Table of Contents
Definition / general | Essential features | Terminology | ICD coding | Epidemiology | Sites | Pathophysiology | Etiology | Clinical features | Diagnosis | Laboratory | Radiology description | Radiology images | Prognostic factors | Case reports | Treatment | Gross description | Gross images | Frozen section description | Frozen section images | Microscopic (histologic) description | Microscopic (histologic) images | Cytology description | Positive stains | Negative stains | Electron microscopy description | Sample pathology report | Differential diagnosis | Additional references | Practice question #1 | Practice answer #1 | Practice question #2 | Practice answer #2Cite this page: Bhardwaj S, Kalir T. Pregnancy luteoma. PathologyOutlines.com website. https://www.pathologyoutlines.com/topic/ovarypregnancyluteoma.html. Accessed August 26th, 2025.
Definition / general
- Pregnancy luteoma falls under the tumor-like lesions category of the WHO 2020 classification of tumors
- Self limited, hyperplastic (nonneoplastic) proliferation of large, luteinized ovarian cells during pregnancy, resulting in a tumor-like mass of the ovary that regresses spontaneously during puerperium
Essential features
- Pregnancy luteomas are nonneoplastic (hyperplastic) proliferations of luteinized cells, occurring, by definition, during pregnancy
- Regress spontaneously after delivery
- Microscopic features include diffuse sheets of round cells with abundant eosinophilic cytoplasm and round nuclei
