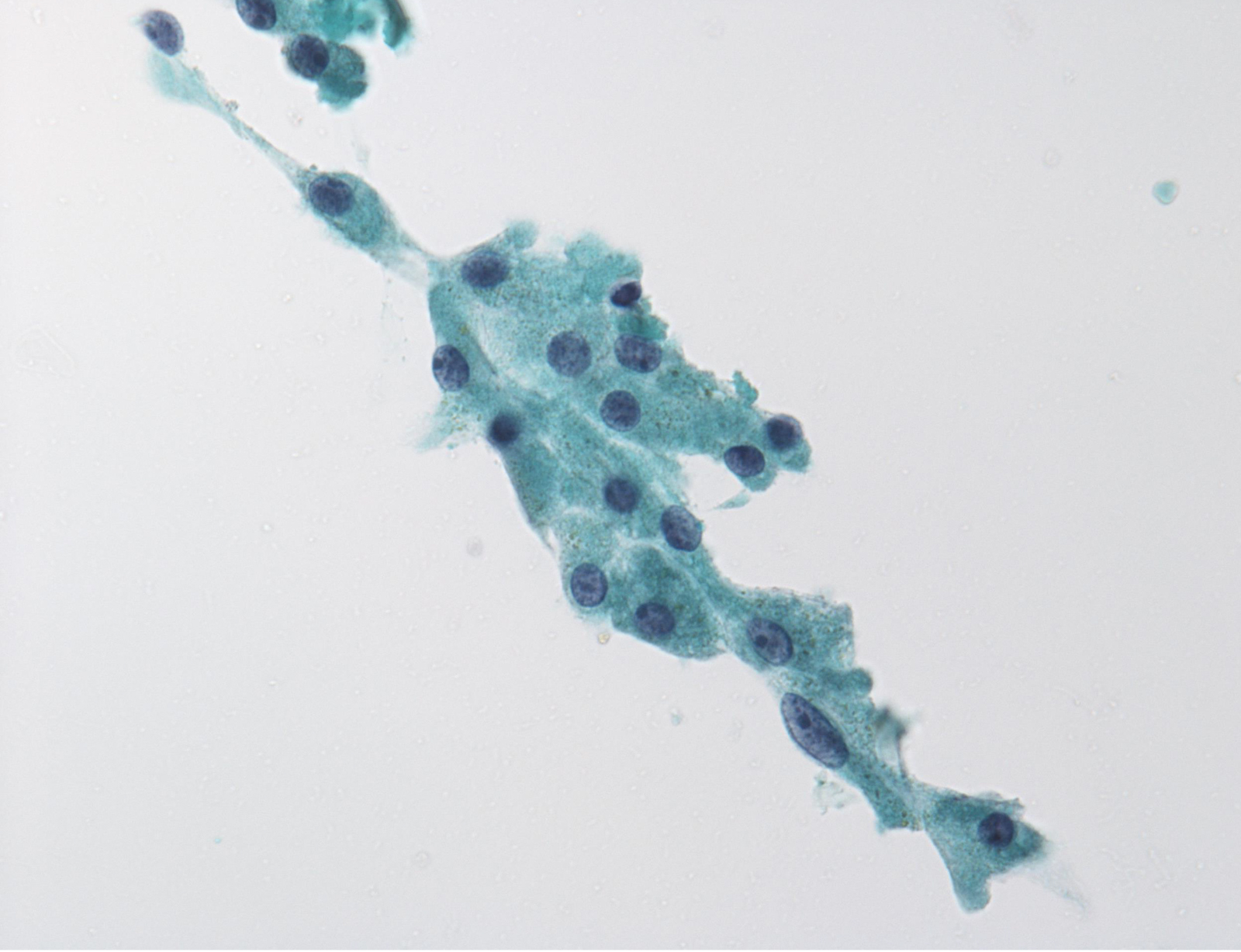
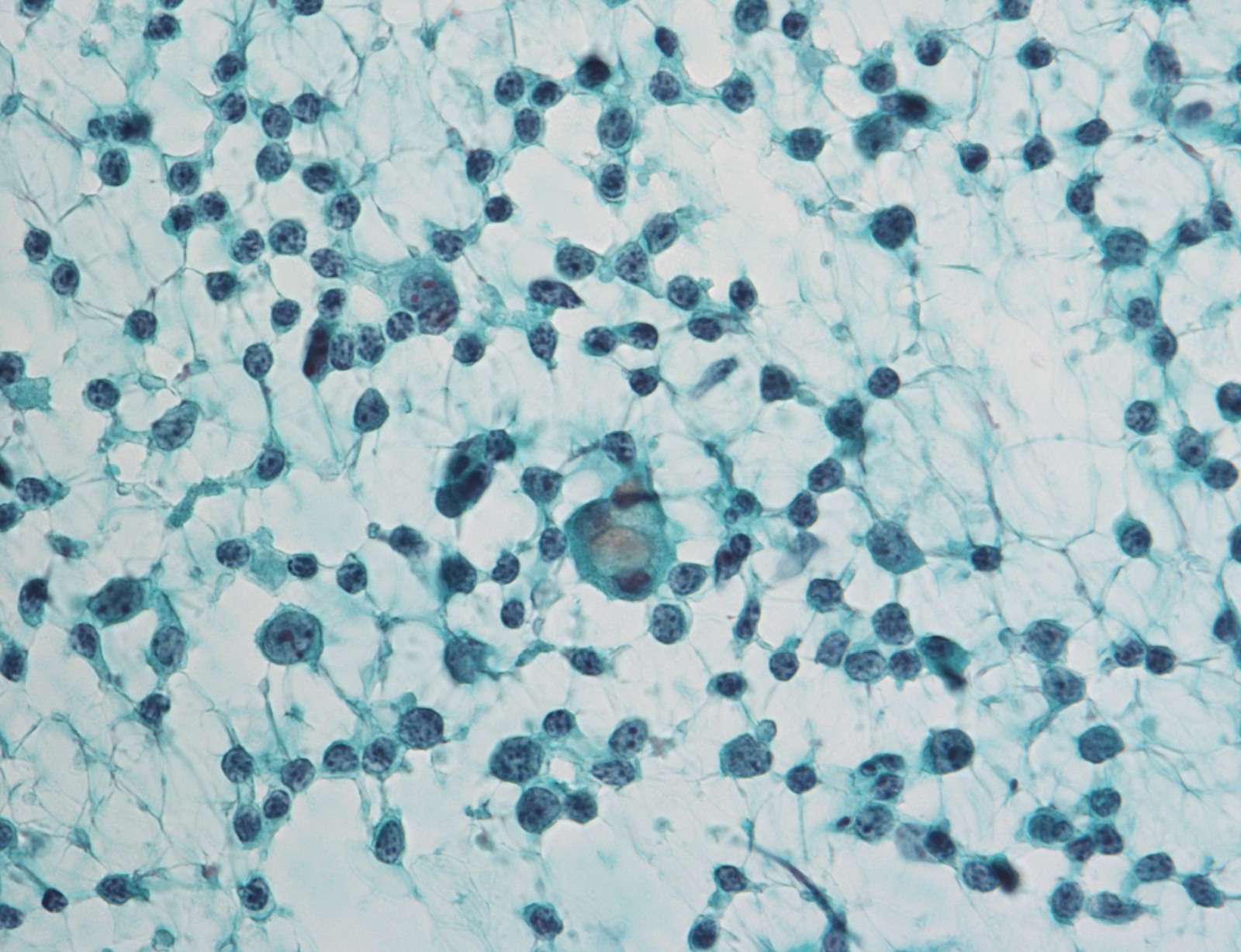
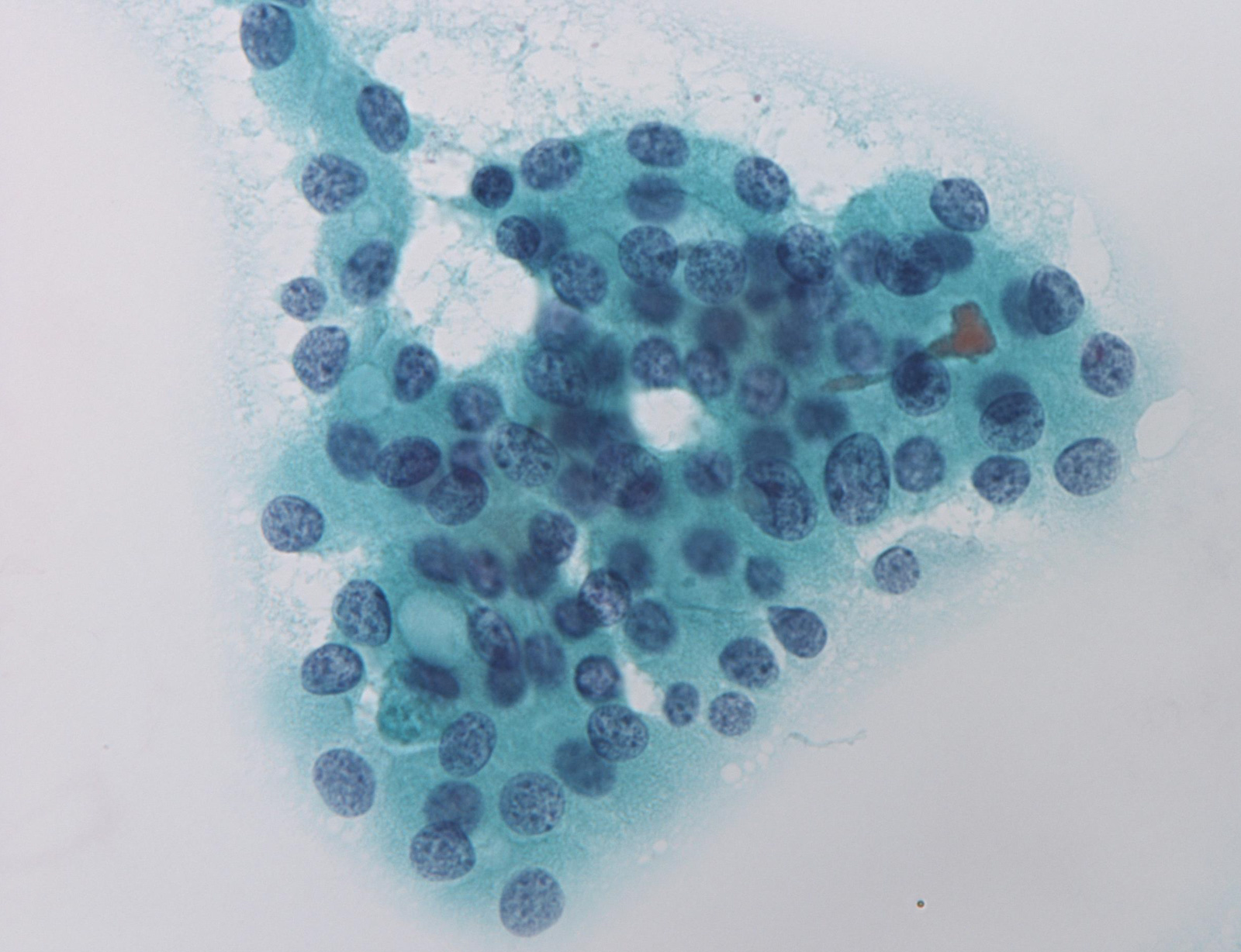
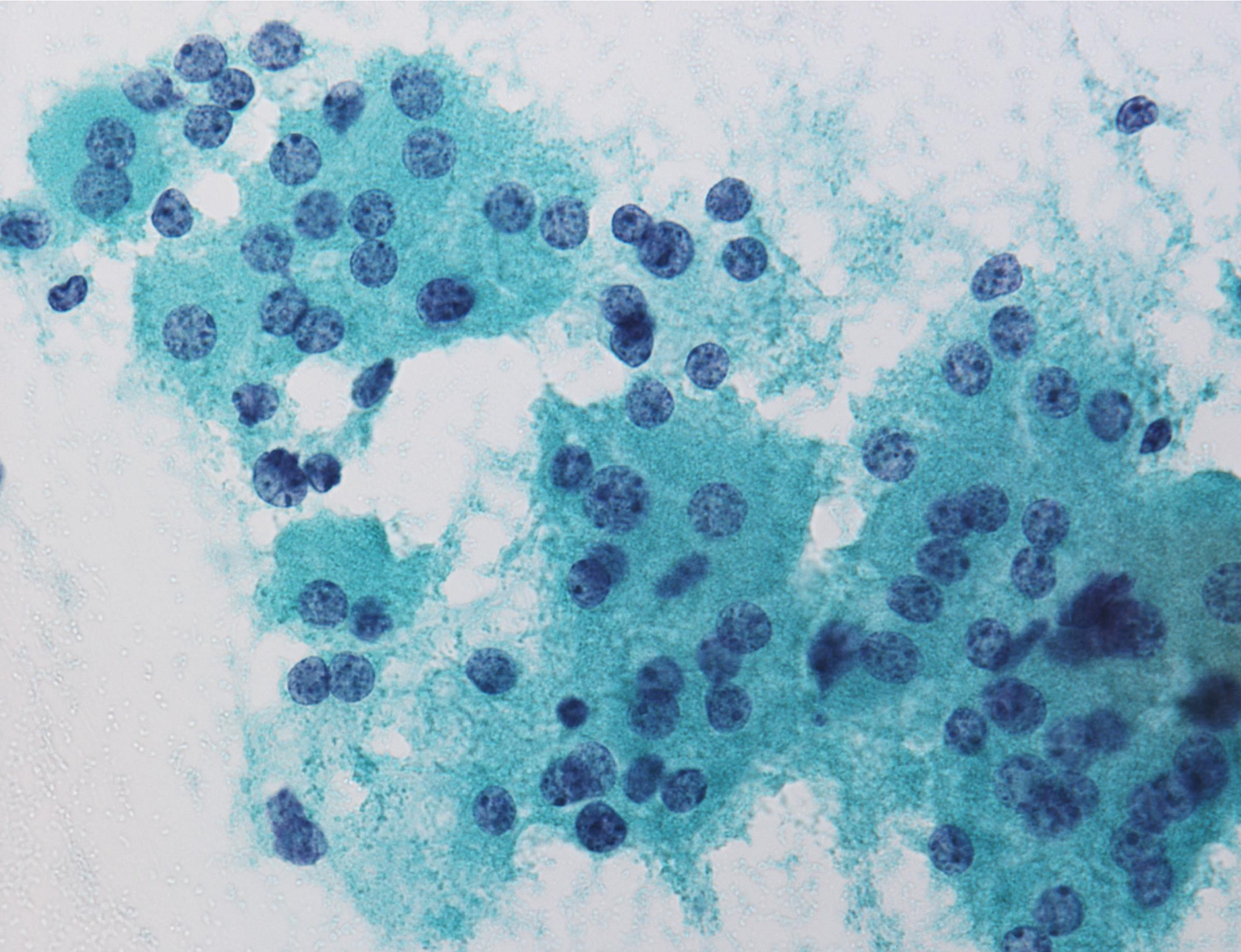
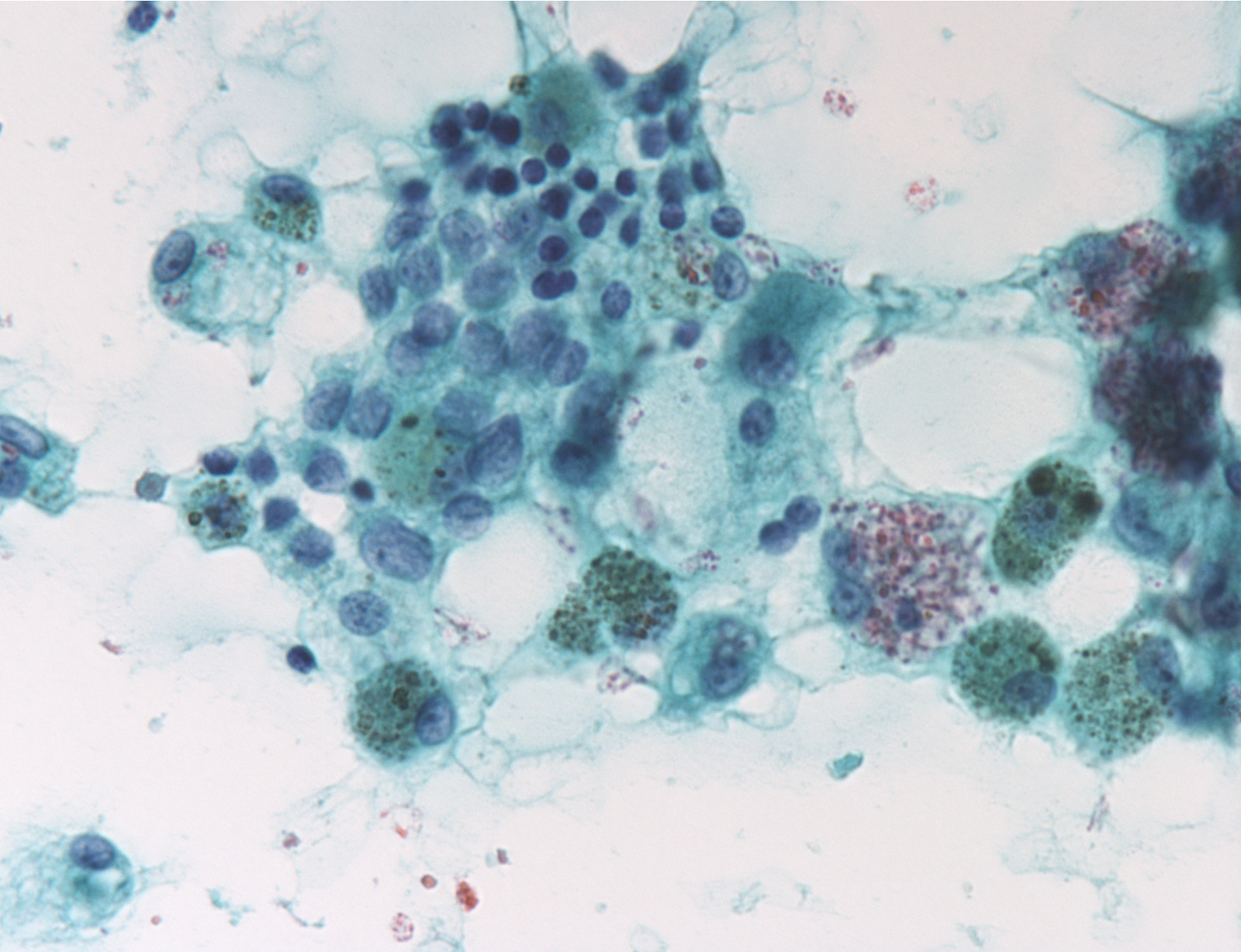
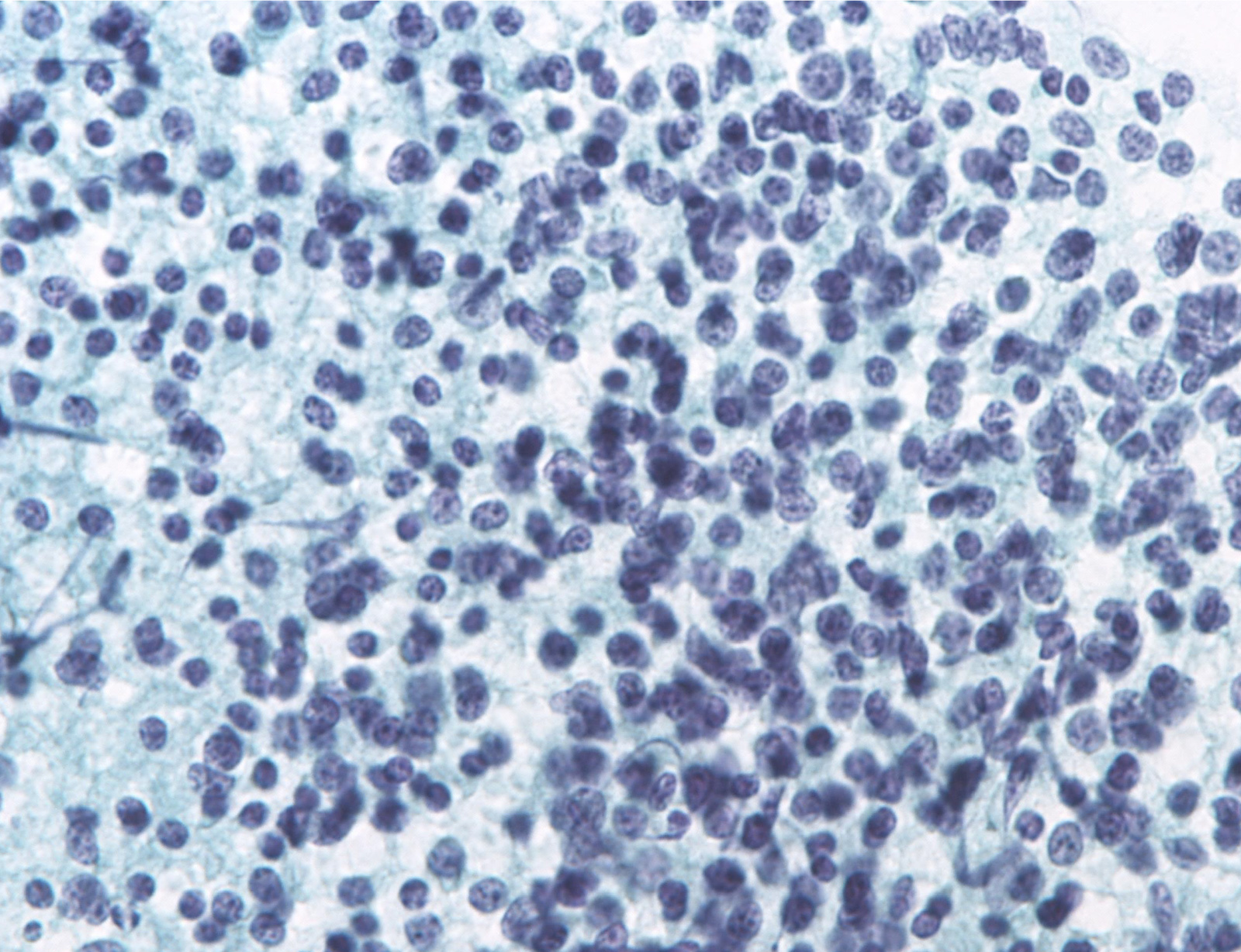
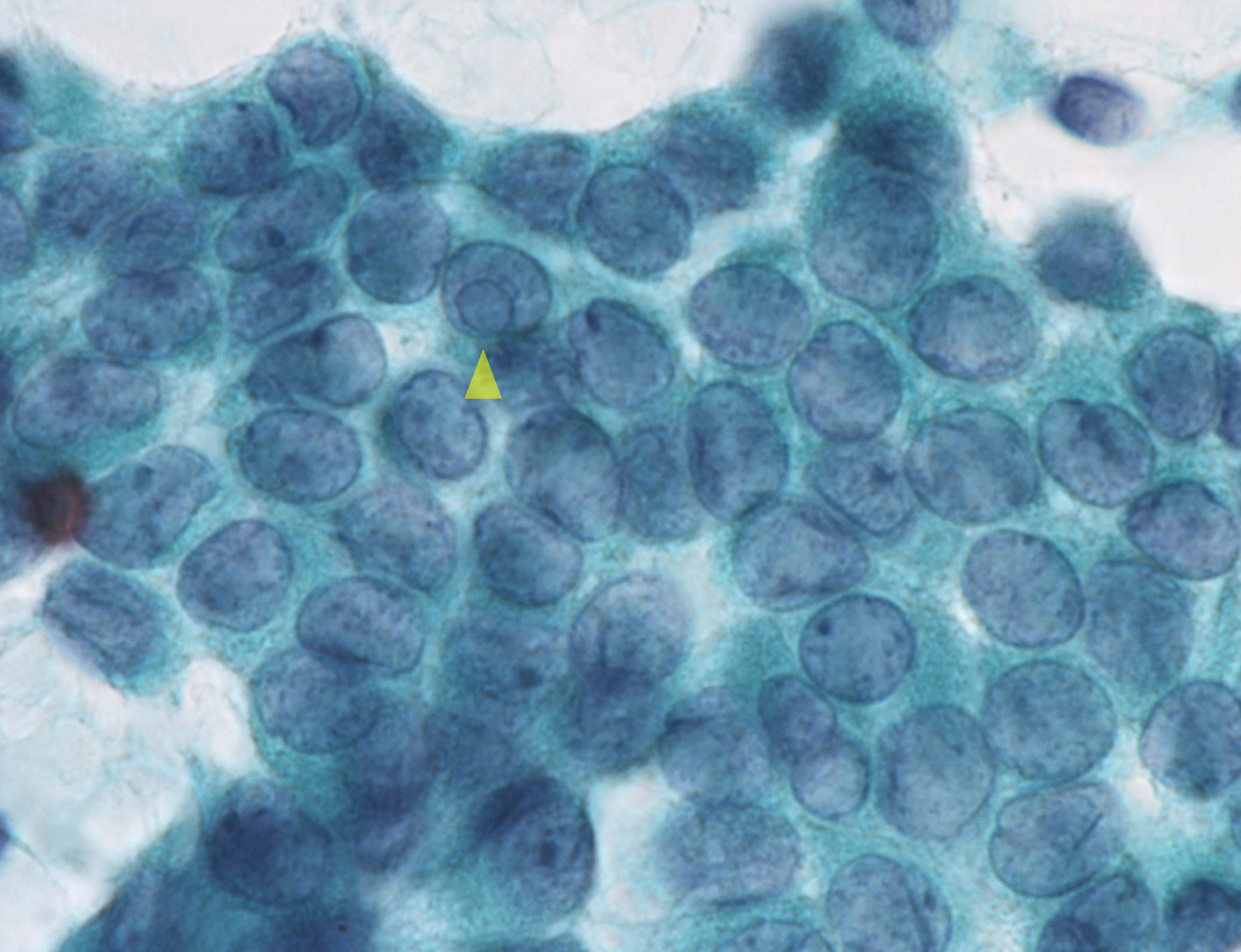
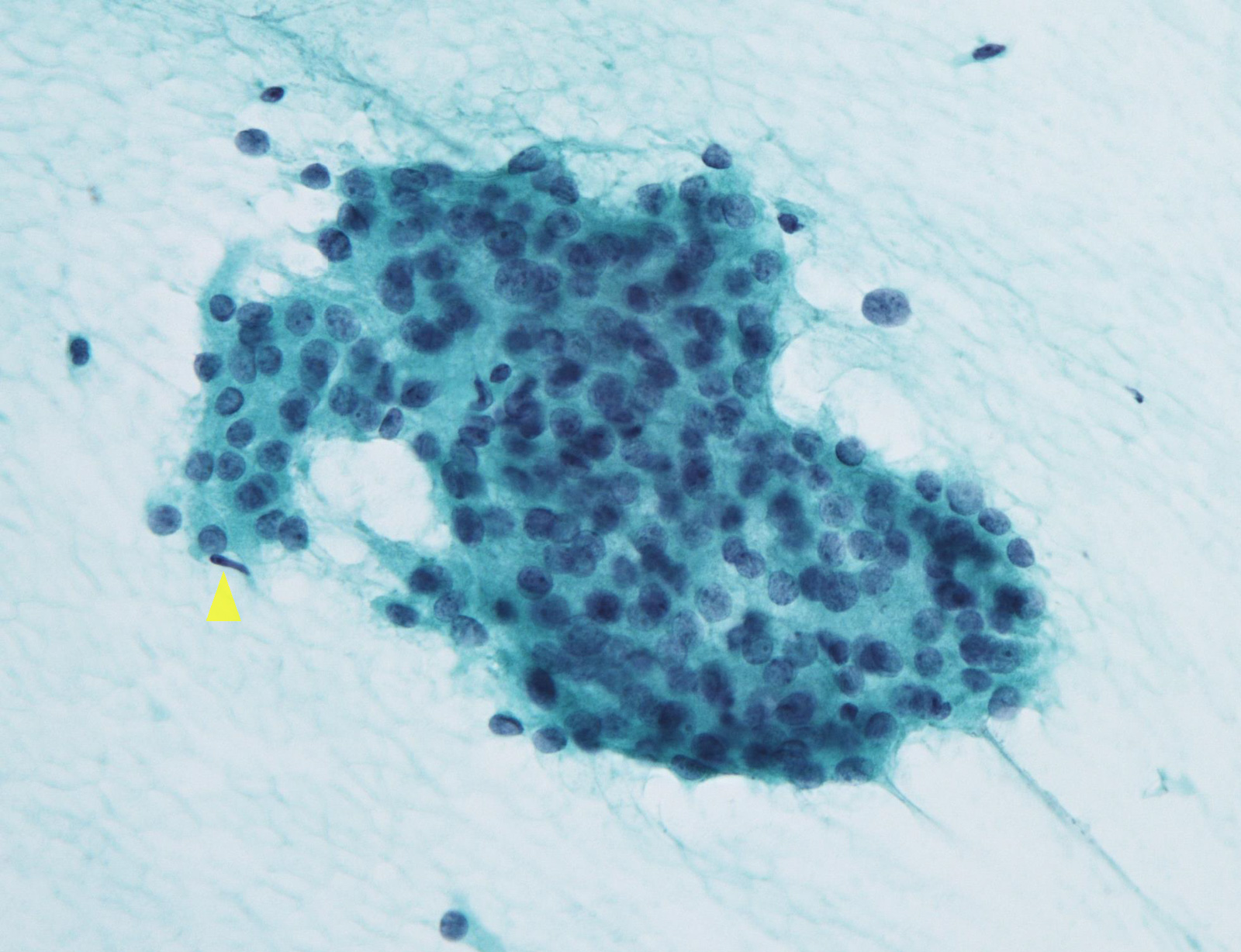
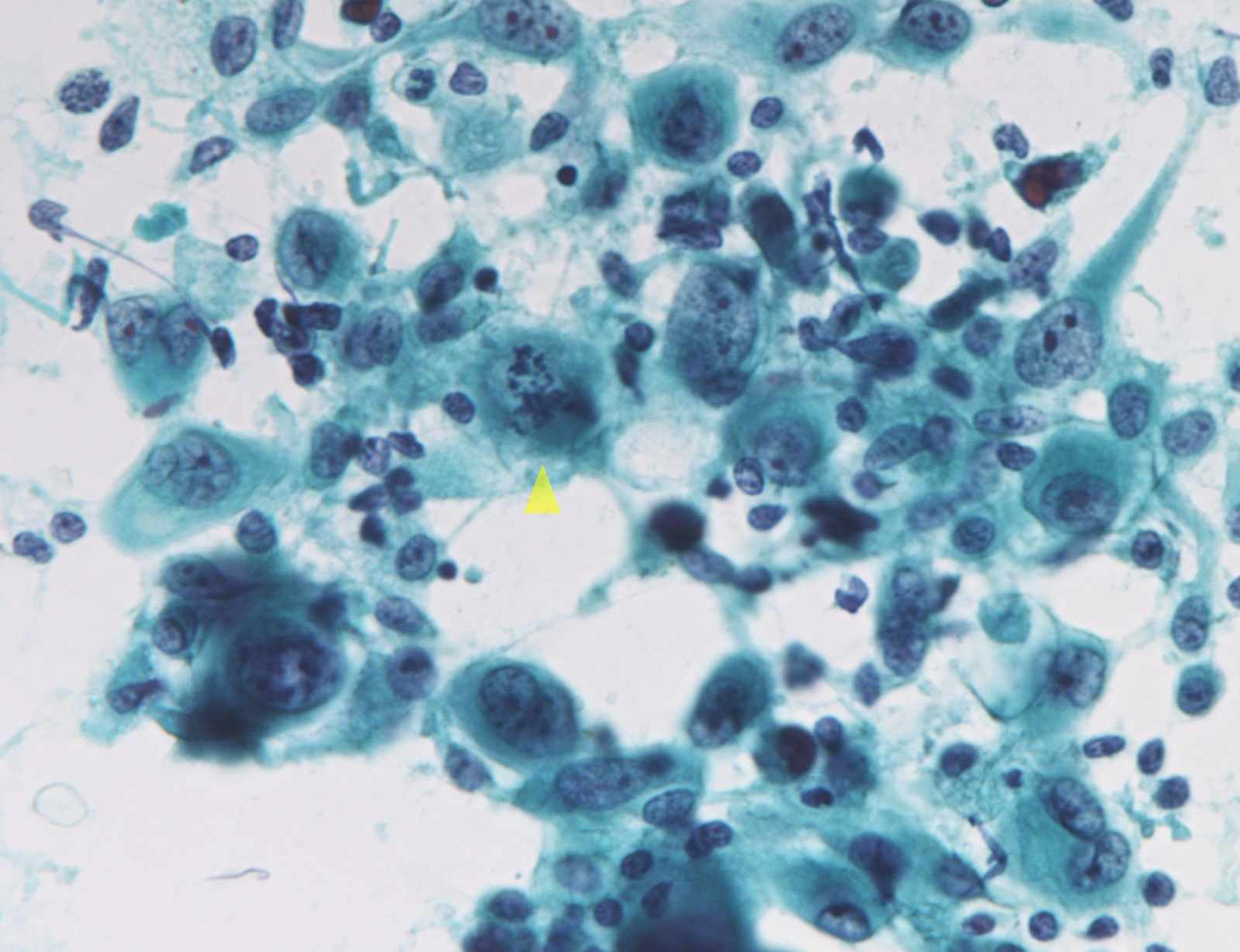
