Table of Contents
Definition / general | Essential features | Terminology | ICD coding | Epidemiology | Sites | Pathophysiology | Etiology | Clinical features | Diagnosis | Laboratory | Prognostic factors | Case reports | Treatment | Clinical images | Gross description | Gross images | Microscopic (histologic) description | Microscopic (histologic) images | Positive stains | Molecular / cytogenetics description | Sample pathology report | Differential diagnosis | Additional references | Practice question #1 | Practice answer #1 | Practice question #2 | Practice answer #2Cite this page: Martinez Ciarpaglini C. Well differentiated neuroendocrine tumor. PathologyOutlines.com website. https://www.pathologyoutlines.com/topic/colontumorneuroendocrinetumor.html. Accessed September 16th, 2025.
Definition / general
- Well differentiated tumors demonstrating morphological and immunohistochemical neuroendocrine differentiation
Essential features
- May be indolent and incidental or present with widespread metastases and have aggressive clinical course
- The term neuroendocrine tumor (NET) comprises WHO G1, G2 and rare G3 lesions (depending on the proliferative activity)
Terminology
- Previously termed carcinoid tumor (Arch Pathol Lab Med 2016;140:437)
- According to the 2017 WHO classification, the term MiNEN (mixed neuroendocrine neoplasm) comprises mixed neuroendocrine and nonneuroendocrine proliferations (World J Gastrointest Oncol 2020;12:791)
- The nonneuroendocrine component may be adenocarcinoma, squamous cell carcinoma, among others
- Each component must be ≥ 30% to fall into the category of MiNEN
ICD coding
- ICD-10: D3A.8 - other benign neuroendocrine tumors
Epidemiology
- Incidence of colorectal neuroendocrine tumors has been continuously increasing over the past few decades (incidence: 1.04 per 100,000 persons) (JAMA Oncol 2017;3:1335)
- Colorectal neuroendocrine tumors comprise roughly 30% of neuroendocrine tumors in the digestive system (Cancer 2003;97:934)
- According to the WHO classification (5th edition), there is a slight male predominance and the median age of presentation is 56 years for rectal tumors and 65 years for colonic tumors
Sites
- In the large intestine (excluding the appendix), the most frequent location is the rectum (34% of all gastrointestinal neuroendocrine tumors) (Ann Oncol 2013;24:3040)