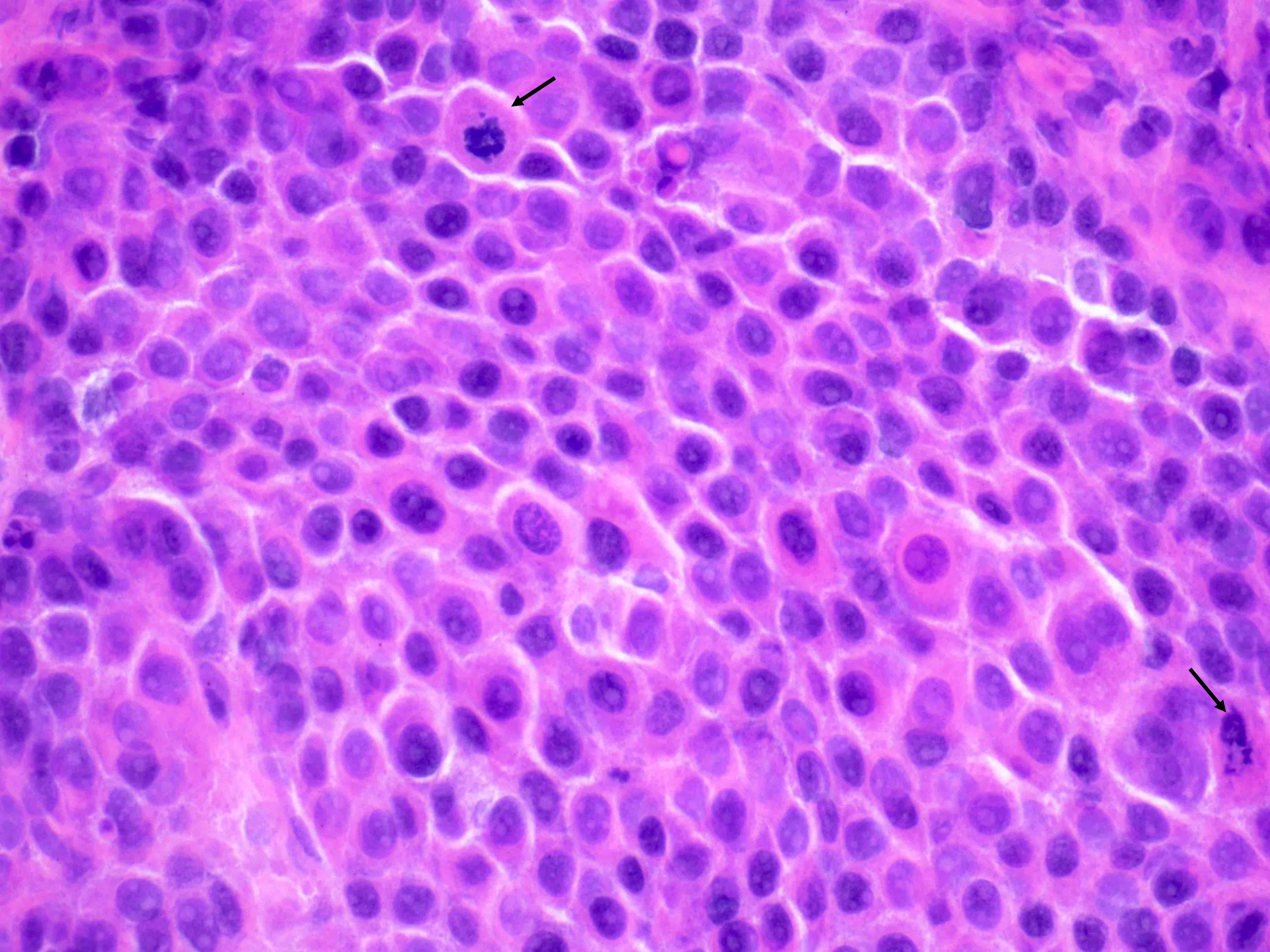
- Some luteomas may show brisk mitoses
- This may be a diagnostic pitfall, especially during intraoperative evaluation
- Presence of uniform bland cells, despite the high mitotic rate and a supportive clinical history, is the key to making a diagnosis in these cases
Terminology
- Luteoma of pregnancy
ICD coding
- ICD-O: 8610/0 - luteoma, NOS
- ICD-11: 2F32.Y & XH40J2 - other specified benign neoplasm of ovary & luteoma, NOS
Epidemiology
- Age group: 15 - 44 years; by definition, during pregnancy (Int J Gynecol Pathol 1993;12:108)
- Commonly seen in mid to late pregnancy (Int J Gynaecol Obstet 2022 Feb 25 [Epub ahead of print])
- 80% of patients are multiparous; luteomas are rare in primiparous women
- 80% of patients are African Americans
- Rarely, may recur in consecutive pregnancies
Sites
- Ovary
Pathophysiology
- Occurs due to nodular hyperplasia of theca interna cells
- Previously, elevated hCG was implicated as a direct causative agent of luteomas, due to the finding of elevated hCG levels in luteoma patients and spontaneous regression after pregnancy
- hCG shares a common alpha subunit with luteinizing hormone (LH) and is thus able to bind to the LH-hCG receptor on the ovarian stromal cells; this causes proliferation and luteinization of ovarian stromal cells
- Factors against hCG being the sole causative agent in luteoma pathogenesis:
- There are other conditions (such as trophoblastic tumors) that are more common, that have elevated hCG levels; luteomas have not been found to be associated with trophoblastic tumors
