Table of Contents
Definition / general | Essential features | Terminology | ICD coding | Epidemiology | Sites | Pathophysiology | Etiology | Diagrams / tables | Clinical features | Diagnosis | Laboratory | Radiology description | Radiology images | Prognostic factors | Case reports | Treatment | Clinical images | Microscopic (histologic) description | Microscopic (histologic) images | Cytology description | Positive stains | Negative stains | Electron microscopy description | Electron microscopy images | Molecular / cytogenetics description | Molecular / cytogenetics images | Videos | Sample pathology report | Differential diagnosis | Additional references | Practice question #1 | Practice answer #1 | Practice question #2 | Practice answer #2Cite this page: Waqas O, Raza M, Ud Din N. Langerhans cell histiocytosis. PathologyOutlines.com website. https://www.pathologyoutlines.com/topic/mandiblemaxillalch.html. Accessed August 25th, 2025.
Definition / general
- Most common histiocytic disorder characterized by aberrant function and differentiation or proliferation of mononuclear phagocyte system cells
- Langerhans cell histiocytosis (LCH) (clonal neoplasm of myeloid dendritic cells) expresses CD1a and CD207 (Langerhans cell phenotype)
Essential features
- LCH is particularly a disease of childhood (in adults it is less common)
- Most commonly presents as an incidental lytic bone lesion and a skin lesion
- Characteristic LCH cell (large cell with nuclear grooves that are CD1a and CD207 positive) in an eosinophil rich mixed inflammatory background
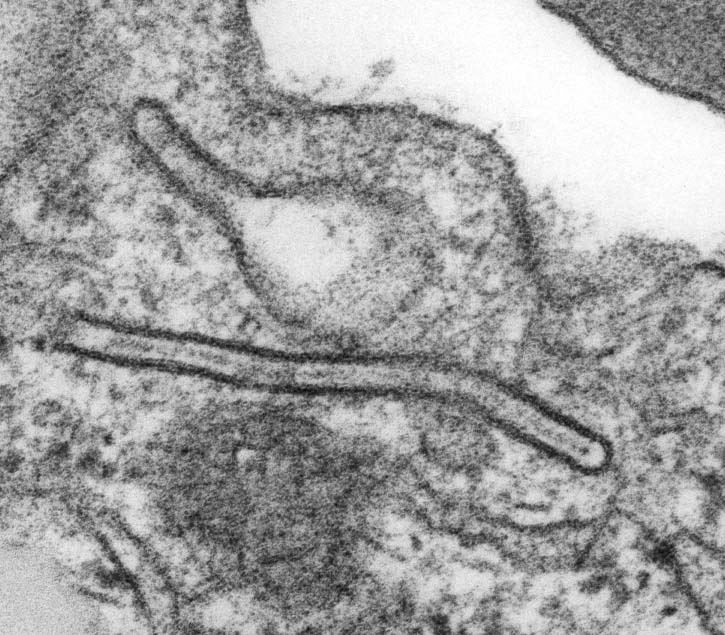
