Table of Contents
Definition / general | Essential features | Terminology | ICD coding | Epidemiology | Sites | Pathophysiology | Etiology | Clinical features | Diagnosis | Laboratory | Radiology description | Prognostic factors | Case reports | Treatment | Gross description | Gross images | Microscopic (histologic) description | Microscopic (histologic) images | Virtual slides | Cytology description | Positive stains | Negative stains | Electron microscopy description | Molecular / cytogenetics description | Sample pathology report | Differential diagnosis | Practice question #1 | Practice answer #1 | Practice question #2 | Practice answer #2Cite this page: Lobo J, Henrique R. Yolk sac tumor. PathologyOutlines.com website. https://www.pathologyoutlines.com/topic/testisyst.html. Accessed September 29th, 2025.
Definition / general
- Germ cell neoplasm composed of cells / structures reminiscent of embryonic / fetal yolk sac, allantois and extraembryonal mesenchyme
Essential features
- 2 biologically distinct entities:
- Postpubertal type (germ cell neoplasia in situ [GCNIS] related and usually as part of mixed tumor)
- Prepubertal type (unrelated to germ cell neoplasia in situ and usually pure)
- Multiplicity of histological features with various mimickers
- Most overlooked germ cell tumor subtype
- Elevated serum alpha fetoprotein (AFP) should raise the suspicion for a yolk sac component
Terminology
- First described / studied by Gunnar Teilum and later by Aleksander Talerman (Am J Surg Pathol 1981;5:719)