- Luteomas occur in mid to late pregnancy, while hCG levels are higher in the first trimester
- Therefore, factors other than hCG are also involved in the pathogenesis; exact pathogenesis is unknown (Am J Obstet Gynecol 1969;104:871)
- Pre-existent endocrinopathy, such as stromal hyperthecosis or polycystic ovarian syndrome (PCOS), may predispose to the development of luteomas in some patients
Etiology
- Elevated hCG levels have been implicated in causing hyperplasia of luteinized cells
Clinical features
- Usually asymptomatic, with the mass being incidentally detected at cesarean section or tubal ligation at the end of pregnancy
- Pelvic mass: rarely seen, can rarely cause obstruction of birth canal
- Virilization (Obstet Gynecol 1982;59:105S):
- Seen in 25% of patients, appearing or increasing in the third trimester of pregnancy
- Androgen levels are increased up to 70 times normal
- High androgen levels may be seen in women who do not exhibit virilizing symptoms; this is due to the androgen blunting effect of sex hormone binding globulin (SHBG)
- Virilization symptoms seen with luteomas include:
- Hirsutism
- Deepening of voice
- Acne
- Frontal baldness
- Clitoral hypertrophy, etc.
- Virilization features can be seen in up to 70% of female infants born to women that exhibit virilization features under the influence of androgens, leading to clitoromegaly and labial fusion (Obstet Gynecol 1982;59:105S)