- Cytoplasmic Birbeck granules (tennis racket shaped structures) on electron microscopy (pathognomonic)
- BRAF V600E mutation: therapeutic and prognostic significance (Blood 2022;139:2601)
- Stage of the disease (including high risk organ involvement) determines the prognosis
Terminology
- Langerhans cell histiocytosis (current WHO 5th edition; recommended)
- Histiocytosis X and eosinophilic granuloma (not recommended)
- Hand-Schüller-Christian disease (lytic bone lesions, diabetes insipidus [DI] and exophthalmos)
- Letterer-Siwe disease (systemic disease with spleen, liver and bone marrow involvement)
- Hashimoto-Pritzker disease (congenital, self resolving skin lesions; benign variant of LCH)
ICD coding
Epidemiology
- Annual incidence rate in the pediatric population (below 2 years) is 5 - 9 new cases per 1 million population; in adults, 1 - 2 new cases per 1 million population (Pediatr Blood Cancer 2008;51:71, Br J Haematol 2022;199:728)
- Slight male predominance
- Pulmonary Langerhans cell histiocytosis (PLCH) is more common during the third and fourth decades of life (Immunol Rev 2010;234:213)
- More commonly found in Hispanics and individuals of European descent (Blood 2016;127:2672)
Sites
- Can arise in virtually any organ system but has a particular affinity for bone (79%), skin (36%) and pituitary gland (25%)
- Among bones, skull is the most common site; less common sites include mandible and orbit (World J Pediatr 2019;15:536)
- Lung is a common site in young men with smoking history
- Other organs include liver, spleen, lymph node, gastrointestinal tract and parotid gland
Pathophysiology