- Complex nomenclature over time, reflecting the remarkable heterogeneity: primitive endodermal tumor, yolk sac carcinoma, endodermal sinus tumor, mesoblastoma vitellinum, orchioblastoma, polyvesicular vitelline tumor, adenocarcinoma of the infant testis, extraembryonic mesoblastoma; malignant endothelioma of perithelioma type (currently less used and not recommended by WHO 2016) (Int J Surg Pathol 2014;22:677, Histopathology 2012;60:1023)
ICD coding
Epidemiology
- Postpubertal type:
- Almost always part of mixed tumors (44% of nonseminomas); pure form exceedingly rare (0.6% of testicular germ cell tumors)
- Epidemiological trends common to all germ cell neoplasia in situ related tumors (15 - 40 years, rising incidence, environmental, genetic and epigenetic triggers) (Int J Mol Sci 2019;20:258)
- Prepubertal type:
- Rare (2 - 3 cases/year/1 million) and usually pure (mixed forms with teratoma are the exception)
- Children < 6 years (median age 16 - 20 months)
- Does not follow epidemiological trends / associations of postpubertal type tumors
Sites
- Testis
- Also found in ovary, extragonadal (along midline of the body)
Pathophysiology
- Postpubertal type:
- Derived from germ cell neoplasia in situ (primordial germ cells / gonocytes arrested in maturation → germ cell neoplasia in situ → reprogramming → extraembryonal differentiation)
- Type II germ cell tumor (Nat Rev Cancer 2019;19:522)
- Prepubertal type:
- Not derived from germ cell neoplasia in situ