- Androgen levels are high in female infants but lower than maternal levels
- Other uncommon symptoms include:
- Abdominal pain
- Abdominal mass
- Ascites
- Regression of luteomas:
- Regression occurs spontaneously, several weeks after delivery (starts within days after delivery)
- Androgen levels fall to baseline 2 weeks after delivery
- Symptoms of virilization disappear during first 2 weeks after delivery
Diagnosis
- Excisional biopsy, frozen section examination
Laboratory
- Elevated hCG levels
- Elevated androgen levels
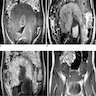
Radiology description
- Ultrasound and MRI show a predominantly solid, heterogenous mass
Prognostic factors
- All pregnancy luteomas invariably regress spontaneously after pregnancy
Case reports
- 28 year old woman, gravida 2, para 1 (G2P1), with large ovarian mass causing hirsutism identified intraoperatively (Case Rep Obstet Gynecol 2021;2021:6695117)
- 30 year old woman with pregnancy luteoma associated with ectopic pregnancy (J Reprod Infertil 2017;18:333)
- 33 year old woman with enlarged ovaries discovered incidentally during cesarean section (Int J Appl Basic Med Res 2016;6:282)
Treatment
- Treatment is usually not necessary as these are spontaneously resolving lesions
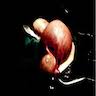
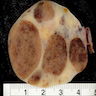
Gross description
- Mostly unilateral; bilateral in 33% of cases (Am J Surg Pathol 2014;38:239)
- Multiple lesions are seen in 50% of cases
- Usually measure 5 - 10 cm but can be smaller or rarely larger, up to 20 cm