- Constitutive activation of MAPK gene pathway with universal phosphorylated ERK (p-ERK) expression leading to disrupted cell migration, apoptosis inhibition and oncogene induced senescence (N Engl J Med 2018;379:856)
- Majority harbor somatic BRAF V600E mutation (most common) and MAP2K1 (second common)
- Misguided myeloid differentiation model: stage of differentiation in which the myeloid cell acquires activating MAPK mutations determines the extent of LCH
- High risk, multisystem LCH: self renewing stem or progenitor cells from bone marrow (J Exp Med 2015;212:281)
- Low risk, multisystem LCH: MAPK activation of committed dendritic cell (DC) precursors or monocytes
- Low risk, single lesion: arises from a regional dendritic cell precursor
Etiology
- Etiology of LCH remains unknown
- Cigarette smoking (chronic irritant) plays role in PLCH (N Engl J Med 2022;387:2449)
Clinical features
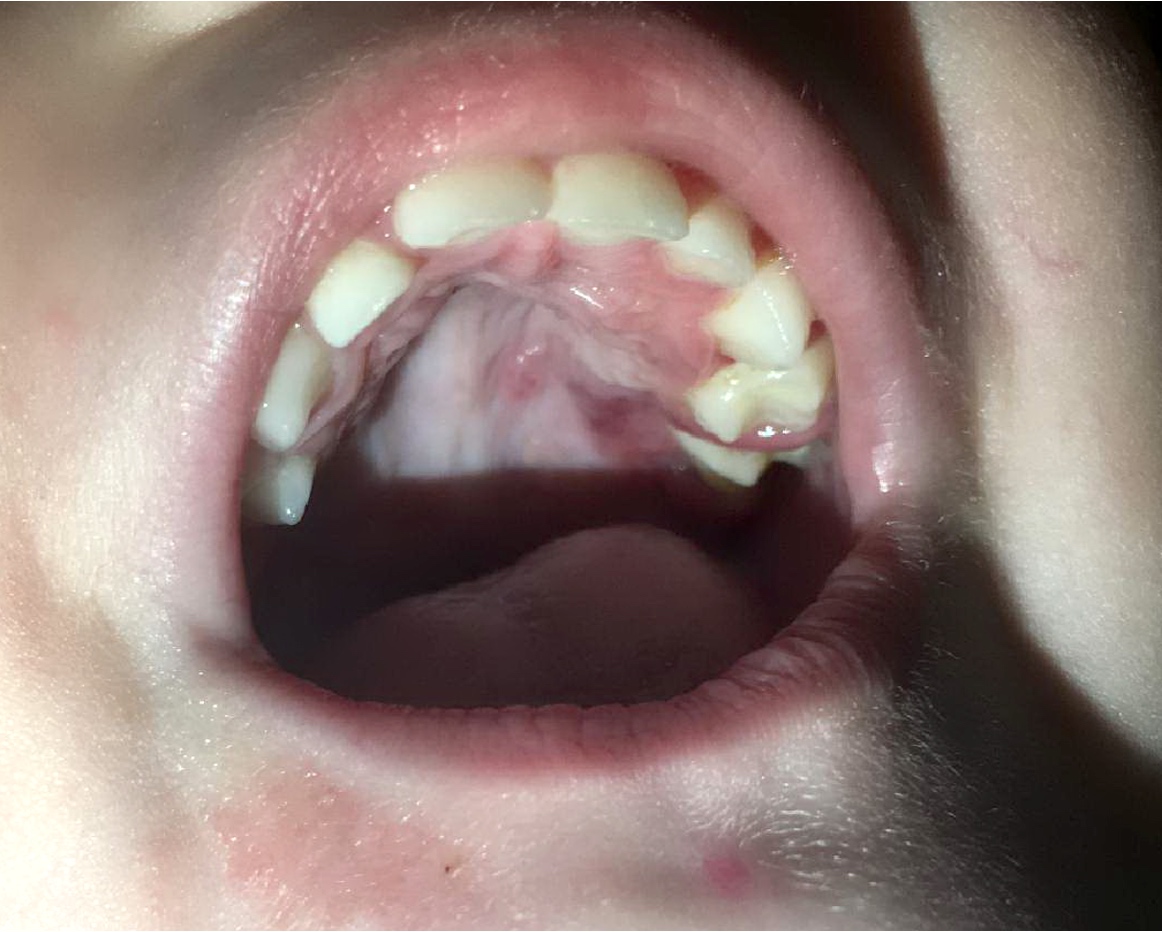
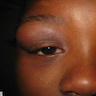
- Facial swelling and preauricular pain (most common) due to tooth mobility and involvement of the posterior mandibular region, indicating alveolar process damage (Medicine (Baltimore) 2019;98:e16331)
- Skin manifestations (particularly scalp): seborrheic dermatitis-like eruptions, erythematous, crusted, scaly papules and eczematous lesions (Head Neck Pathol 2021;15:41)
- Pituitary gland involvement: central diabetes insipidus (most common initial sign of central nervous system [CNS] LCH), growth hormone deficiency and thyroid dysfunction (Pediatr Blood Cancer 2018;65:e26784)
- LCH associated neuroinflammation and neurodegeneration: neurological deficit, cognitive impairment and behavioral changes
Diagnosis