- In the colon, neuroendocrine tumors are more frequent in the cecum (69.6%), followed by sigmoid (13.0%), ascending colon (13.0%) and transverse colon (4.3%) (Arq Gastroenterol 2009;46:288)
- Colon proper is the least common site for intestinal well differentiated neuroendocrine tumors; around 7.5% of all neuroendocrine tumors are of colonic origin (Surg Oncol Clin N Am 2020;29:267)
Pathophysiology
- Colonic NETs arise from Kulchitsky cells or enterochromaffin cells located within the crypts of Lieburkuhn of the colon (World J Gastrointest Oncol 2020;12:791)
- Rare cases are familial (Tech Coloproctol 2006;10:143)
Etiology
- Etiology remains unknown
Clinical features
- Patients with colonic neuroendocrine tumors may present with pain, bleeding, altered bowel habits, weight loss, anorexia or even bowel obstruction (Best Pract Res Clin Gastroenterol 2012;26:775)
- Carcinoid syndrome occurs in patients with liver metastasis
- Prognosis largely depends on the grade and stage of the tumor; median overall survival ranges from 22 months in G1 - G2 tumors to 12 months in G3 tumors according to the WHO classification (5th edition)
- Increased risk of lymph node metastasis according to size: around 1% in tumors < 10 mm, 23% in tumors from 10 - 20 mm and 50% in tumors > 20 mm (J Korean Soc Coloproctol 2011;27:17)