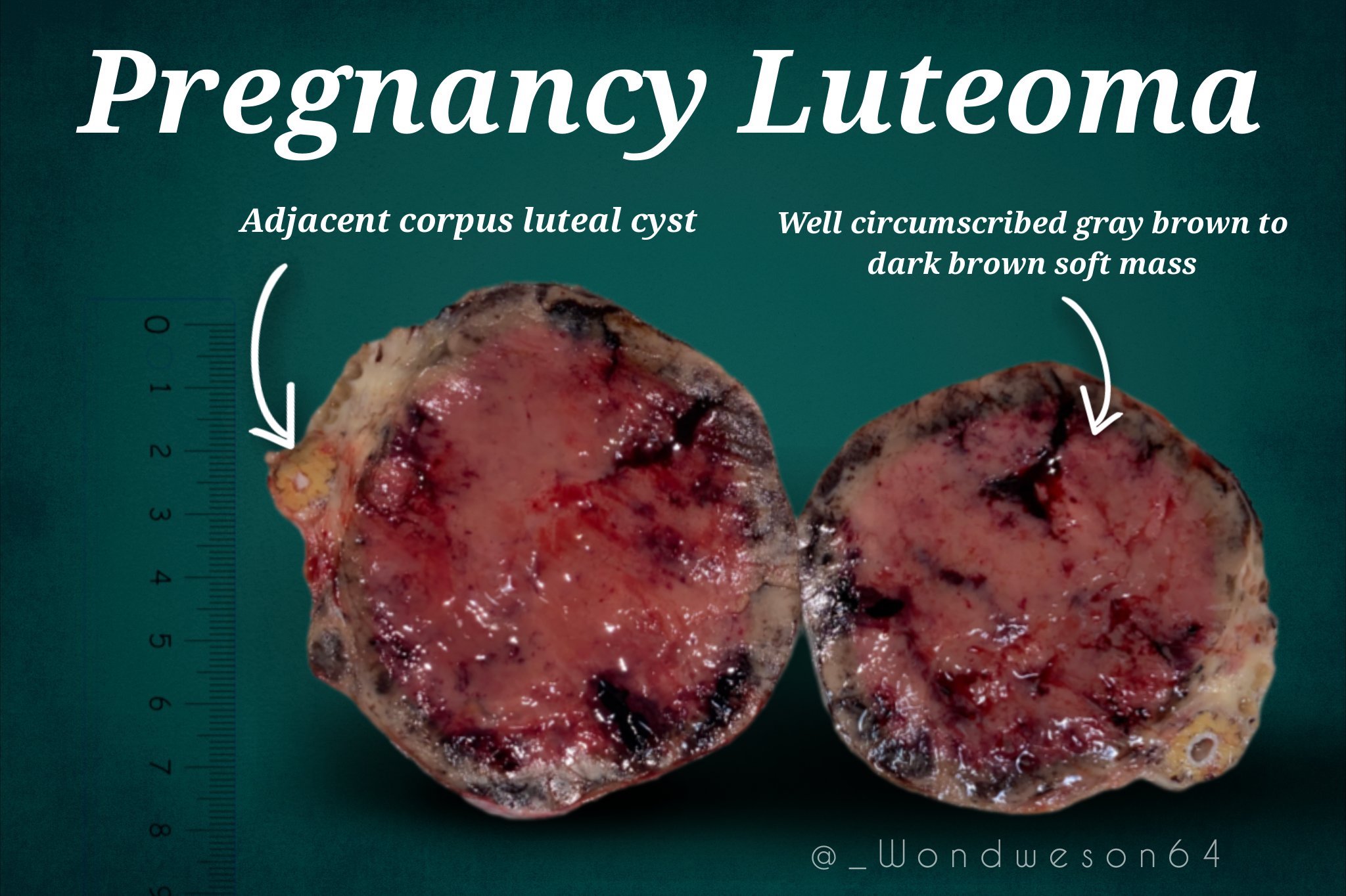
- Cut surface:
- Well circumscribed, multinodular or multiple lesions, brown with focal hemorrhage, without areas of necrosis
- When necrosis is seen (rarely), it is central and due to infarction
Gross images
Frozen section description
- Similar to permanent sections, frozen sections show round to oval cells with abundant eosinophilic cytoplasm, arranged in diffuse sheets
- Caution is advised in interpreting mitoses
- Historically, some luteomas may show brisk mitoses misleading a pathologist into making a diagnosis of malignancy on intraoperative evaluation
- Even with brisk mitoses, cells are uniform and stroma is rarely prominent (Am J Surg Pathol 2014;38:239)
Frozen section images
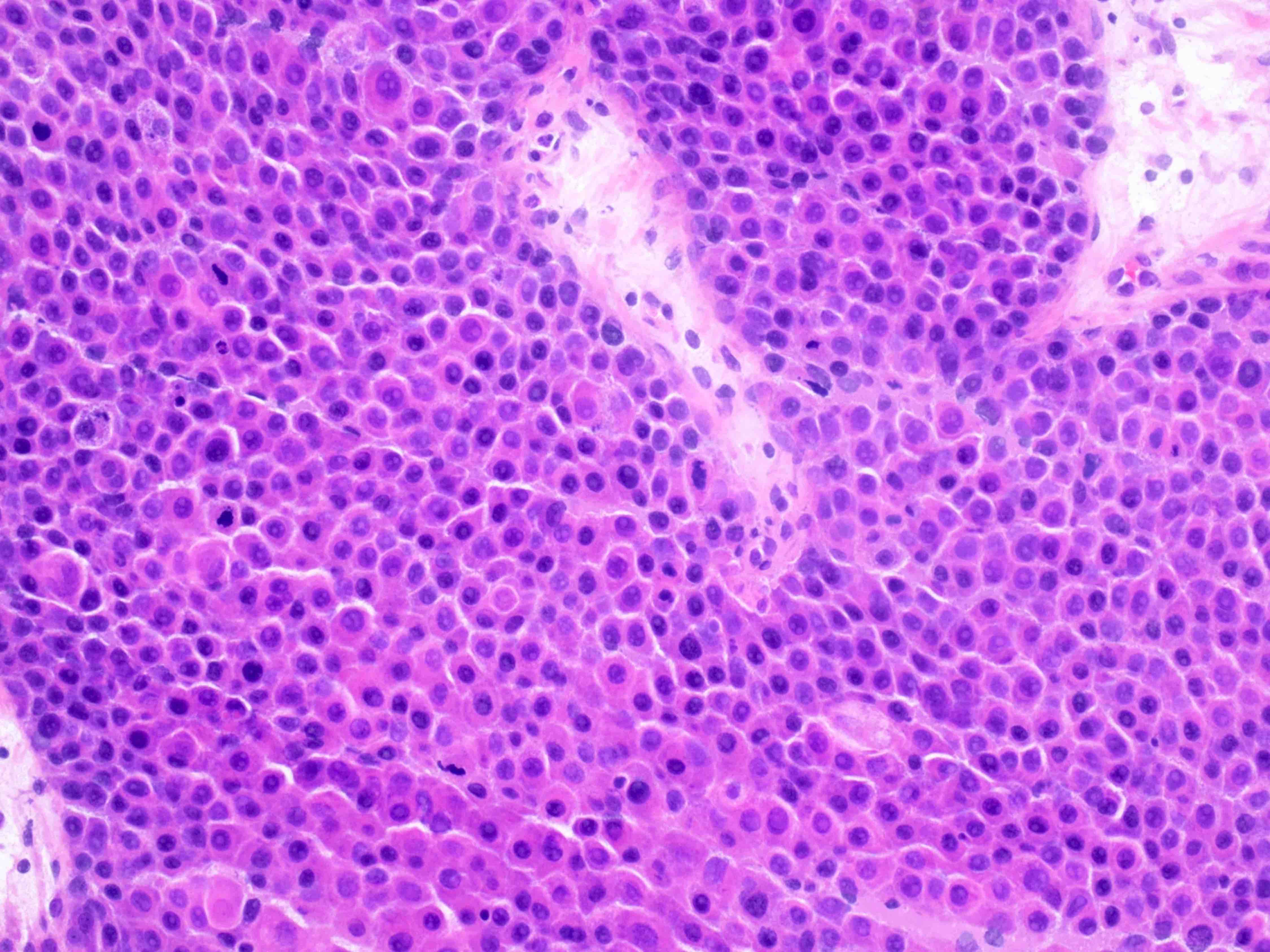
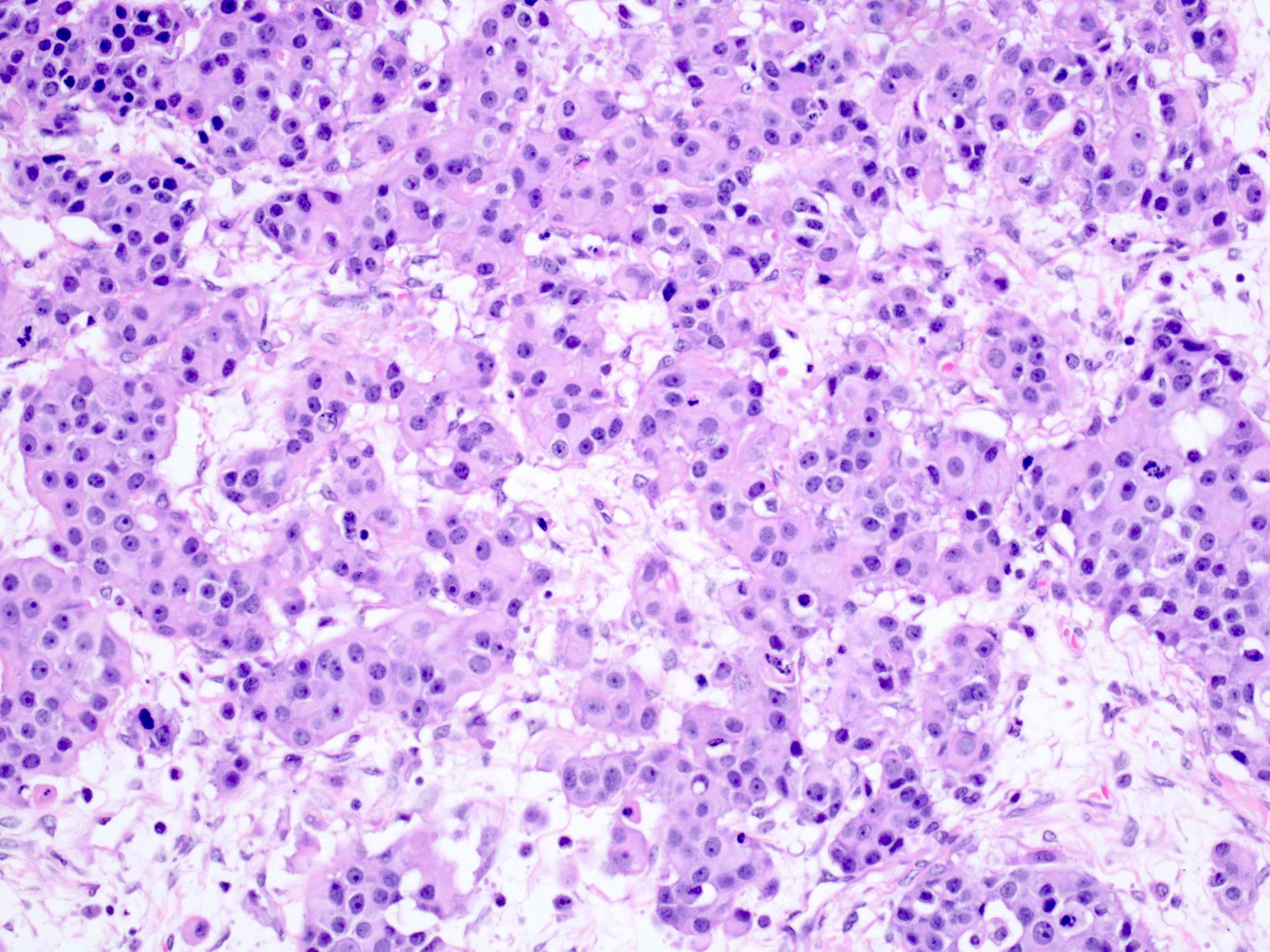
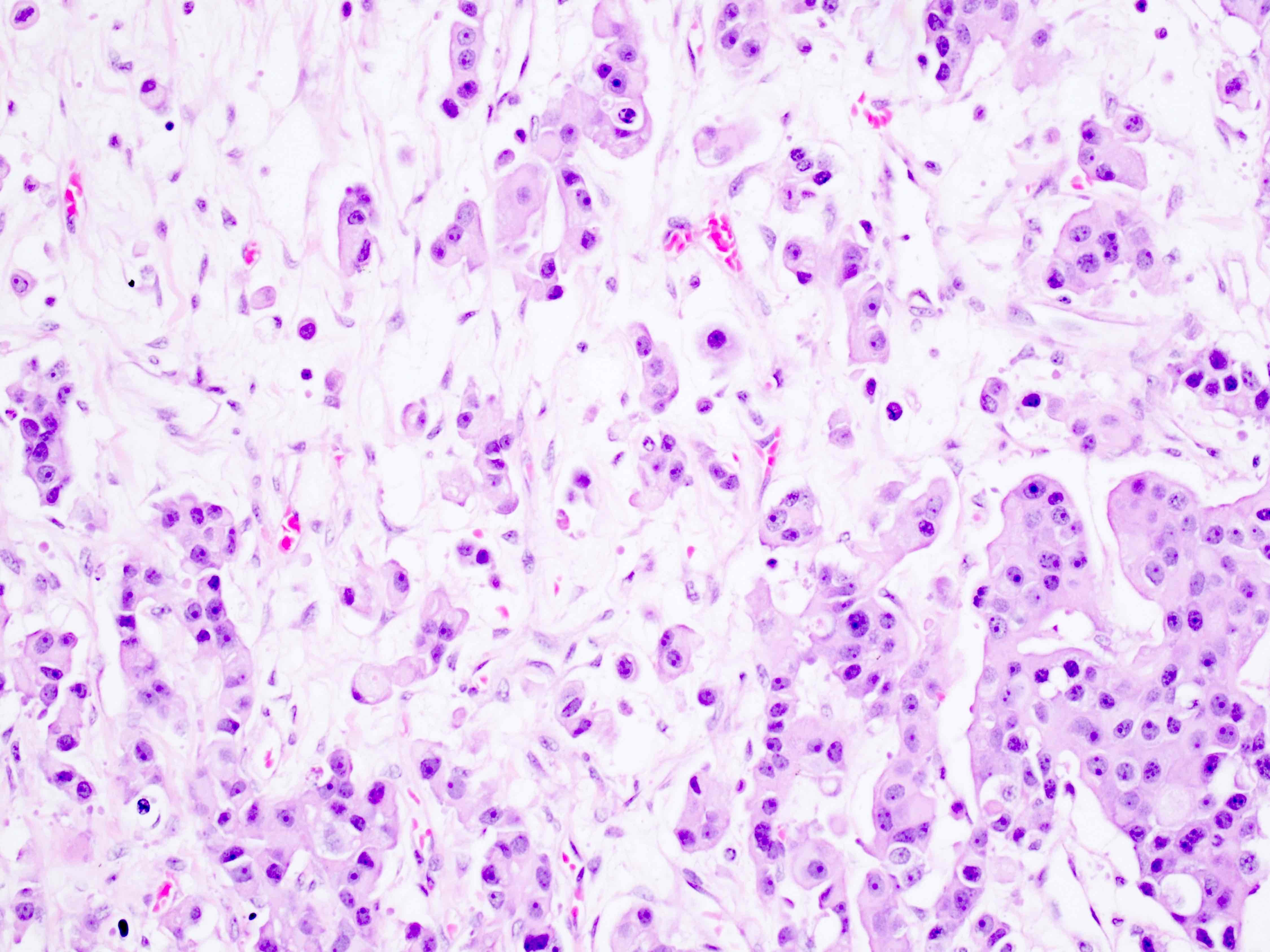
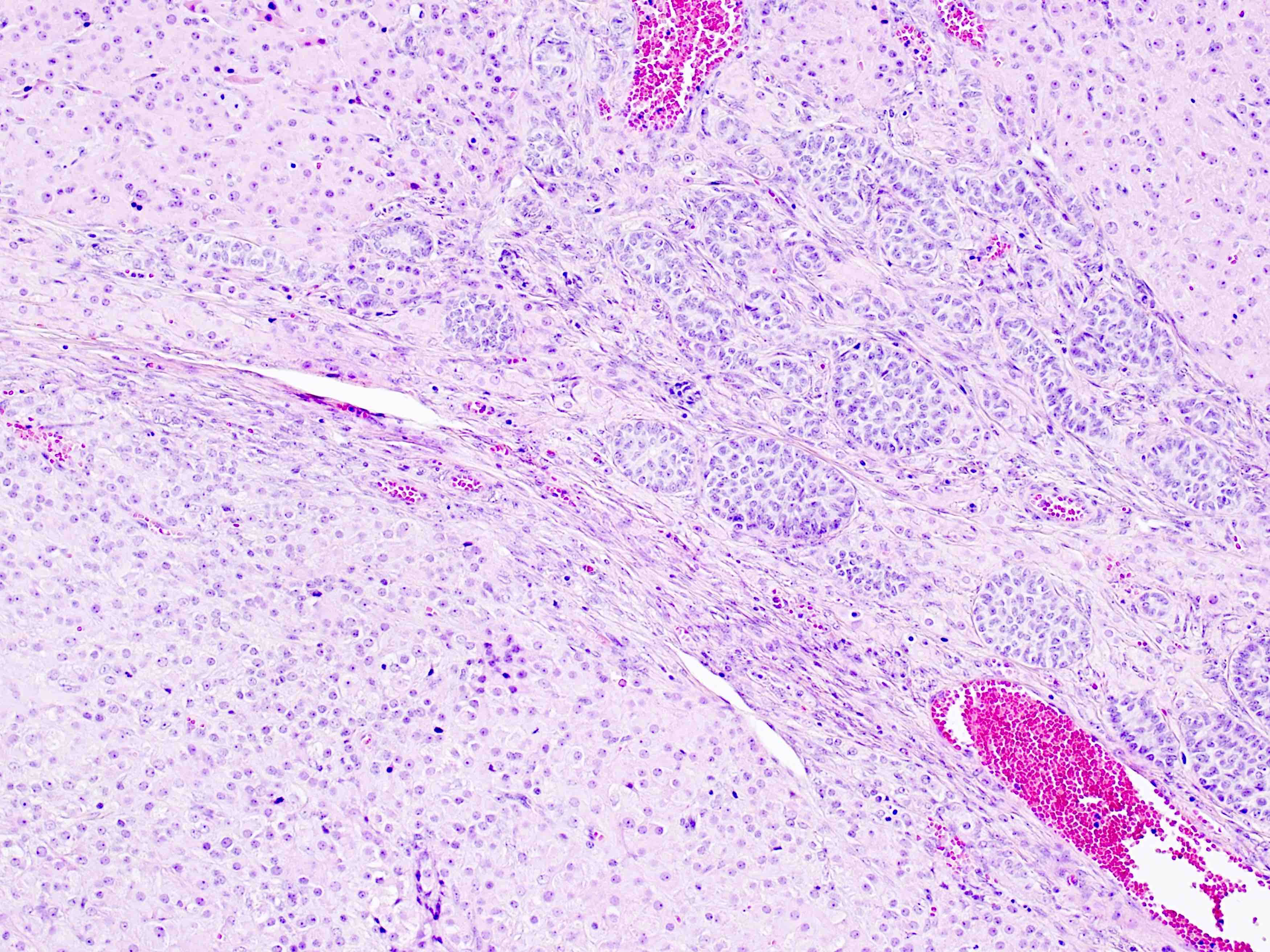
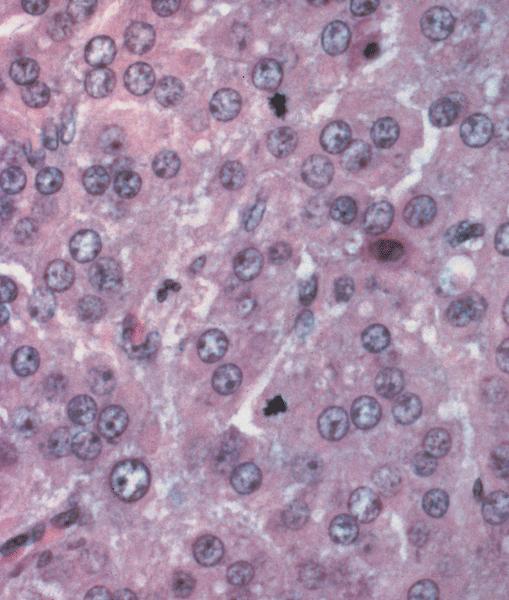
Microscopic (histologic) description
- Arrangement:
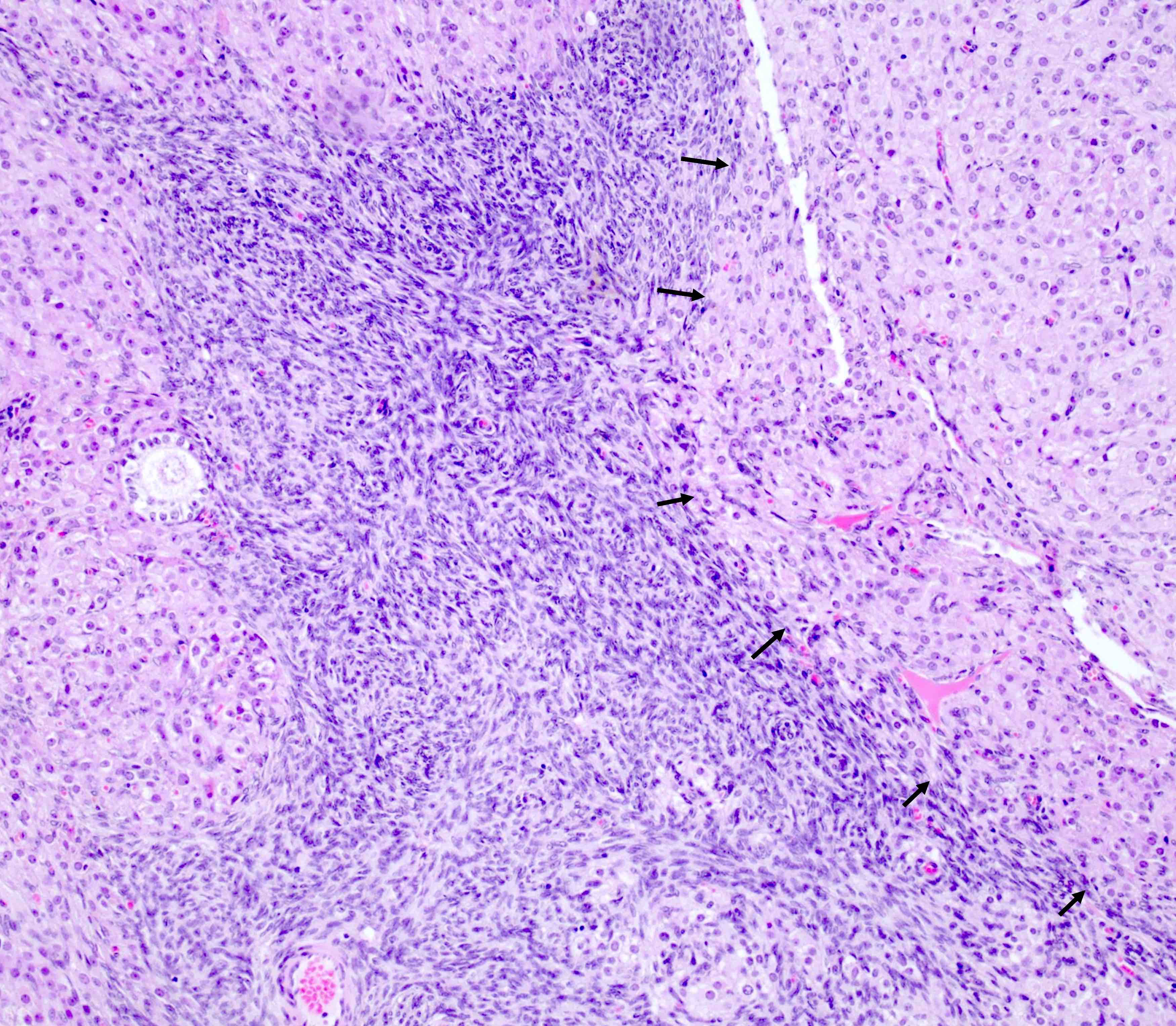
- Lesion is well circumscribed; even when there are multiple nodules, they are sharply circumscribed from the stroma
- Cells are arranged in sheets
- Occasionally, stromal edema may result in separation of cell clusters giving rise to a vaguely nested appearance (Am J Surg Pathol 2014;38:239)
- There may be follicle-like spaces that give rise to a thyroid-like or (when smaller), acinus-like appearance
- Sometimes, a vague reticular or nested pattern may be seen (Am J Surg Pathol 2014;38:239)