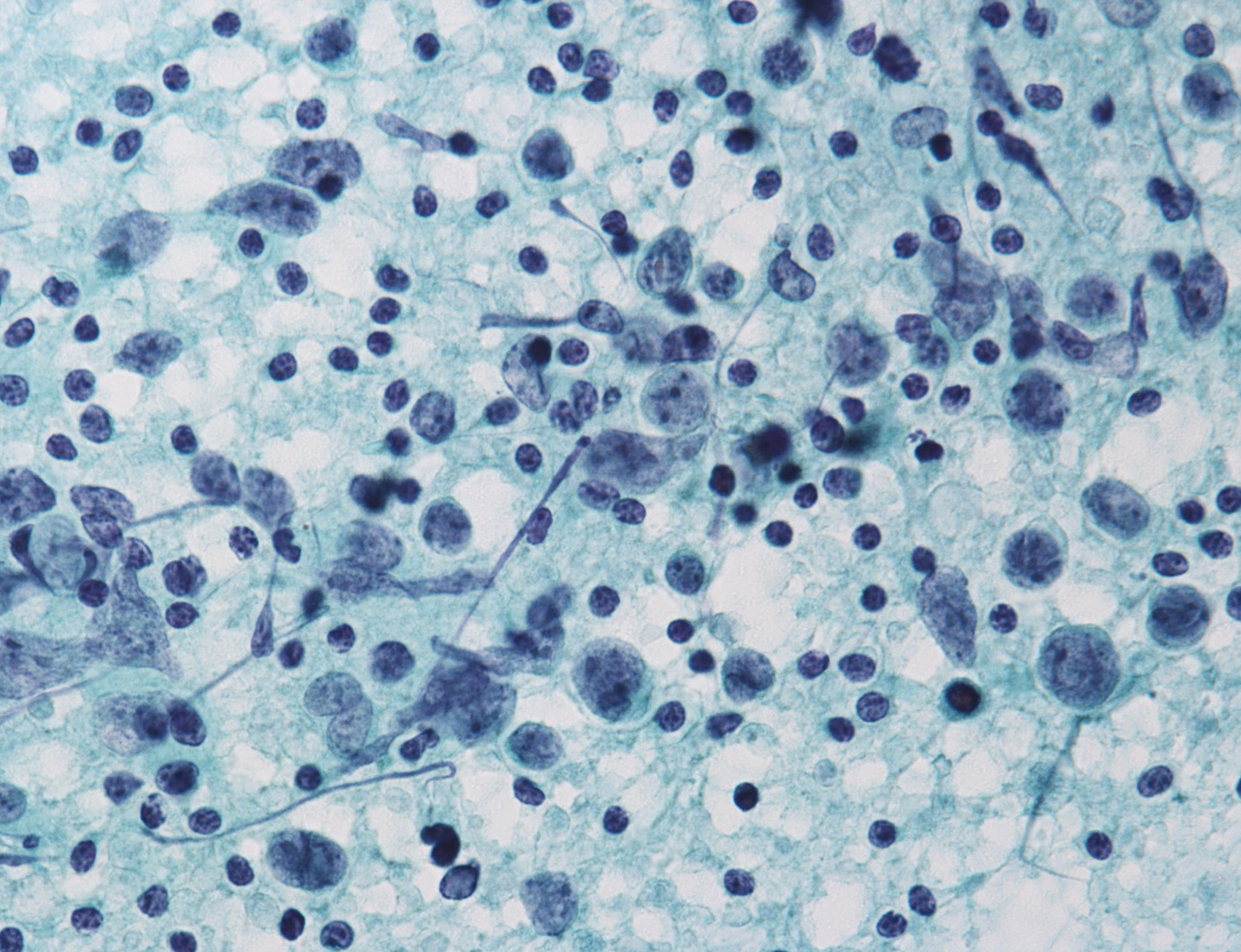
Cytology images
Contributed by Ayana Suzuki, Ph.D.
Unsatisfactory
Benign
Atypia of undetermined significance
Suspicious for malignancy
Malignant
Videos
Algorithmic approach to thyroid FNA
Head & tail of the Bethesda system
Thyroid cytology: approach
Thyroid cytology: cases
Thyroid cytology: ND, benign and FN