- G3 colorectal neuroendocrine tumors show an intermediate behavior between G2 neuroendocrine tumors and neuroendocrine carcinomas (Cancer 2014;120:2814)
Diagnosis
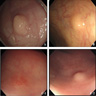
- > 50% are discovered during routine colonoscopy for colorectal cancer screening (Best Pract Res Clin Gastroenterol 2012;26:775)
- Rectal tumors may present with symptoms such as bleeding or change in bowel habits (Surg Oncol Clin N Am 2020;29:267)
Laboratory
- Since few colorectal neuroendocrine tumors produce serotonin, routine analysis of plasma or urinary 5-hydroxyindoleacetic acid (5-HIAA) is not useful (Neuroendocrinology 2012;95:88, PLoS One 2017;12:e0188876)
- Serum chromogranin A (CgA) is the most common biomarker used to assess the bulk of disease and monitor treatment; it is elevated in both functioning and nonfunctioning neuroendocrine tumors (Neuroendocrinology 2012;95:88, World J Gastrointest Oncol 2020;12:791)
Prognostic factors
- Rectal neuroendocrine tumors are commonly small and generally low to intermediate grade (G1 or G2), whereas colonic tumors are often aggressive, poorly differentiated and higher grade (G3) (Neuroendocrinology 2016;103:139)