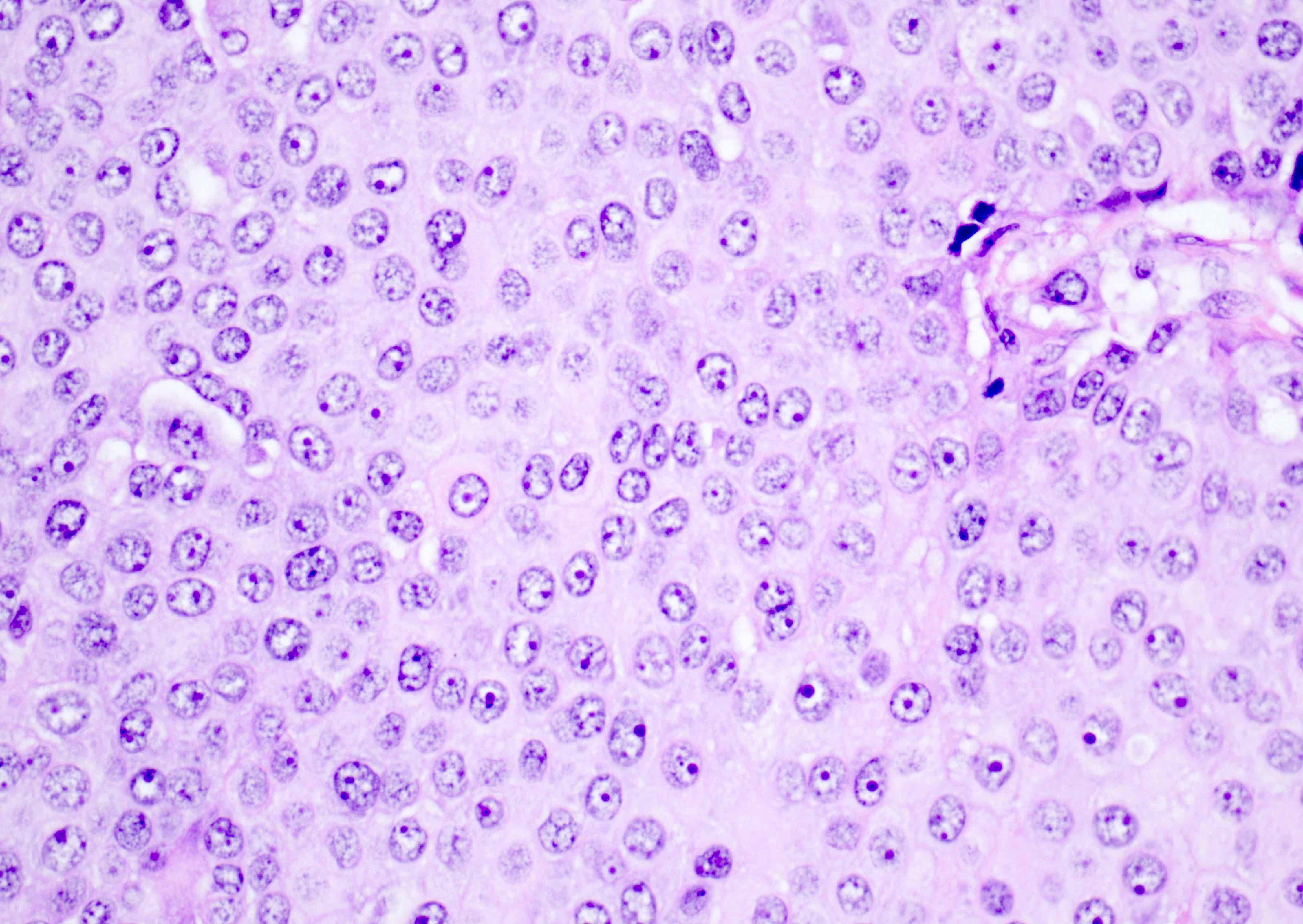
- Cells are round to oval with abundant eosinophilic cytoplasm, round nuclei with prominent nucleoli
- Sometimes, cells may have pale, frothy cytoplasm instead of eosinophilic cytoplasm; nuclei are uniform (no nuclear pleomorphism)
- Mitoses: mitoses are infrequent; in some cases, brisk mitoses can be seen, as high as 2 - 3/10 high power fields (Am J Surg Pathol 2014;38:239)
- Necrosis: not usually seen; can be seen in very large tumors, in a central location, due to infarction of the tumor as it outgrows its blood supply
- Stroma: usually scant; some cases may show stromal edema or fibrosis giving rise to a vaguely nested arrangement of tumor cells
- Additional features: some authors have described the presence of granulosa cell proliferations of pregnancy in the ovary uninvolved by pregnancy luteoma (Am J Surg Pathol 2014;38:239)
- Granulosa cell proliferations of pregnancy may be observed in the ovary involved by luteoma
Microscopic (histologic) images
Contributed by Swati Bhardwaj, M.B.B.S., M.D., Tamara Kalir, M.D., Ph.D. and AFIP images
Cytology description
- Moderate to large cells with abundant eosinophilic cytoplasm and uniform, round nuclei
Positive stains
Negative stains
Electron microscopy description
- Electron microscopy shows features of steroid hormone producing cells (Hum Pathol 1976;7:205, Pathol Res Pract 1999;195:859):
- Lipid droplets
- Mitochondria with tubular cristae
- Abundant smooth endoplasmic reticulum