- Lesional tissue biopsy for confirmation (even if the case is highly suggestive of LCH through clinical and imaging findings with a reasonable exception for single system PLCH with classic radiologic and clinical findings) (Blood 2022;139:2601)
- Baseline full body (vertex to toes) FDG PET / CT (including distal extremities) to define the extent of the disease
- Magnetic resonance imaging (MRI) of the brain with gadolinium, with a dedicated examination of the sella turcica for pituitary dysfunction and neurological symptoms
- All patients should undergo BRAF V600E mutational analysis for the diagnosis and treatment
- For wild type BRAF V600E LCH cases, next generation sequencing (NGS) can be performed to assess MAPK-ERK pathway mutations for questionable diagnosis or second line treatment
Laboratory
- Liver function tests (LFTs) to assess liver function
- Complete blood count with differential (CBC) for cytopenia
- Inflammatory markers (C reactive protein)
- Morning urine and serum osmolality and water deprivation test (to evaluate DI)
- Morning serum cortisol, thyroid stimulating hormone (TSH) and free T4, follicle stimulating hormone (FSH) / luteinizing hormone (LH), prolactin, with testosterone (men) and estradiol (women) in case of pituitary dysfunction or involvement on MRI (Blood 2022;139:2601)
- Low insulin-like growth factor 1 levels due to suspected growth hormone deficiency
- Cell free DNA analysis on peripheral blood for BRAF V600E mutation in cases of absent or insufficient tissue (variable sensitivity but category A consensus recommendation)
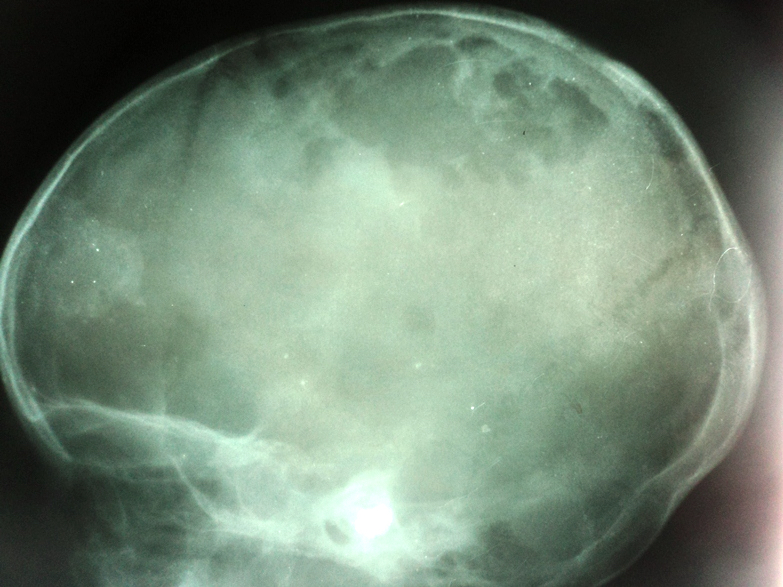
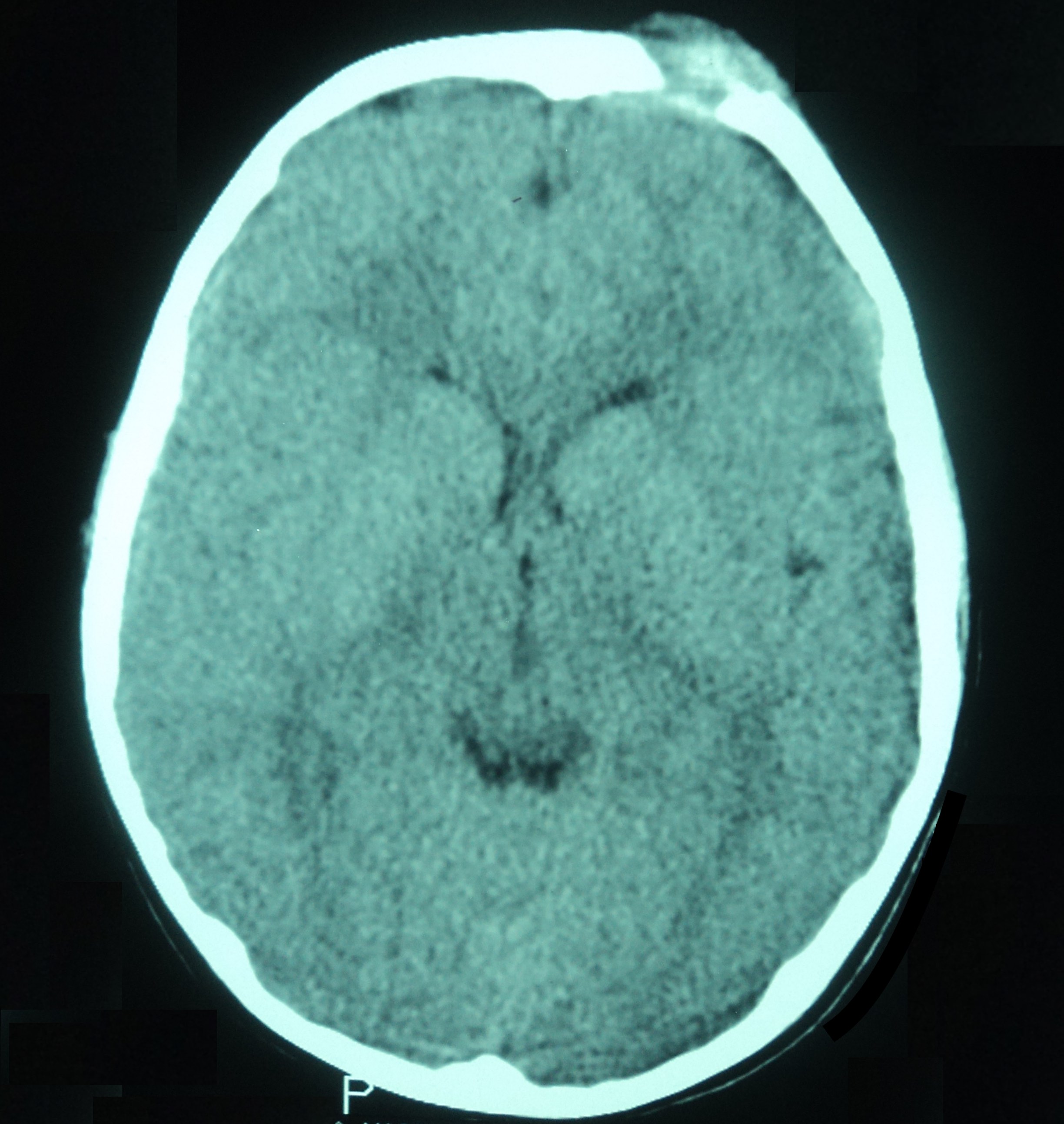
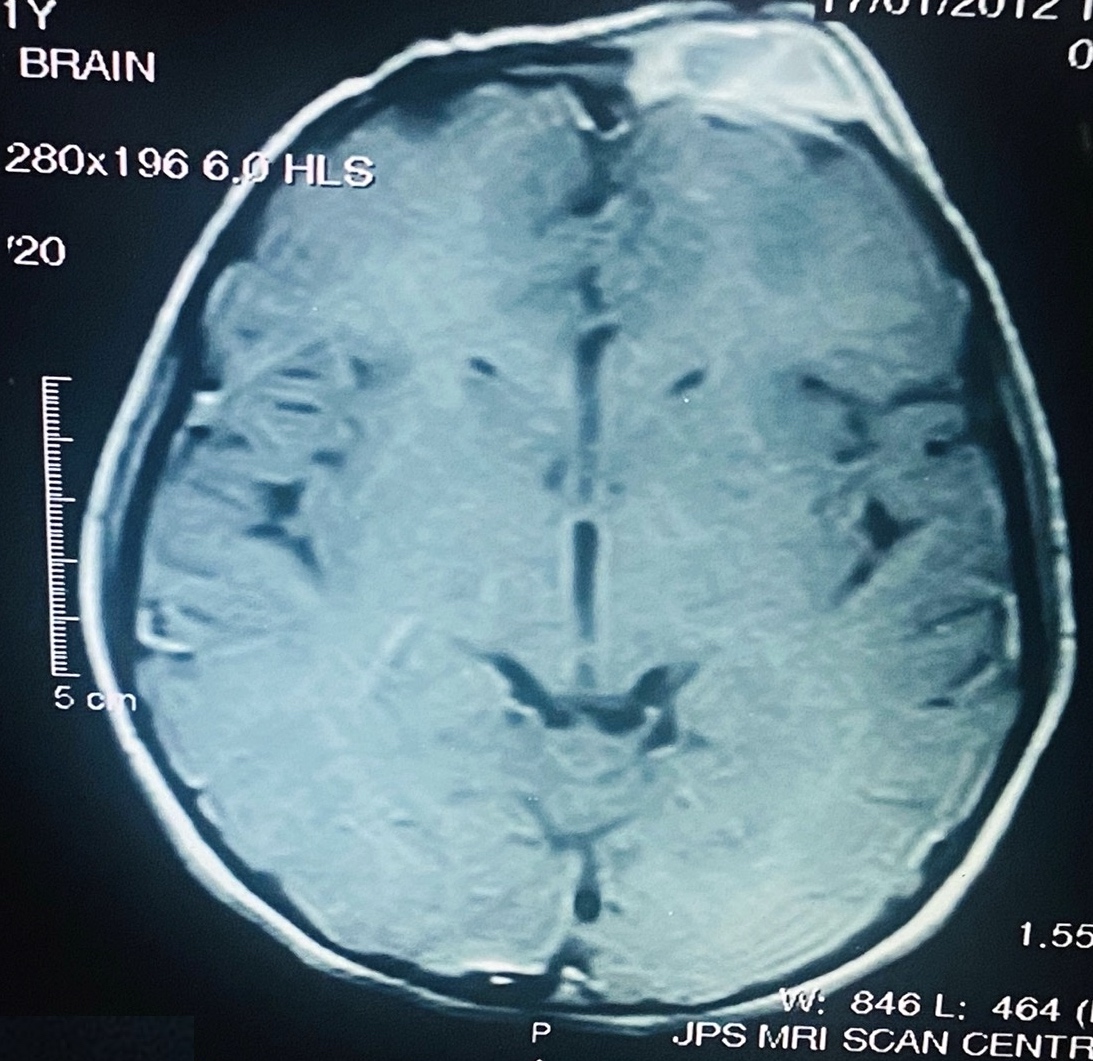