- Colorectal tumors < 10 mm have a low rate (1.95%) of lymph node metastasis (J Korean Soc Coloproctol 2011;27:17, Gut 2007;56:863)
- Lymphatic invasion and WHO grade are independent predictive factors of lymph node metastasis (Front Oncol 2016;6:173, Sci Rep 2019;9:2404, Ann Surg Oncol 2020;27:855)
- Number of positive locoregional lymph nodes is an independent prognostic factor of survival (Ann Surg Oncol 2019;26:2028)
- HES77 expression is also linked to worse prognosis (Anticancer Res 2015;35:3767)
Case reports
- 47 year old man with a polypoid mass in the right colic flexure (Pan Afr Med J 2021;40:243)
- 57 year old man with perforated colonic tumor with liver metastasis (Am J Case Rep 2019;20:920)
- 64 year old man presented with carcinoid syndrome (Ann Ital Chir 2017;6:S2239253X17027694)
- 85 year old woman presented with mixed neuroendocrine and nonneuroendocrine neoplasms (Int J Surg Case Rep 2020;76:125)
Treatment
- For lesions < 10 mm with no involvement of muscularis propria, transanal endoscopic microsurgery is usually sufficient (J Laparoendosc Adv Surg Tech A 2006;16:435, Neuroendocrinology 2016;103:139)