- Dispersed golgi apparatus
- No crystalloid lattice structures are seen
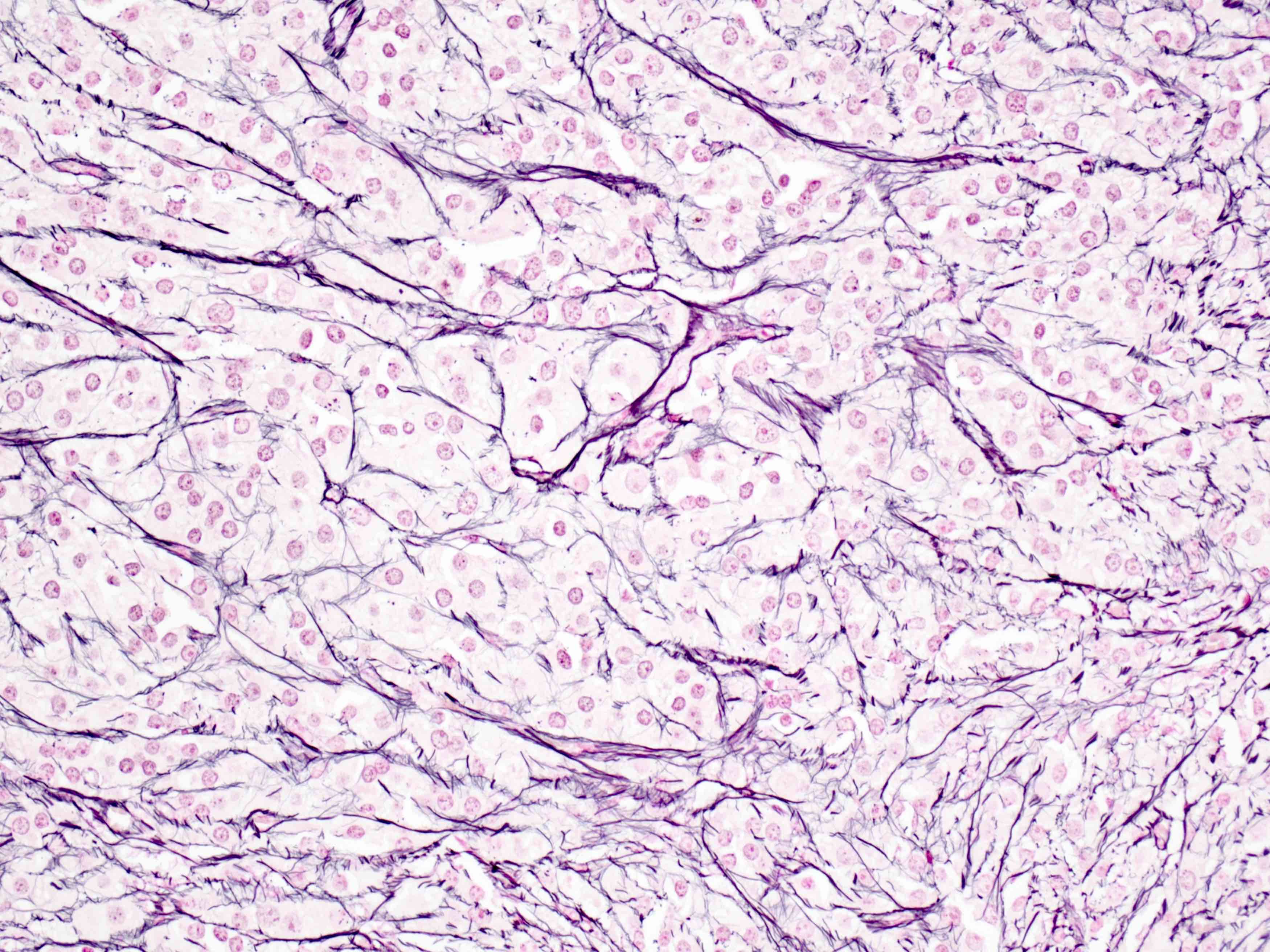
Sample pathology report
- Ovary, oophorectomy specimen:
- Pregnancy luteoma (see comment)
- Comment: Reticulin staining revealed reticulin fibers encircling groups of luteinized cells, consistent with the diagnosis.
- Microscopic description: There are sheets of uniform, round to oval cells with abundant eosinophilic cytoplasm with round nuclei. Background stromal luteosis is seen.
- Ovary, oophorectomy specimen:
- Pregnancy luteoma (see comment)
- Comment: Mitotic activity is brisk but attributed to the hormonal milieu of pregnancy.
- Microscopic description: There are sheets of uniform, round to oval cells with abundant eosinophilic cytoplasm with round nuclei. 5 - 6 mitoses are seen per 10 high power fields. Brisk mitotic activity has been described in pregnancy luteomas. The presence of bland and uniform cells is compatible with a luteoma, despite the brisk mitoses.
Differential diagnosis
- Steroid cell tumor:
- Rarely bilateral
- Grossly, yellow in color
- Cells are smaller than luteoma cells (cells of pregnancy luteoma are 1.5 - 2 times larger than the cells of steroid cell tumor)
- Eosinophilic cells may be accompanied by pale staining lipid rich cells
- Follicle-like spaces are rarely seen
- Distinction between steroid cell tumor and luteomas is important; a subset of steroid cell tumors are clinically malignant
- Dense reticulum pattern and the presence of lipochrome pigment are other features favoring a diagnosis of steroid cell tumor