Thyroid cytology: malignant, SUS and AUS
Thyroid cytopathology
How to observe thyroid FNA
Additional references
Practice question #1
Practice answer #1
E. Nondiagnostic. When the aspirated material contains only foamy histiocytes and no follicular epithelium or colloid, it is qualified as nondiagnostic; however, in some local reporting systems (e.g., Japanese), these cases are reported as adequate, cyst fluid only, because their malignancy risk is almost the same as that of the benign category and lower than that in the nondiagnostic category. Answer B is incorrect because there are no cells in the photo that meet the adequate and benign criteria. Answers A, C and D are incorrect because there are no atypical cells in the photo.
Comment Here
Reference: Bethesda system diagnostic categories
Comment Here
Reference: Bethesda system diagnostic categories
Practice question #2
Which is the most recommended management for the benign category of The Bethesda System for Reporting Thyroid Cytopathology?
- Follow up
- Molecular testing
- Reaspiration
- Thyroid lobectomy
- Total thyroidectomy
Practice answer #2
A. Follow up. The American Thyroid Association recommends follow up based on the ultrasound pattern for a benign lesion. Answer B is incorrect because molecular testing is typically reserved for indeterminate categories, such as atypia of undetermined significance (AUS) or follicular neoplasm (FN) / suspicious for a follicular neoplasm (SFN), where the risk of malignancy is higher and the cytological findings are ambiguous. Answer C is incorrect because reaspiration is not routinely recommended for benign cytology unless there are new clinical concerns or suspicious ultrasound features. Answer D is incorrect because lobectomy is typically considered for categories with a higher risk of malignancy, not benign lesions. Answer E is incorrect because total thyroidectomy is usually reserved for confirmed malignant cases or when there are other indications, such as large goiters causing compressive symptoms.
Comment Here
Reference: Bethesda system diagnostic categories
Comment Here
Reference: Bethesda system diagnostic categories