- Larger or high risk tumors may require surgery (Arch Surg 2008;143:471)
- Positive margins may not increase risk of recurrence (Am Surg 2011;77:198)
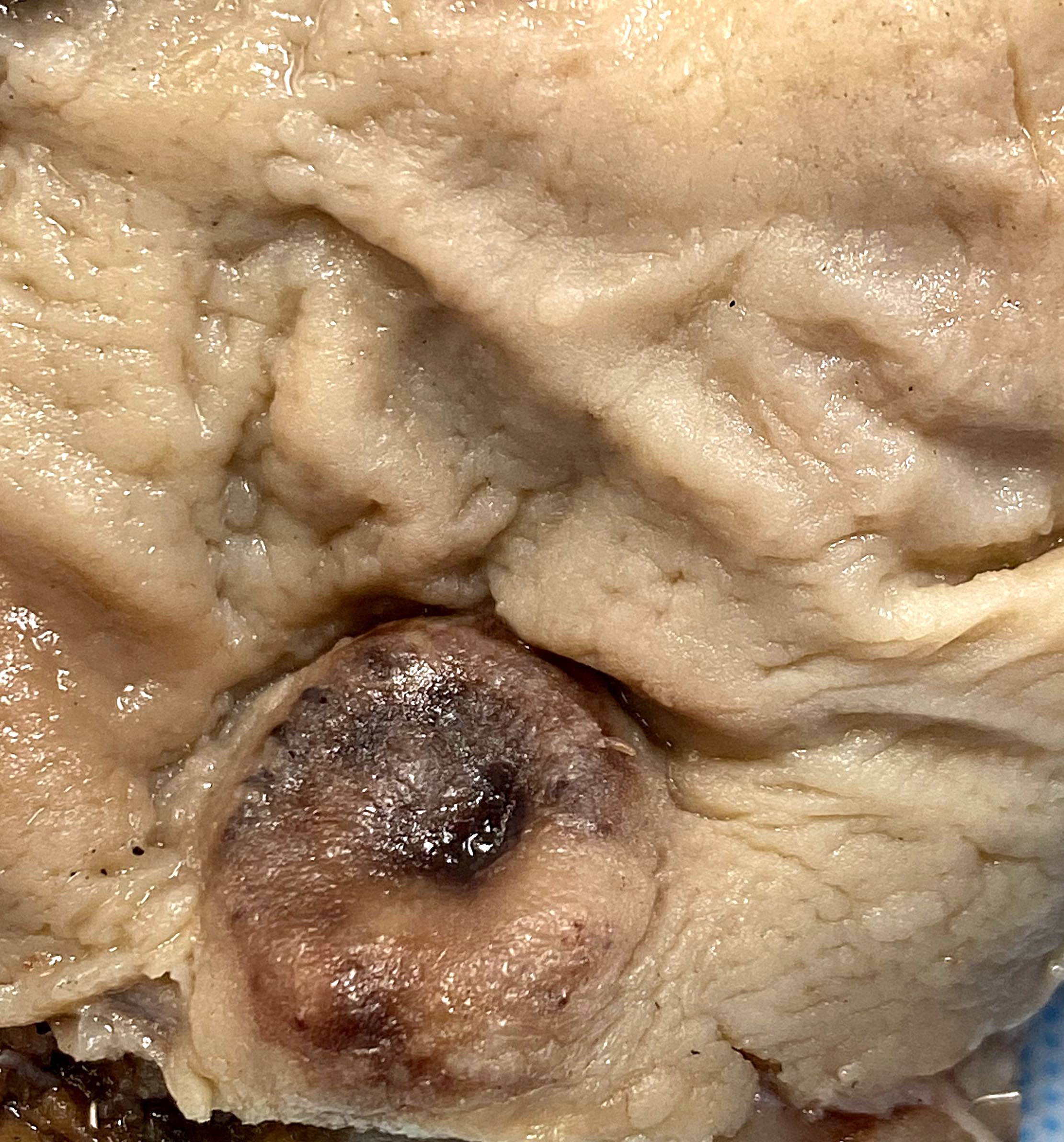
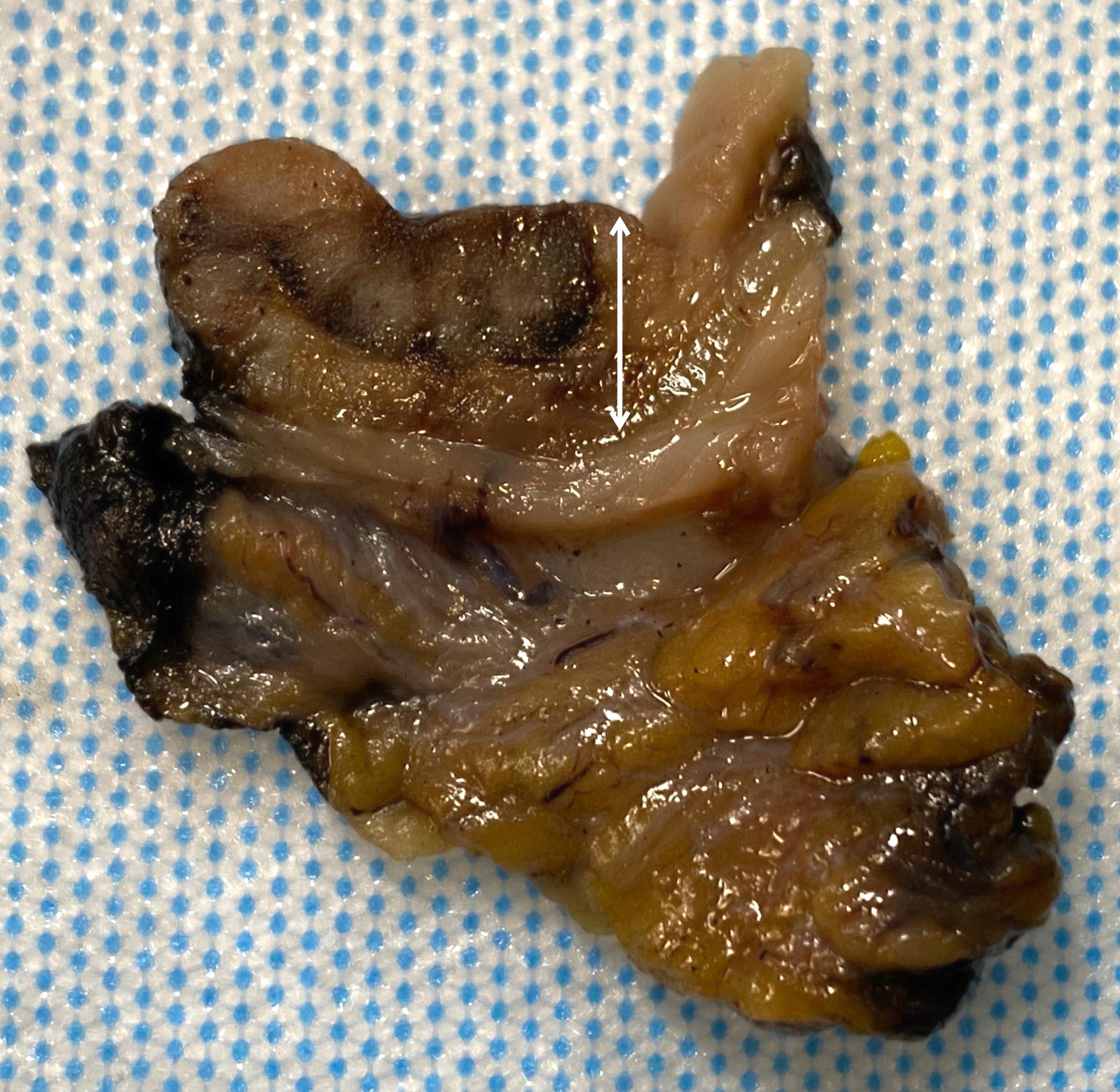
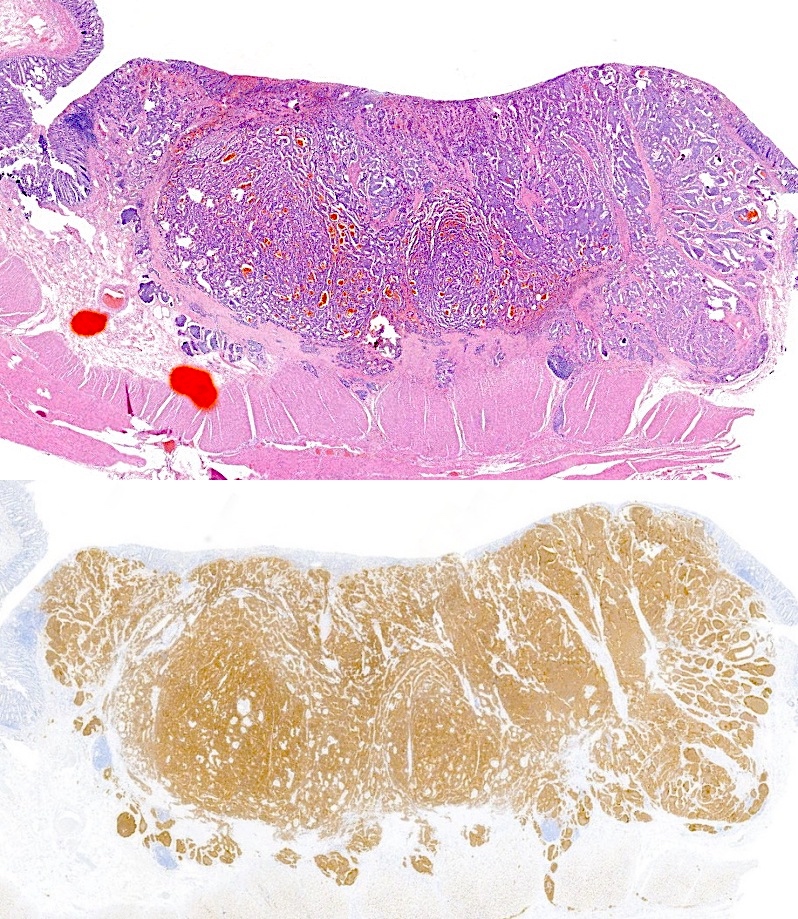
Gross description
- Most colorectal neuroendocrine tumors appear as yellow or pale, polypoid or flat, doughnut shaped lesions or submucosal nodules (Neuroendocrinology 2016;103:139)
Gross images
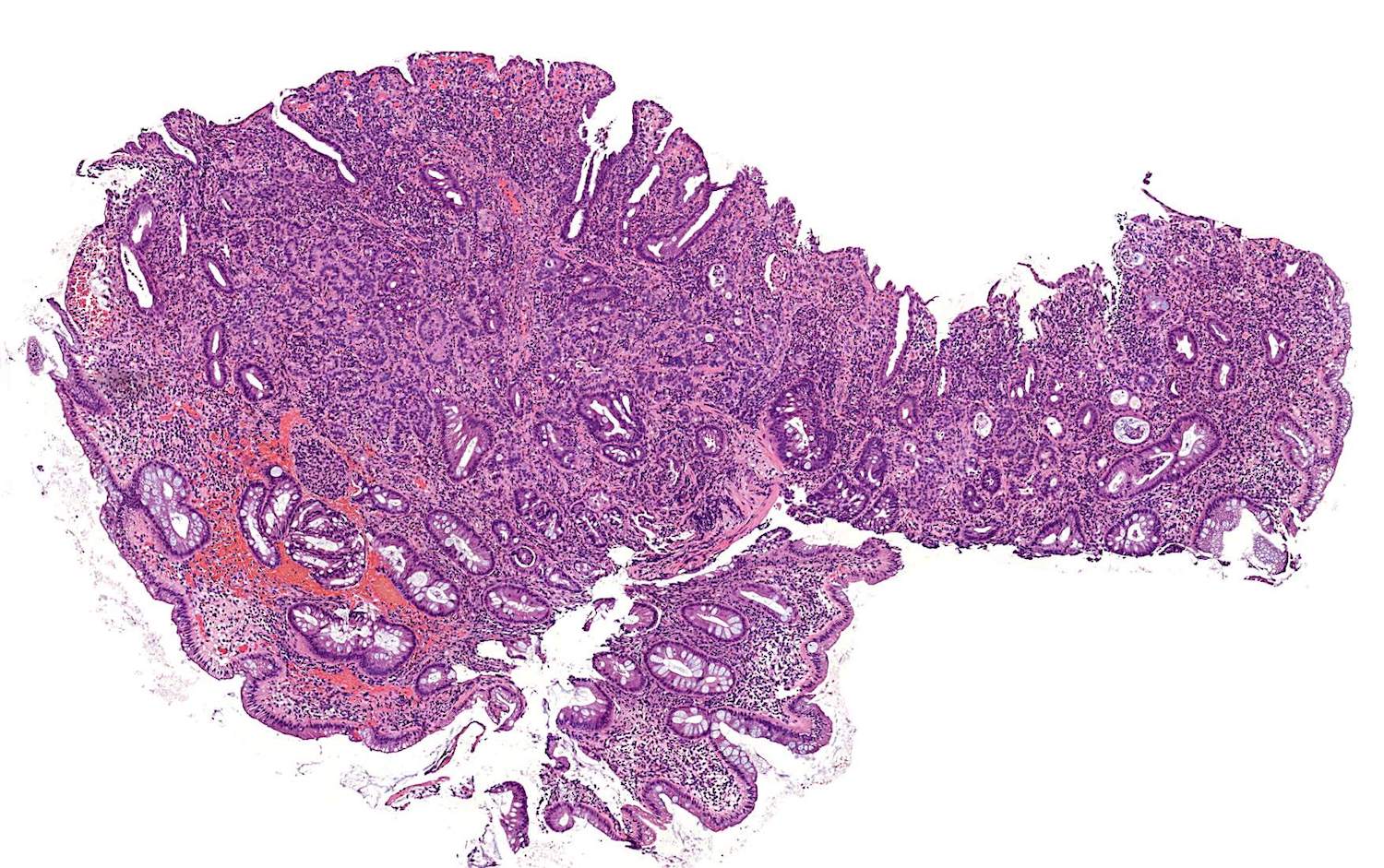
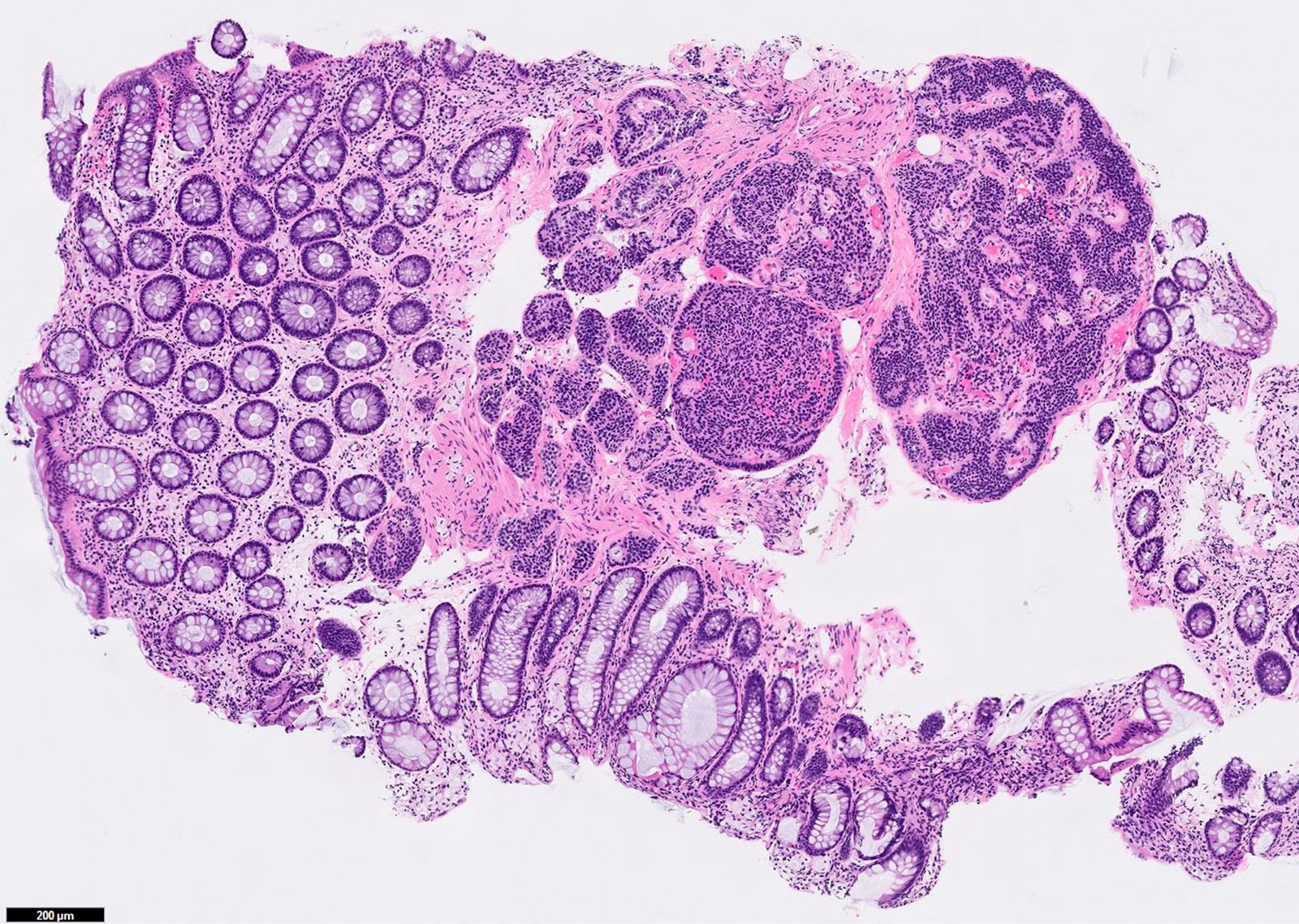
Microscopic (histologic) description
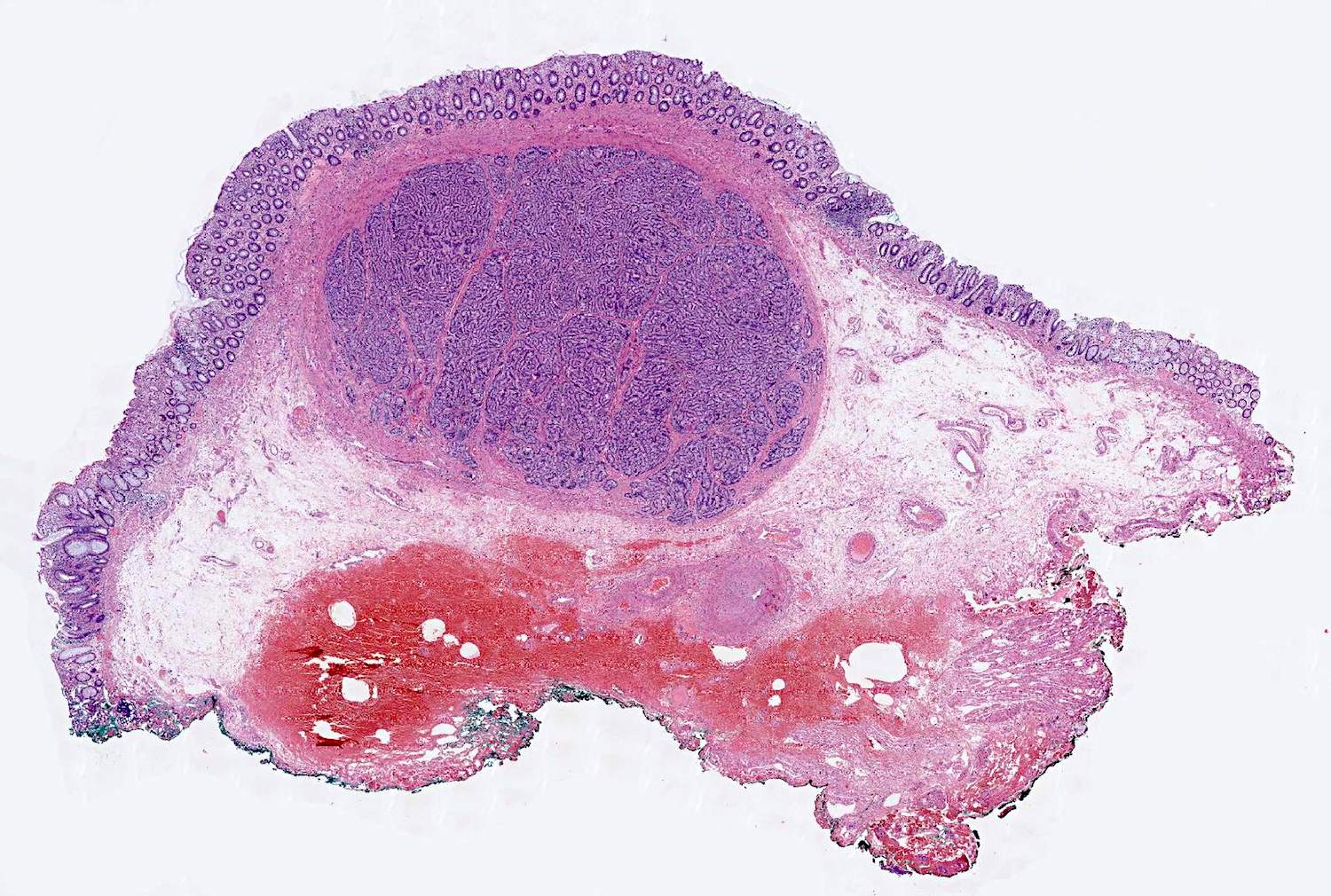
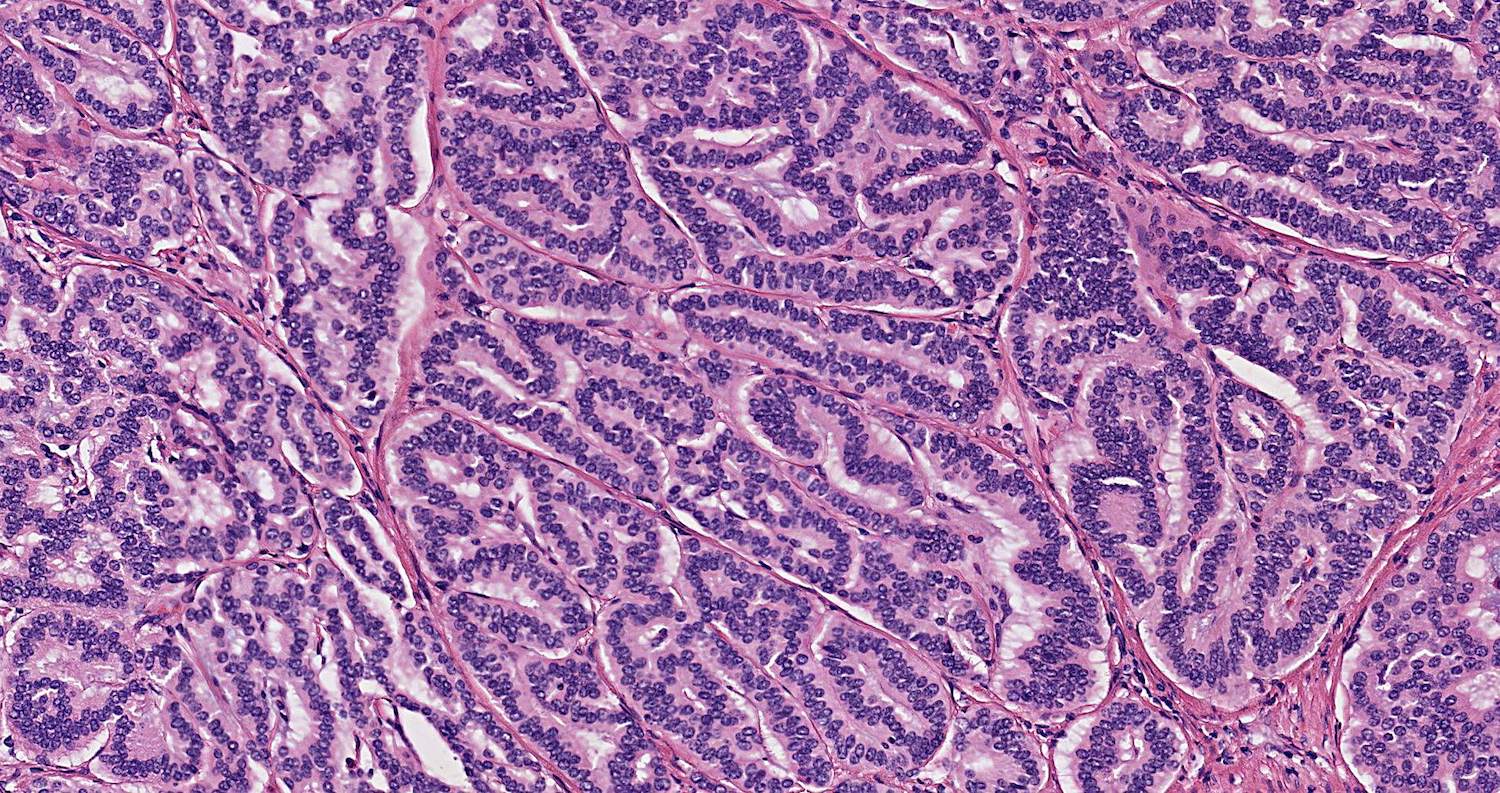
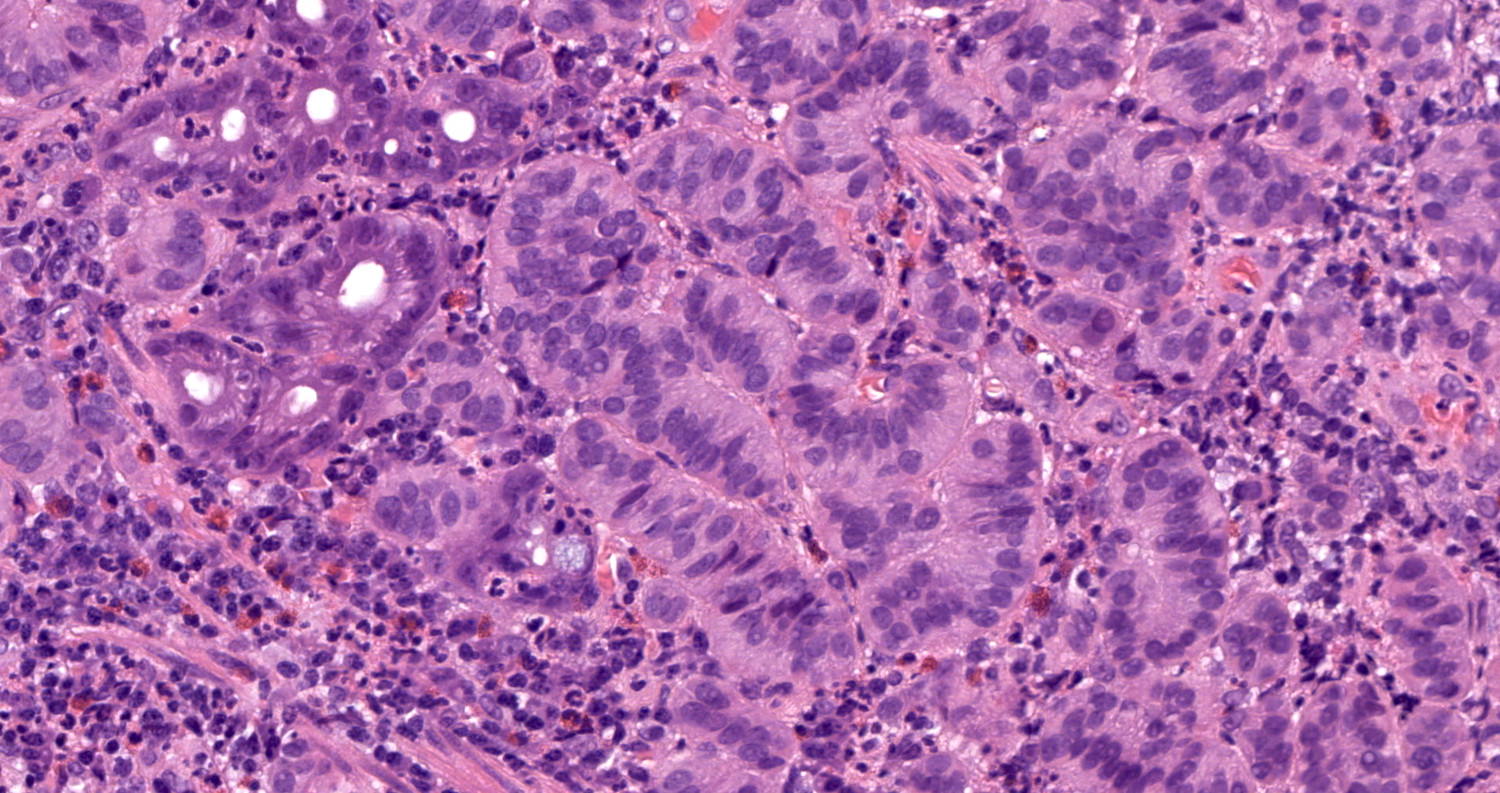
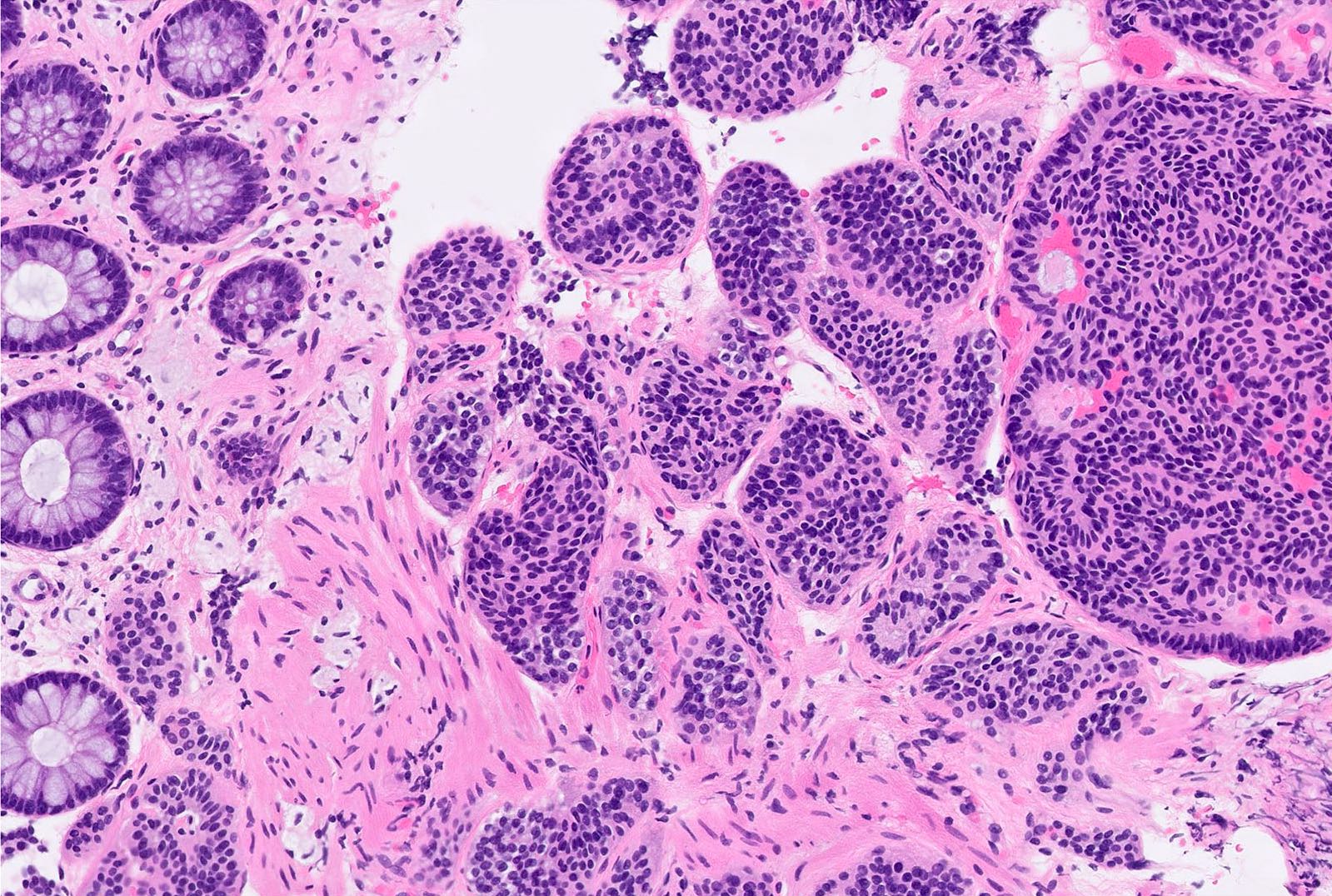
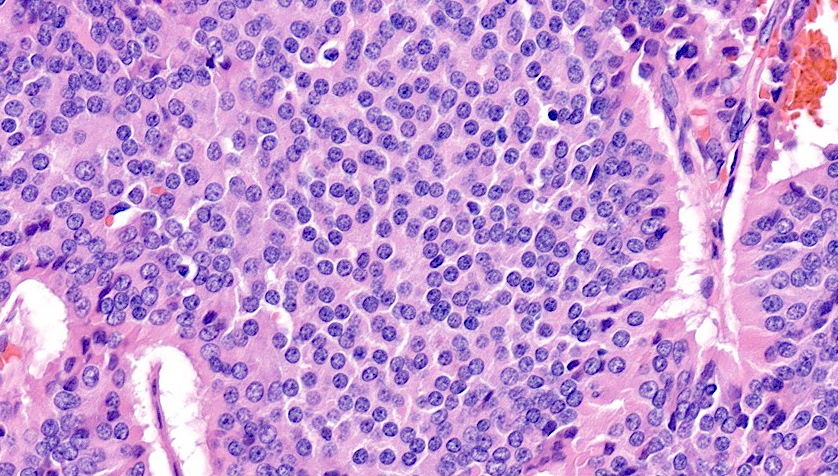
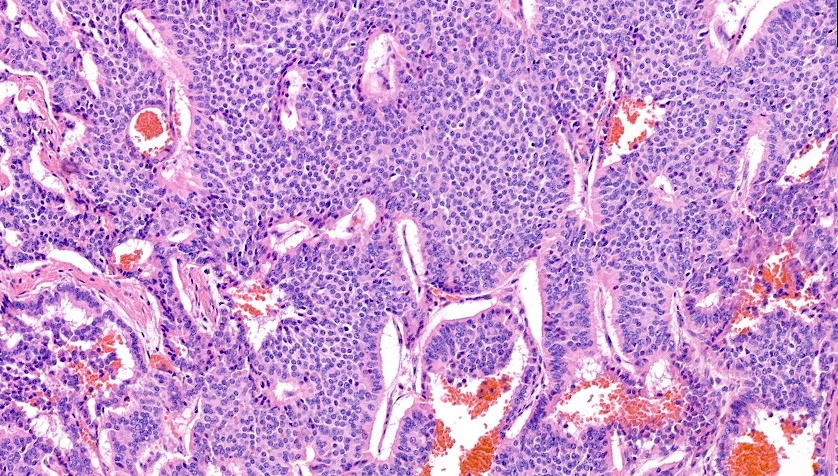
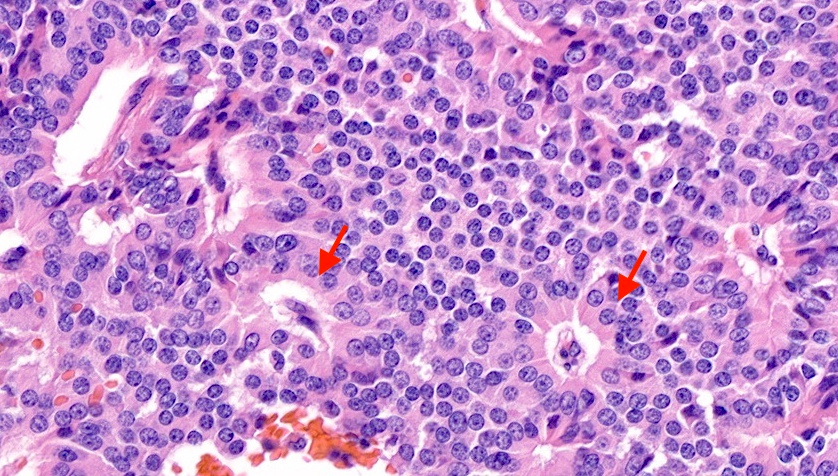
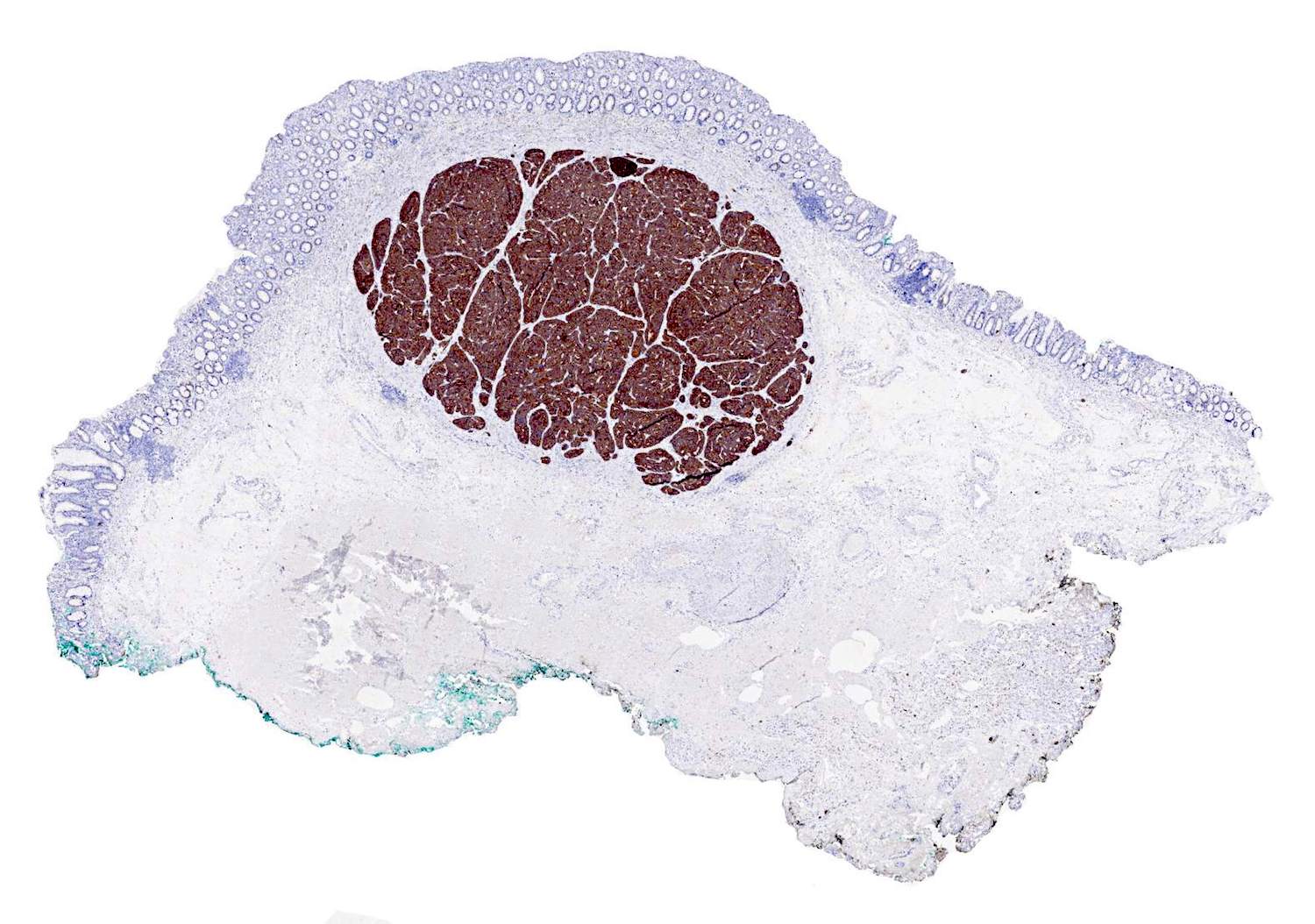
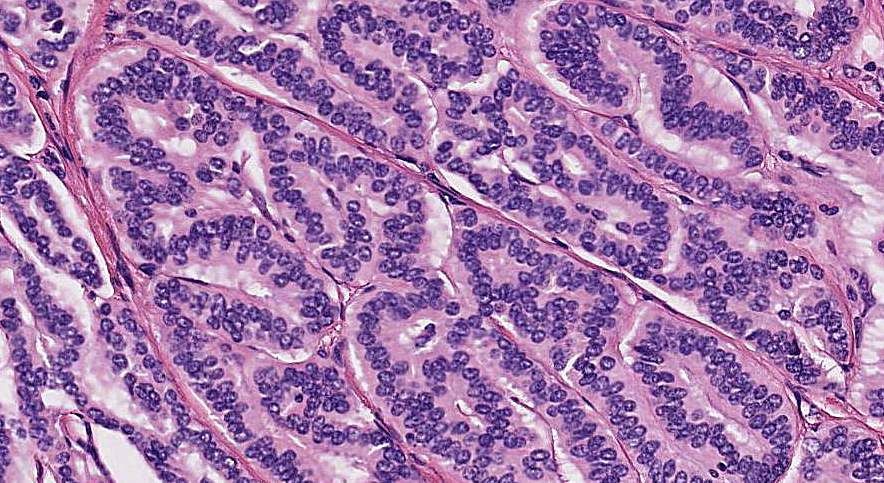
- Well differentiated neuroendocrine pattern:
- Monotonous regular cells with round or oval nuclei with salt and pepper chromatin and moderate eosinophilic granular cytoplasm
- Organoid architecture: tumor cells arranged in nests, trabecular or insular pattern (Oncologist 2016;21:1191)