Radiology description
- Punched out, lytic osseous lesions, often involving flat bones (skull, sternum, ribs, pelvis)
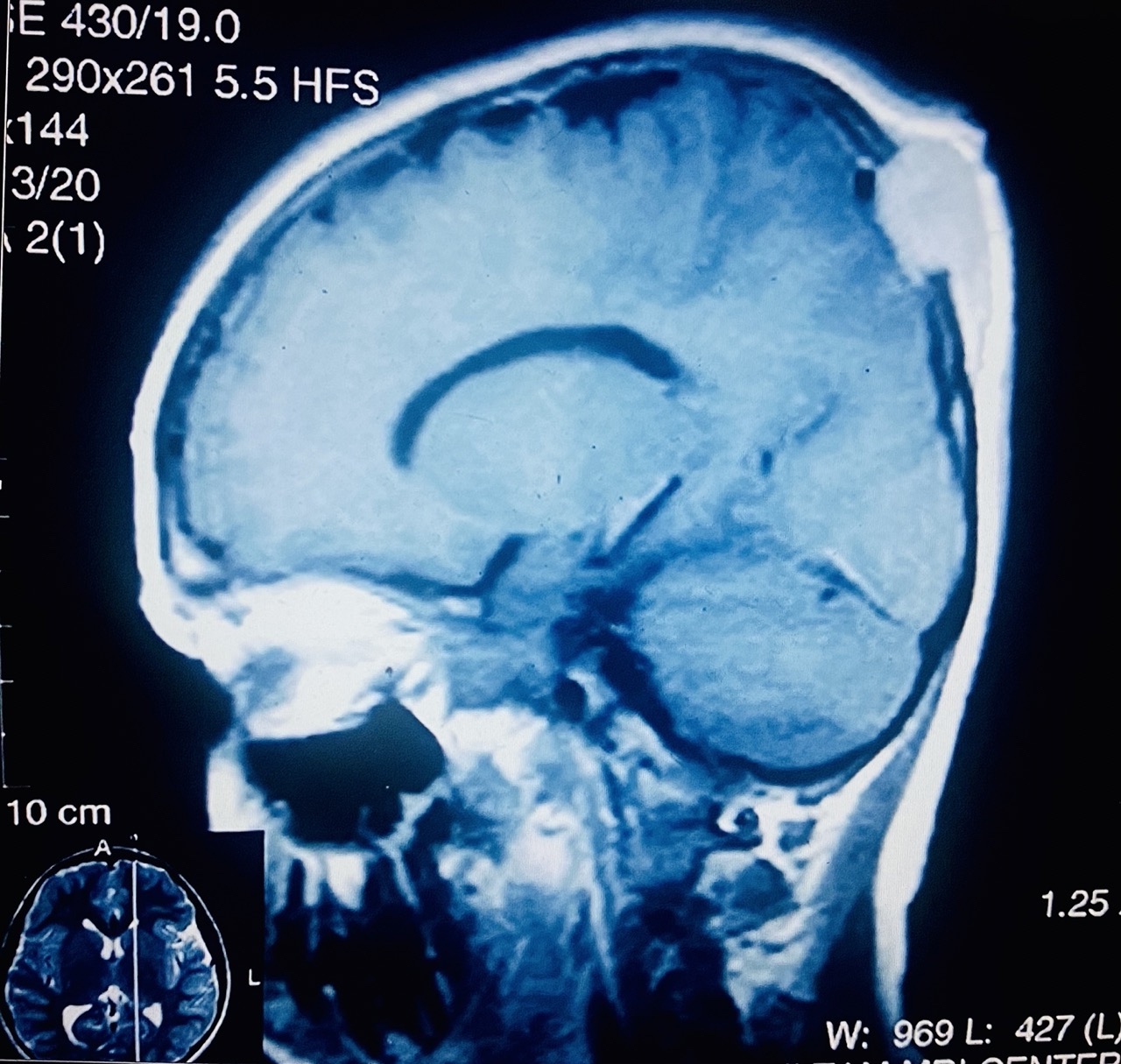
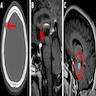
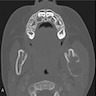
- In jaw: expansile soft tissue mass with mixed attenuation or nonenhancing, ill defined, infiltrating lesion with hyperintense T2 signal intensity and radiolucent lesion with thinning of the cortex (mandible) on plain orthopantomogram (OPG) (Medicine (Baltimore) 2019;98:e16331)
- Upper lobe predominant nodular and cystic lung lesions in a smoker (J Thorac Imaging 2019;34:W144)
Radiology images
Prognostic factors
- Largely dependent on the extent of the disease, high risk organ involvement (brain, lung and liver) and response to therapy (Blood 2022;139:2601)
- Unifocal LCH has an excellent prognosis with a 5 year survival of > 90%
- Single system (SS) LCH (lung) has variable course depending upon pulmonary function and development of pulmonary hypertension (HTN) (death occurs due to respiratory failure)
- Multisystem (MS) LCH / disseminated LCH has improved overall 5 year survival by current therapy regimes
- Permanent consequences of diabetes insipidus and neurodegeneration have worse clinical outcome
Case reports
- 4 year old boy presented with painful swelling left cheek (Clin Case Rep 2021;9:e04321)
- 10 year old boy presented with right temporoparietal region swelling after a fall from bicycle (Int J Surg Case Rep 2021;85:106179)
- 46 year old male nonsmoker with a history of fever and dry cough, longstanding hypothyroidism and diabetes insipidus (World J Clin Cases 2021;9:11029)