- Hypothesis: derived from teratoma
- Type I germ cell tumor
Etiology
- Postpubertal type:
- Environmental factors: testicular dysgenesis syndrome, subfertility
- Genetic factors: single nucleotide polymorphism (SNP) variants (KITLG, SPRY4, DMRT1)
- Prepubertal type:
- Unknown
- References: Int J Mol Sci 2019;20:258, Nat Rev Cancer 2019;19:522
Clinical features
- Unilateral testicular mass, often painless (World J Surg Oncol 2014;12:400)
- Postpubertal type:
- More aggressive (compared to prepubertal): around 40% stage I disease; metastatic rate of 33%; stage III patients with yolk sac elements display poorer prognosis; increased prevalence in late recurrences (chemoresistance)
- Prepubertal type:
- Less aggressive (compared to postpubertal): 80% stage I disease; of these 6% relapse on surveillance only
- Higher tendency to bypass retroperitoneal lymph nodes and spread hematogenously (compared to postpubertal)
Diagnosis
- Physical examination
- Testicular ultrasound
- Serum tumor markers
- Definitive diagnosis: histological examination of orchiectomy specimen
- References: Surg Pathol Clin 2015;8:687, Hum Pathol 2018;82:113
Laboratory
- Elevated alpha fetoprotein in 95 - 98% of the cases (may also be elevated in 20 - 25% of teratomas via hepatoid differentiation, mucinous glands) (Open Access J Urol 2011;4:1)
- Care in interpretation in neonates (physiologically elevated alpha fetoprotein until 6 months)
Radiology description