- In some cases, nuclear pleomorphism may be encountered (endocrine atypia) but is not associated with tumor aggressiveness (Oncologist 2016;21:1191)
- Rarely mucin secretion
- Grading scheme is based on the proliferative rate (mitoses and Ki67 index) as follows:
- Low grade or grade 1 (G1): mitoses < 2 per 2 mm2 and Ki67 index < 3%
- Intermediate grade or grade 2 (G2): mitoses 2 - 20 per 2 mm2 or Ki67 index 3 - 20%
- High grade or grade 3 (G3): mitoses > 20 per 2 mm2 or Ki67 index > 20%
- Mitotic count should be evaluated in a 2 mm2 hotspot area (roughly equivalent to 10 high power fields with a 40x objective lens) (Arch Pathol Lab Med 2016;140:437)
- Ki67 index should be estimated in ≥ 500 cells in the hotspot regions (Oncologist 2016;21:1191)
- If there is any discrepancy between mitotic index and Ki67 index, the higher should be considered for the classification (World J Gastrointest Oncol 2020;12:791)
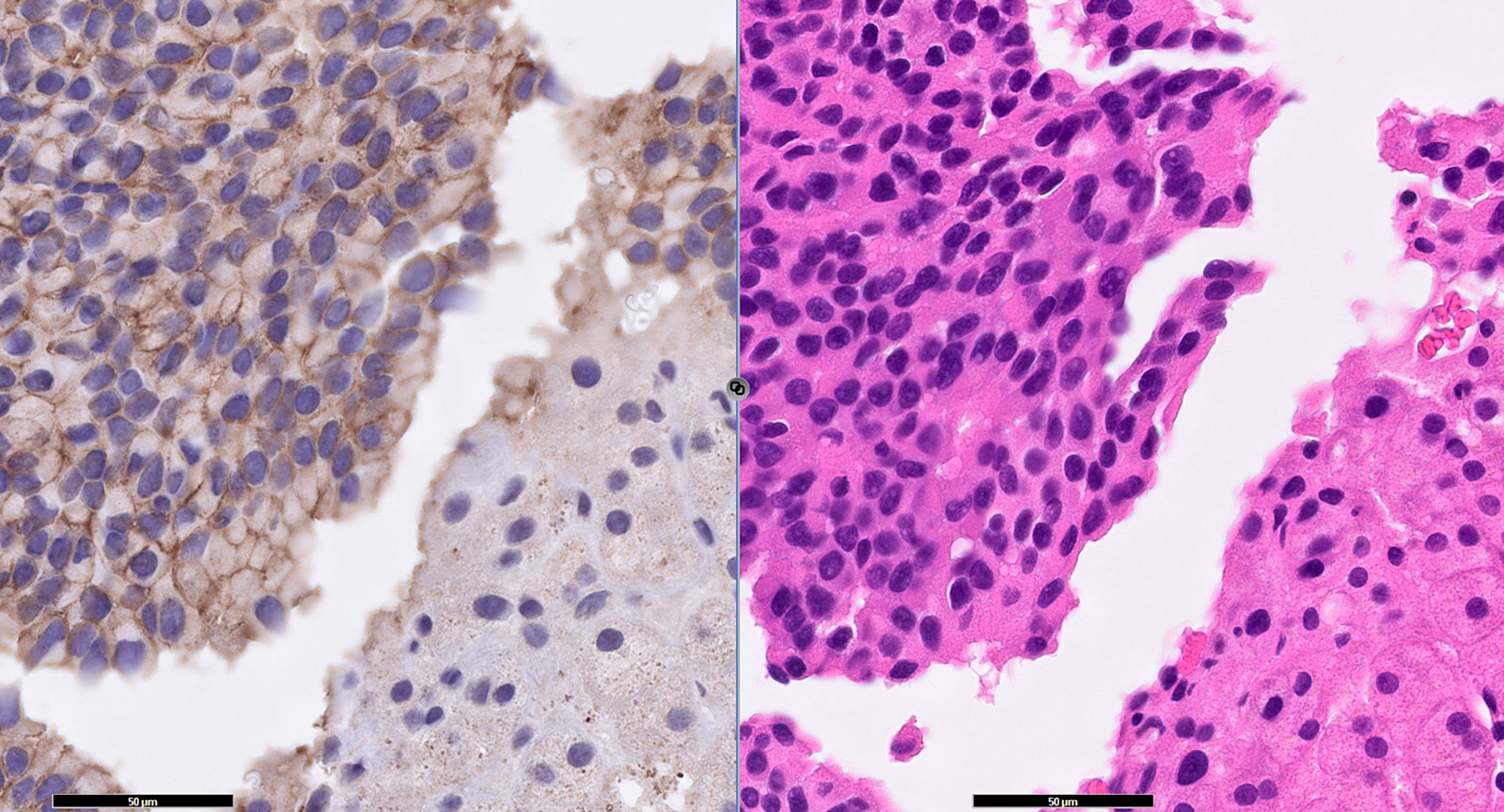
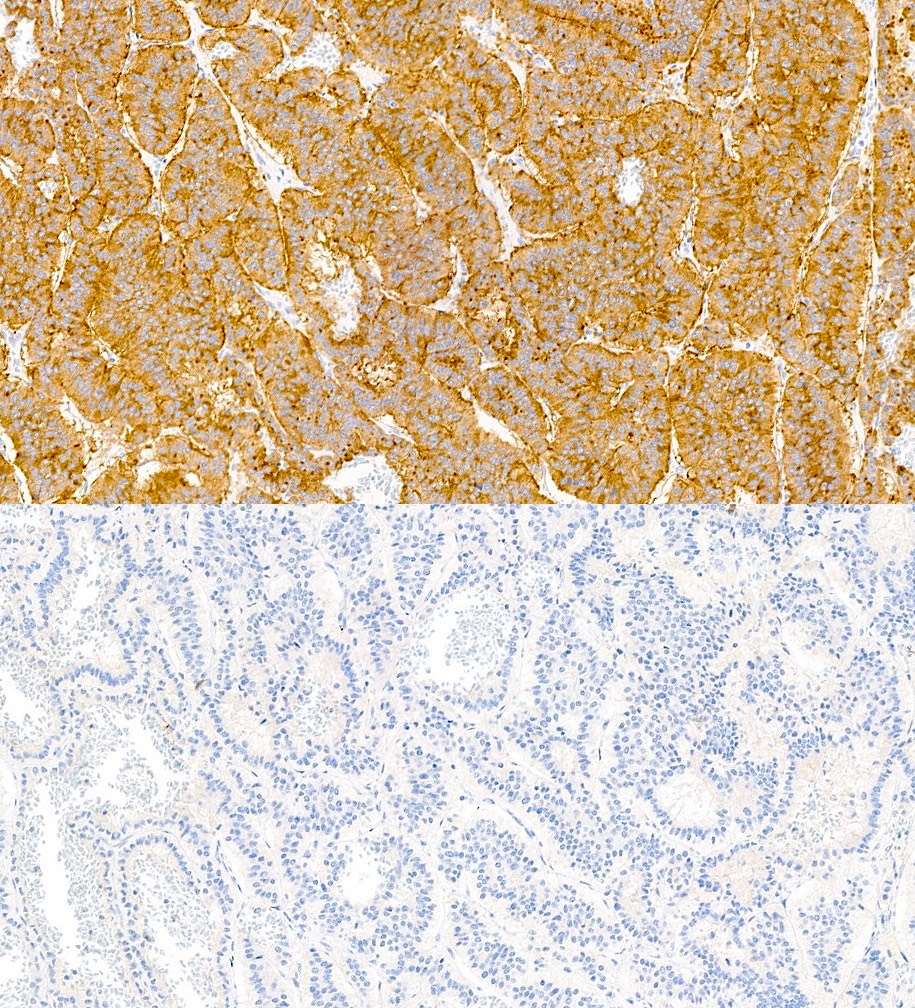
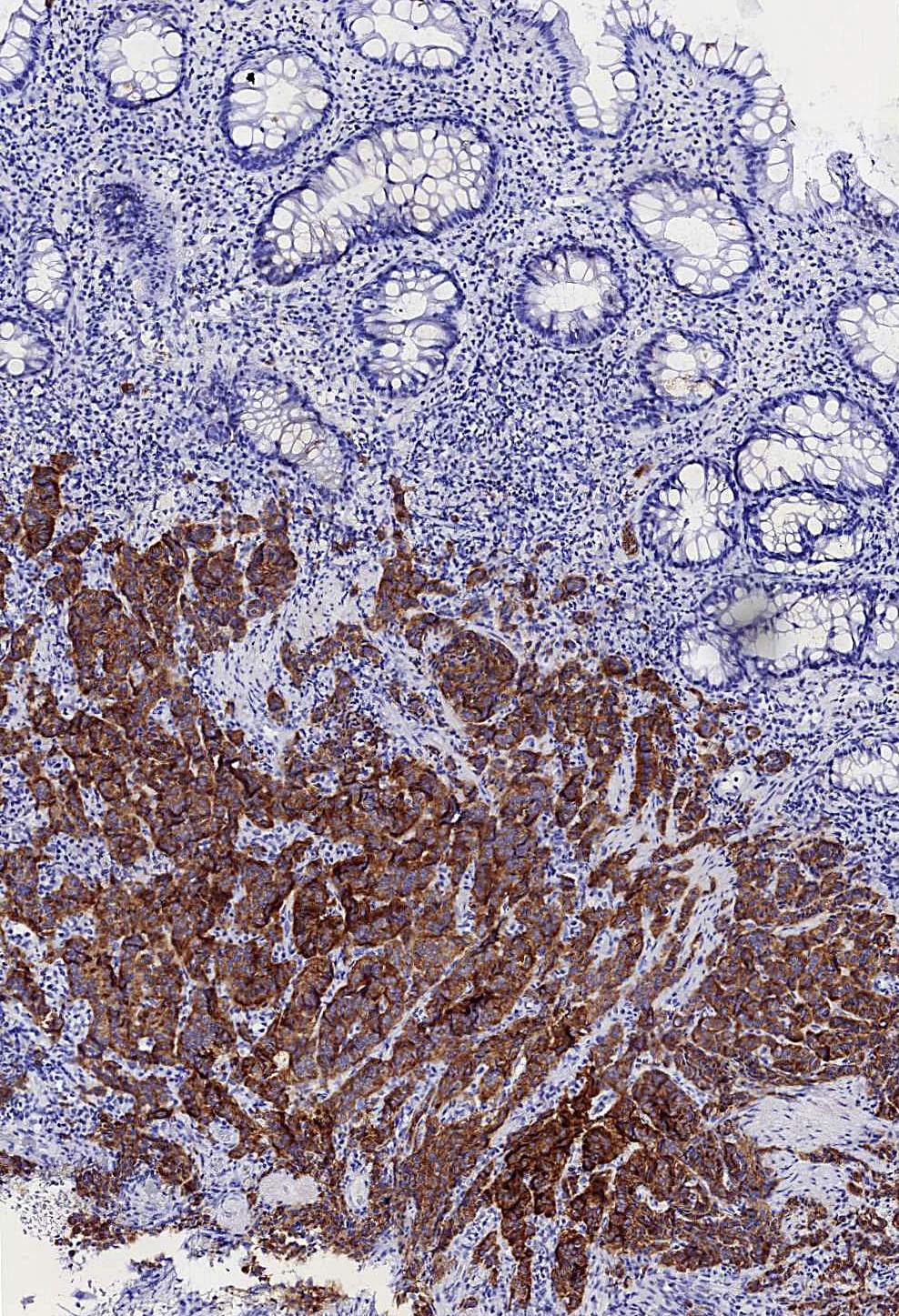
Microscopic (histologic) images
Contributed by Carolina Martinez Ciarpaglini, M.D., Ph.D. and Yuri Tachibana, M.D.