- 47 year old woman presented with headache, weakness in lower extremities and urinary retention (Radiol Case Rep 2022;17:1407)
- 65 year old man with intermittent pruritis, fever and abnormal liver function tests (Cureus 2020;12:e8591)
Treatment
- Unifocal LCH: for bone, limited curettage followed by intralesional corticosteroid therapy / radiation therapy (10 - 20 Gy) in case of soft tissue component
- Single system PLCH: smoking cessation, corticosteroid and cladribine based systemic therapy (Blood 2022;139:2601)
- Multifocal multisystem LCH: cladribine and methotrexate combination
- BRAF and MEK inhibitors under trial
Clinical images
Microscopic (histologic) description
- Active lesions
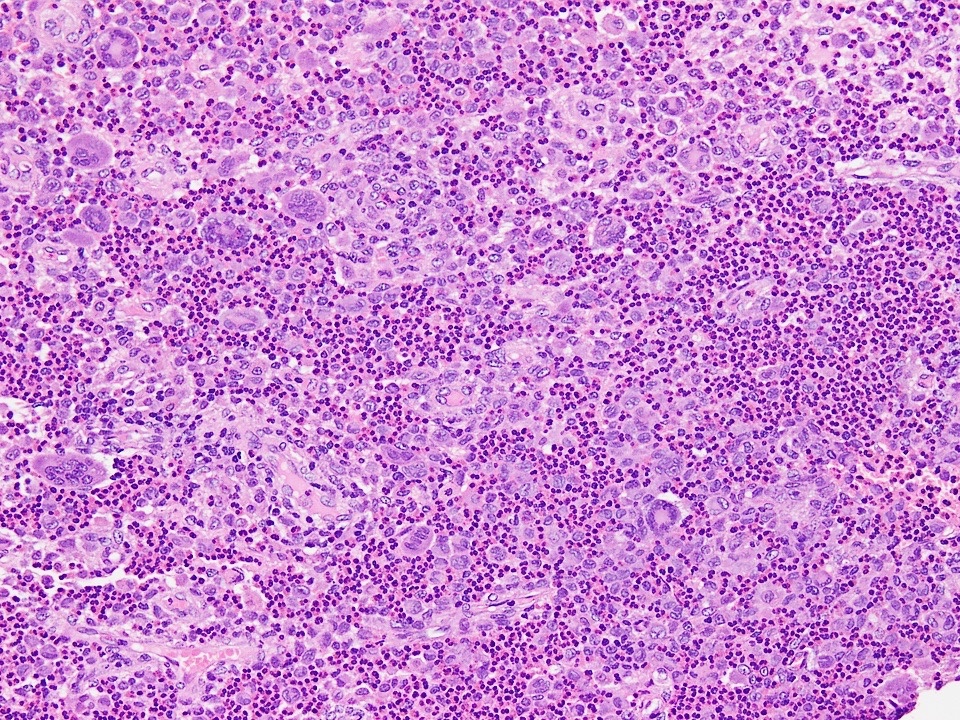
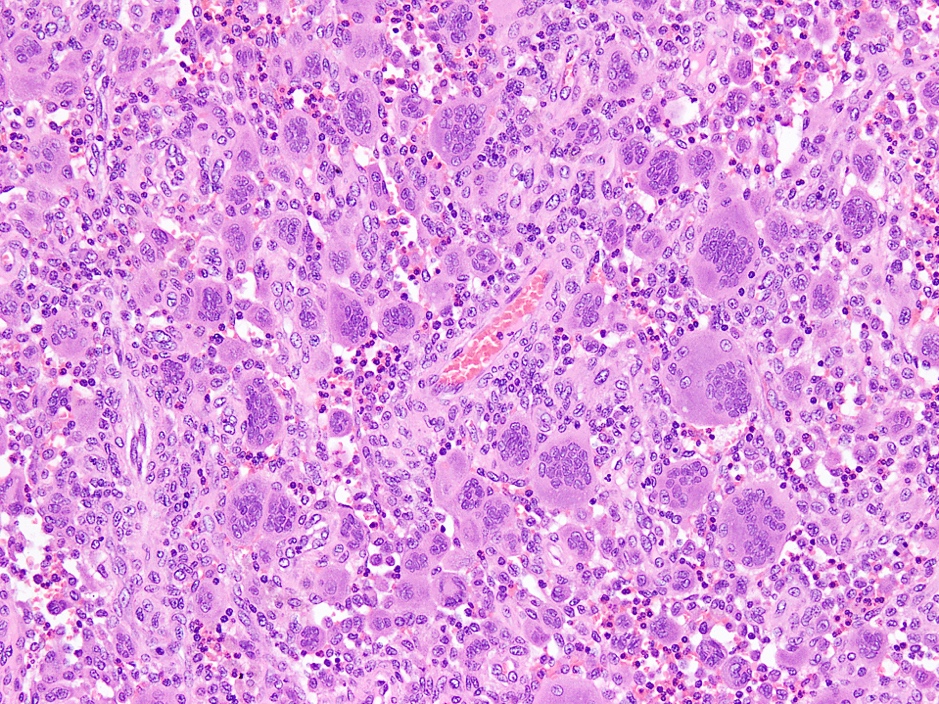
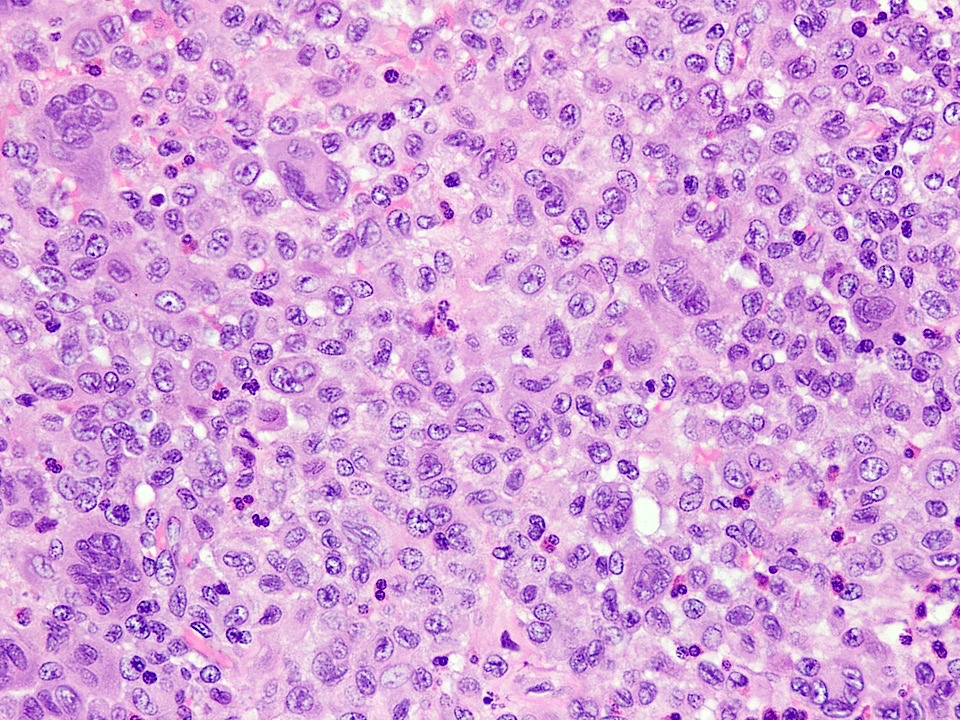
- Sheets of large, round to oval cells with nuclear grooves (coffee bean appearance), abundant eosinophilic cytoplasm, minimal nuclear atypia and variable mitotic count (no atypical forms)
- Background predominantly eosinophil rich with scattered multinucleated histiocytes (LCH type and osteoclast-like); neutrophils and plasma cells are rare
- Central necrosis is common but of no prognostic significance (Int J Surg Case Rep 2021;85:106179)
- Late lesions
- Decreased number of lesional cells, accompanied by fibrosis
- Neurodegenerative lesions: collection of foamy macrophages with CD1a positive cells or perivascular infiltrate of mononuclear cells
- Biliary type lesions: portal noncaseating granuloma with eosinophil rich infiltrate and multinucleated giant cells encasing the duct