- In Leydig cell tumors, a hilar location and the presence of Reinke crystals are helpful
- Luteinized thecoma:
- Shows at least focal classical thecoma cells with oval to round cells with spindled nuclei
- Luteinized granulosa cell tumor:
- Shows at least focal areas of granulosa cell tumor with classical morphology
- Can retain evidence of epithelial differentiation
- Even when they are luteinized, adult granulosa cell tumor do not show luteinization to the extent seen in pregnancy luteomas
- FOXL2 402C → G (C134W) somatic mutation is highly sensitive and specific for diagnosis of adult granulosa cell tumor (N Engl J Med 2009;360:2719, PLoS One 2009;4:e7988, Am J Surg Pathol 2011;35:484)
- Malignant melanoma:
- Shows much greater atypia and higher mitotic rate than that seen in pregnancy luteomas
- IHC is positive for MelanA, HMB45, S100, SOX10 (Case Rep Oncol Med 2020;2020:7206786)
Additional references
Practice question #1
Practice answer #1
D. Reticulin staining fibers enclosing groups of cells, rather than individual cells
Comment Here
Reference: Pregnancy luteoma
Comment Here
Reference: Pregnancy luteoma
Practice question #2
Practice answer #2
D. Lipid rich cells and dense reticulum pattern. Lipid rich cells and dense reticulum patterns favor a diagnosis of steroid cell tumors over pregnancy luteoma. Other features are commonly seen in pregnancy luteomas.
Comment Here
Reference: Pregnancy luteoma
Comment Here
Reference: Pregnancy luteoma