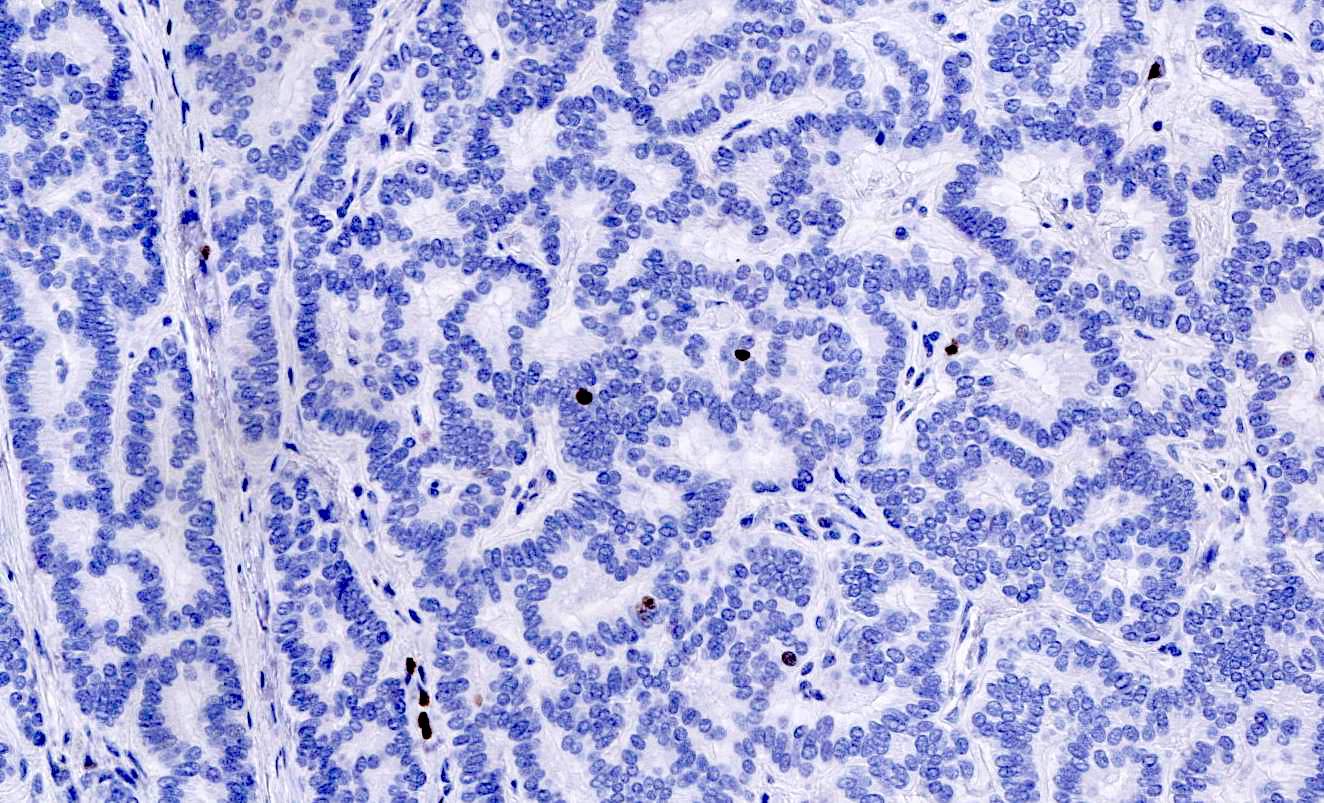
Positive stains
- Synaptophysin
- Chromogranin may occasionally be negative in rectal tumors (Neuroendocrinology 2017;105:196)
- CD56 / NCAM1, Leu7 and neuron specific enolase are not recommended because of their low specificity (Am J Surg Pathol 2010;34:300)
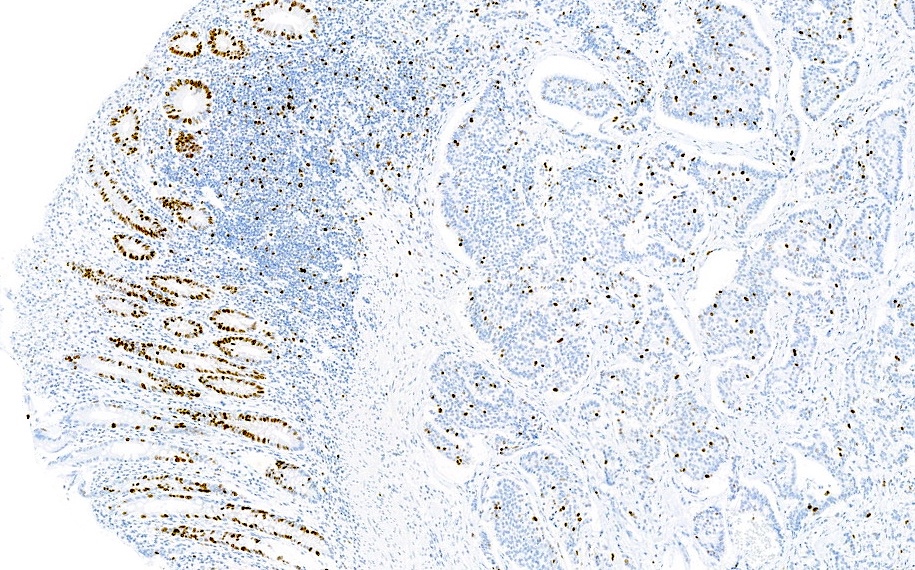
- CDX2 may be useful in determining the site of origin as it would point to the intestine or pancreas (Oncologist 2016;21:1191, Am J Surg Pathol 2010;34:300)
- PAX8 is positive in the majority of rectal and pancreatic well differentiated neuroendocrine tumors (Am J Surg Pathol 2010;34:723)
- CEA
- There are no specific recommendations for the study of the hormone secretion profile, however, somatostatin receptor 2 (SSTR2) immunohistochemical expression has been shown to be an independent prognostic marker (Eur J Nucl Med Mol Imaging 2017;44:468)
Molecular / cytogenetics description
- There is very limited data about molecular alterations in colorectal neuroendocrine tumors; most molecular analyses have been performed in pancreatic and small intestine tumors (Ann Endocrinol (Paris) 2019;80:153, Mod Pathol 2018;31:1770)
Sample pathology report
- Ascending colon mass, endoscopic biopsy:
- Well differentiated neuroendocrine tumor, WHO grade 2 (G2), 7 mm (see comment)
- Comment: There are 2 mitotic figures in 2 mm2 and the Ki67 proliferation index is approximately 5%, consistent with G2 in this material.
Differential diagnosis
- Neuroendocrine carcinoma (NEC):
- High grade tumors with poorly differentiated morphology
- Atypical small or large cells organized in diffuse sheets with little nesting
- Apoptotic bodies and necrosis are usually observed
- They are high grade by definition (mitoses > 20 per 10 high power fields or Ki67 > 20%); however, the Ki67 index is usually > 50% (Neuroendocrinology 2016;103:139)
- Remember that there are some G3 neuroendocrine tumors; poorly differentiated morphology is the main feature to distinguish neuroendocrine carcinomas from G3 neuroendocrine tumors
- BCL2 overexpression, loss of Rb expression and abnormal p53 expression are seen more frequently in neuroendocrine carcinomas and may be useful in the differential diagnosis with well differentiated neuroendocrine tumors (Arch Pathol Lab Med 2016;140:437)
Additional references
Practice question #1
A 5 mm polyp lesion located in the ascending colon was resected. The microscopic image is shown above. Which statement is true regarding this lesion?
- CD56 immunostaining is more specific than synaptophysin for its diagnosis
- CDX2 expression may indicate the site of origin of this type of tumor
- Presence / absence of necrosis is the main feature in defining the tumor grade
- Proliferative activity is not relevant in these tumors
Practice answer #1
B. CDX2 expression may indicate the site of origin of this type of tumor
Comment Here
Reference: Neuroendocrine tumor
Comment Here
Reference: Neuroendocrine tumor
Practice question #2
Which of the following is associated with aggressiveness in colon / rectal neuroendocrine tumors?
- CDX2 expression
- Cytologic atypia
- Lack of expression of chromogranin
- Mitotic activity
Practice answer #2