- PLCH: focal bronchiolocentric eosinophil rich inflammatory infiltrate with clusters of characteristic lesional cells, noncaseating granuloma and multinucleated giant cells
Microscopic (histologic) images
Cytology description
- Typical LCH cells have longitudinal grooves, folded nuclei and abundant pale cytoplasm, which are best appreciated on Pap stain
- Background is eosinophil rich with scattered multinucleated cells
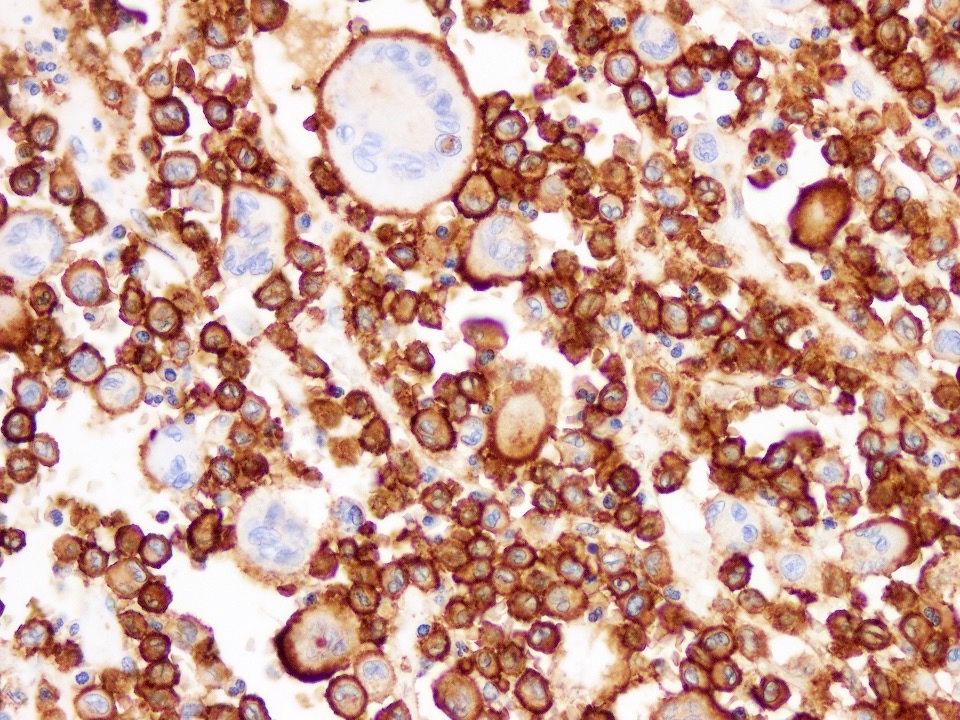
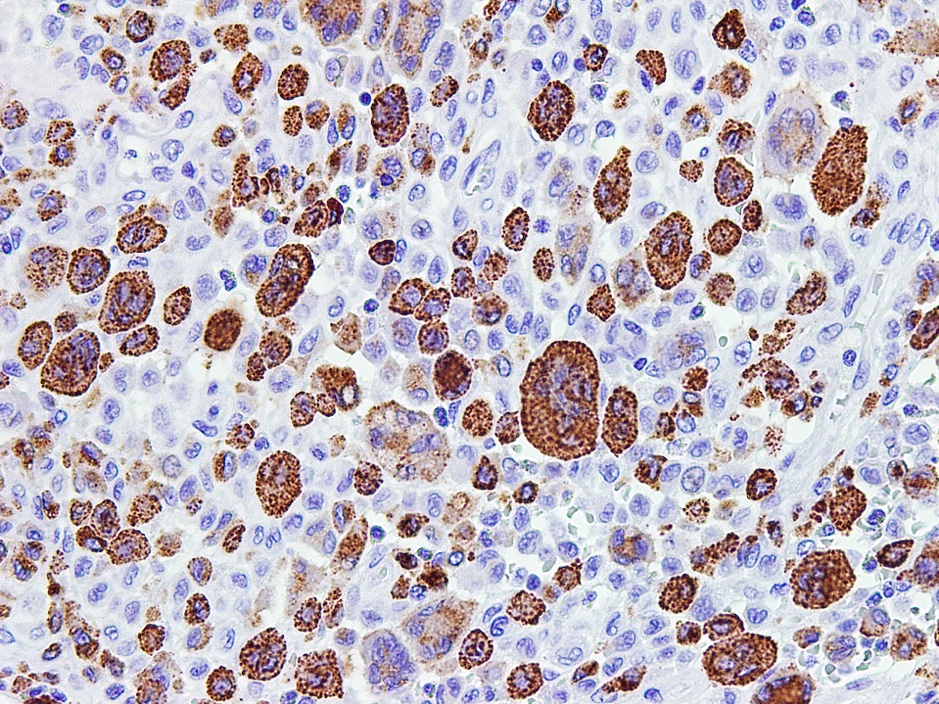
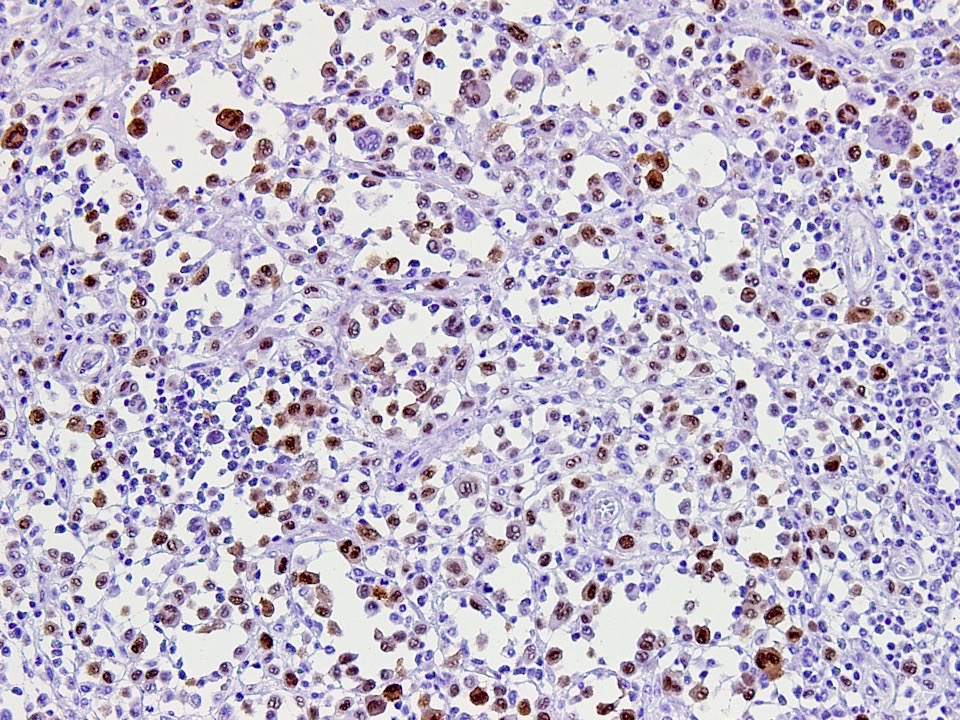
Positive stains
- CD1a: membranous staining
- CD207 (langerin): granular cytoplasmic staining
- CD68: Golgi dot-like staining
- Cyclin D1
- Reference: Blood 2022;139:2601
Negative stains
- CD30
- CD20
- HMB45
- CD21
- Reference: Blood 2022;139:2601
Electron microscopy description
- Indented nucleus with cytoplasmic vacuoles, lamellated dense bodies and Birbeck granules (tennis racket type)
Molecular / cytogenetics description
- BRAF V600E mutation (most common)
- MAPK21 (second most common)
Videos
Radiology and histopathology descriptions
Sample pathology report
- Lytic lesion scalp, biopsy:
- Langerhans cell histiocytosis, not otherwise specified (see comment)
- Comment: The biopsy shows a histiocytic lesion with dense eosinophilic infiltrate, admixed histiocytes with folded nuclei and longitudinal grooves and scattered multinucleated osteoclast-like giant cells. The histiocytes are positive for CD1a and cyclin D1 and negative for CD68, features consistent with Langerhans cell histiocytosis. Radiological skeletal survey, 99mTc bone scan and MRI brain imaging with gadolinium / FDG PET / CT guided imaging is recommended for diagnostic staging.
Differential diagnosis
- Skin
- Angiolymphoid hyperplasia with eosinophilia (ALHE) / epithelioid hemangioma:
- Solitary / multiple skin nodules or papules (J Int Med Res 2021;49:3000605211040976)
- Head and neck region is most common (particularly scalp)
- Female predilection
- Age range: 30 - 50 years
- Microscopy: presence of abundant eosinophils, reactive lymphoid follicles and vascular proliferation (lined by plump endothelial cells)
- Kimura disease:
- Chronic inflammatory disorder (allergic disease)
- More commonly found in Asians with slight male predominance
- Usually deep seated lesion is associated with lymphadenopathy
- Elevated eosinophil count and serum IgE levels (J Int Med Res 2021;49:3000605211040976)
- Abundant lymphocytes, eosinophils (including abscesses) and plasma cells, always with lymphoid follicles
- Juvenile xanthogranuloma:
- Histiocytic (non-Langerhans cell) disorder
- Usually first year of life but can be seen in adults
- Male predominance
- Head and neck predilection (especially orbit) followed by trunk and extremities (BMC Pediatr 2022;22:87)
- Solitary / multiple yellowish papules and nodules
- Touton-like multinucleated giant cells and mononuclear cells (CD68 and CD163 positive)
- Angiolymphoid hyperplasia with eosinophilia (ALHE) / epithelioid hemangioma:
- Head and neck
- Central giant cell granuloma:
- Locally invasive, intraosseous, benign lesion
- Associated with local trauma, repair process or developmental disorder
- Mandible is most commonly affected bone followed by temporal and sphenoid bone (sella turcica)
- Clinical findings: painful jaw swelling, proptosis or headache
- Imaging: well circumscribed, radiolucent and expansile lytic bone lesion with cortical thinning (Dentomaxillofac Radiol 2021;50:20200429)
- Microscopy: numerous multinucleated osteoclast-like giant cells and mononuclear stromal cells, remote hemorrhage and hemosiderin laden macrophages (Histol Histopathol 2008;23:1151)
- Immunostains: CD68 positive and CD1a negative
- Extranodal Rosai-Dorfman disease:
- Benign histiocytic disorder
- Paranasal sinuses (particularly maxilla) and mandible are most common extranodal sites
- Clinical features: presents as nasal obstruction, proptosis or jaw swelling
- Imaging: expansile osteolytic lesion
- More commonly found in individuals who are 22 - 55 years of age with no gender predilection
- Microscopy: sheets of large histiocytes with abundant eosinophilic cytoplasm, mixed inflammatory infiltrate and emperipolesis (engulfment of eosinophils, plasma cells, lymphocytes or neutrophils by large histiocytes) (Head Neck Pathol 2020;14:442)
- Immunostains: S100, CD68 and CD163 positive and CD1a negative
- Erdheim-Chester disease (ECD):
- Rare multisystem histiocytic (non-Langerhans cell type) disorder
- Middle aged adults (mean age: 46 years); pediatric cases are exceedingly rare
- Male preponderance
- Clinical findings: retro-orbital pain and exophthalmos, orbital masses, polydipsia and polyuria (central DI), long bone pains (most common), renal insufficiency and lung function compromise (multisystem disease)
- Imaging: osteosclerotic lesions rather than punched out (as in LCH), abnormal signals in pituitary stalk on MRI
- Microscopy: foamy histiocytes with small nuclei, Touton-like and multinucleated giant cells; in some cases, infiltration of lymphocytes and plasma cells (Blood 2020;135:1929)
- Immunostains: CD68 (cytoplasmic positivity), CD163 (membranous positivity), ALK (cytoplasmic positivity) and CD1a negative
- Central giant cell granuloma:
Additional references
Practice question #1
A 17 year old girl presented with a lesion in the pituitary fossa. Biopsy shows dense mixed inflammation (containing eosinophils) and large cells with pale cytoplasm, containing vesicular nuclei with grooves and membrane folding. Which immunohistochemical stain will be confirmatory for the diagnosis?
- CD1a
- CD30
- Chromogranin
- CK
- SALL4
Practice answer #1
A. CD1a. The histologic findings described above are characteristic of Langerhans cell histiocytosis. CD1a is a specific marker of Langerhans cell histiocyte. Answer B is incorrect because large multinucleated cells (simulating Reed-Sternberg cells in a mixed inflammatory background) do not express CD30 stain. Answers D and C are also incorrect because the morphology shown in the images lacks features of pituitary adenoma (stained by chromogranin) or metastatic carcinoma (stained by CK). Answer E is incorrect because SALL4 is a marker of germ cell tumors and is negative in histiocytic lesions.
Comment Here
Reference: Langerhans cell histiocytosis
Comment Here
Reference: Langerhans cell histiocytosis
Practice question #2
A 35 year old male smoker presents with a history of mild exertional dyspnea. High resolution computed tomography shows reticulonodular opacities and small cysts. The lung biopsy shows CD1a positive histiocytes, eosinophils and admixed plasma cells. The electron microscopy shows tennis racket shaped granules. Which of the following is a surrogate marker of these cytoplasmic granules?
- CD1a
- CD163
- CD207
- CD68
- S100
Practice answer #2
C. CD207. CD207, also called langerin, is a part of type II lectin membrane protein and is responsible for the formation of Birbeck granules. Answer A is incorrect because CD1a stains membrane bound proteins rather than cytoplasmic. Similarly, answers B, D and E are incorrect because these are markers of non-Langerhans cell histiocytes.
Comment Here
Reference: Langerhans cell histiocytosis
Comment Here
Reference: Langerhans cell histiocytosis