- Ultrasound: heterogenous echogenicity, frequently with cystic spaces, ill defined margins and calcifications (Radiographics 2017;37:1085)
Prognostic factors
- Classical factors for testicular germ cell tumors in general, including staging and International Germ Cell Cancer Collaborative Group grouping
- In postpubertal mixed tumors, higher percentage of yolk sac tumor is associated with lower stage (Am J Clin Pathol 2016;145:341)
- For stage I prepubertal type tumors: size > 4.5 cm, invasion of rete testis / epididymis and necrosis (Am J Surg Pathol 2015;39:1121)
Case reports
- 7 month old child with bilateral synchronous testicular prepubertal type and teratoma (Urology 2018;117:142)
- 2 year old boy with yolk sac tumor of cryptorchid testis, diagnosed by fine needle aspiration (J Cytol 2015;32:53)
- 30 year old man with metastatic yolk sac tumor descending from an intra-abdominal testis (Oncol Lett 2015;10:3647)
- 46 year old man with postpubertal type, arising from cryptorchid testes (BMJ Case Rep 2019;12:e229541)
- 50 year old man with pure yolk sac tumor of testis (Med J Dr DY Patil Univ 2016;9:499)
Treatment
- Orchiectomy, with or without adjuvant (cisplatin based) chemotherapy
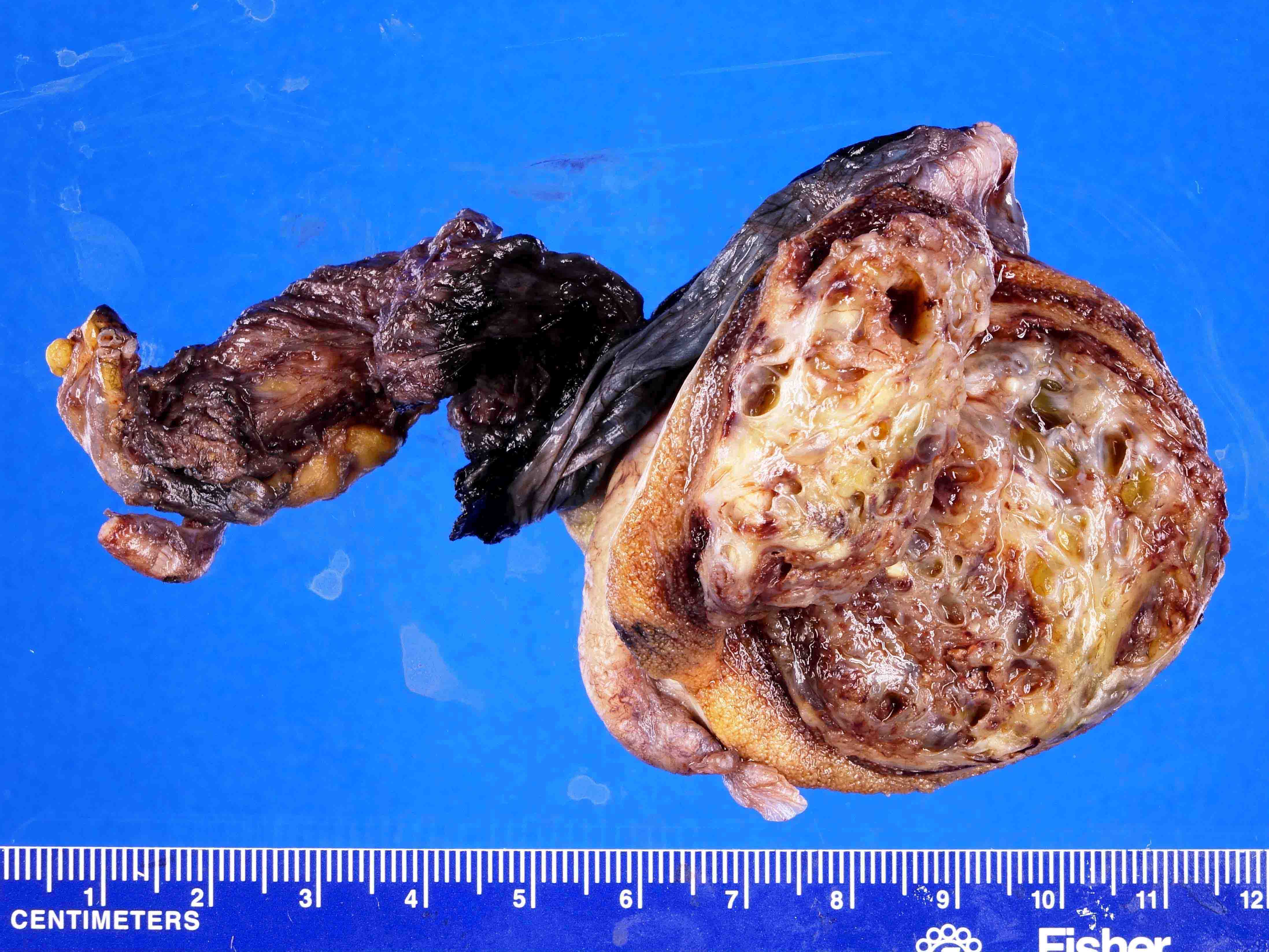
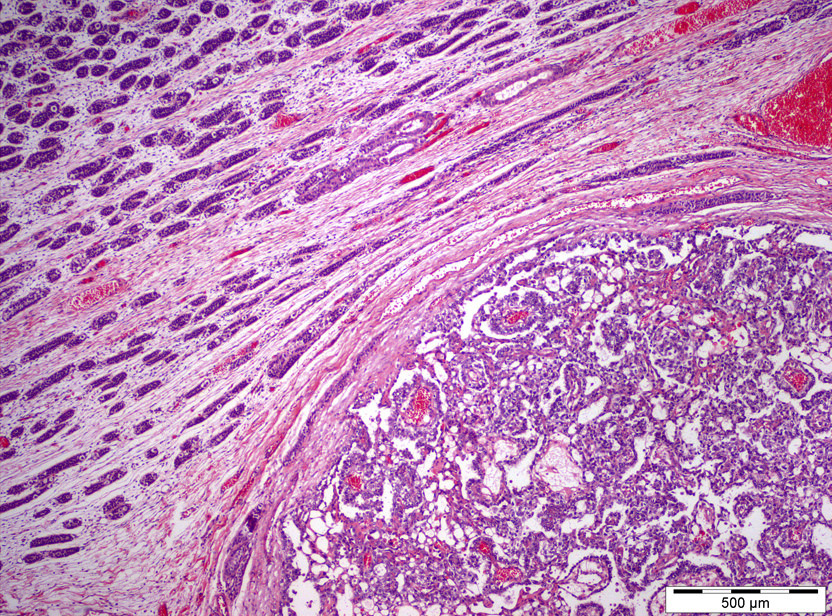
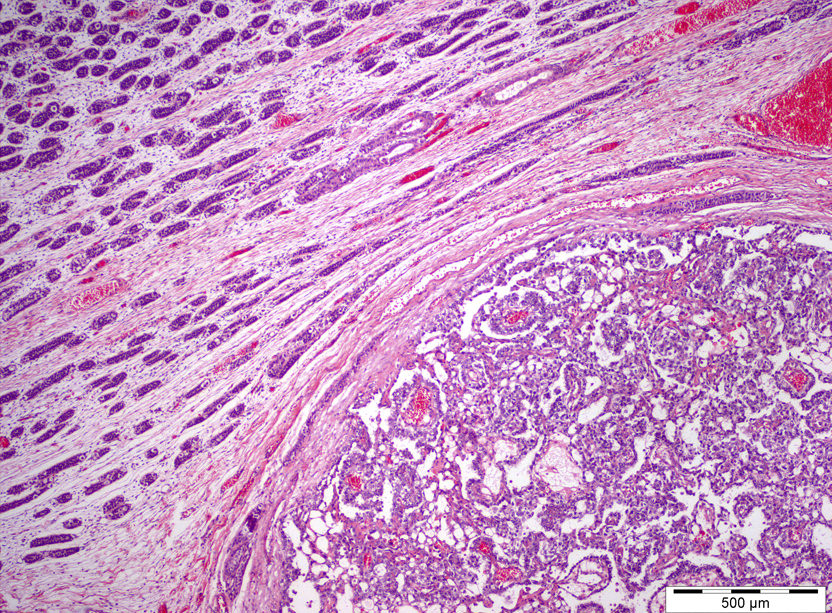
Gross description
- Postpubertal type:
- Poorly circumscribed, nonencapsulated, predominantly solid
- Gray to white to yellow to tan, gelatinous surface
- Hemorrhage, necrosis and cystic areas are common
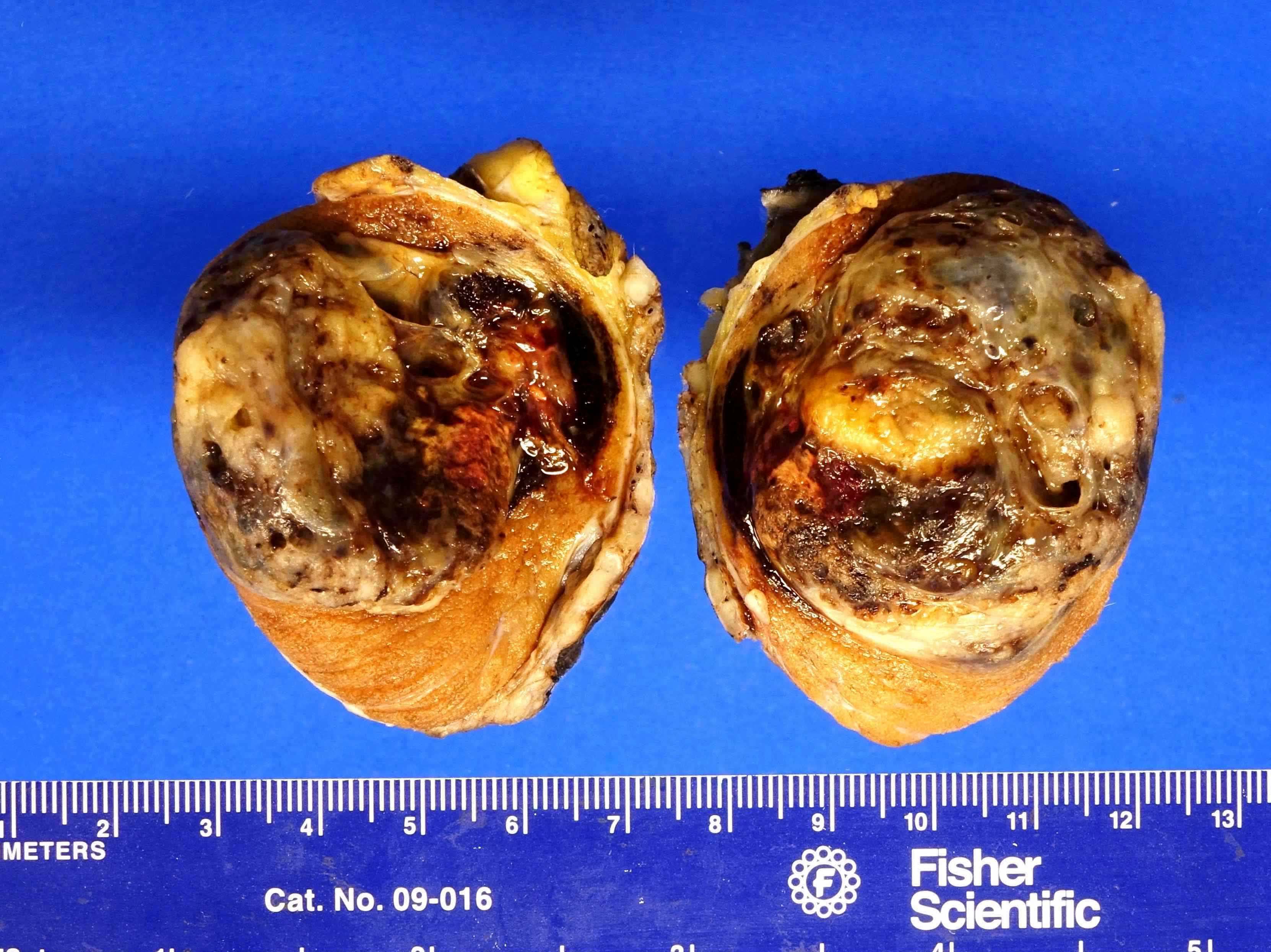
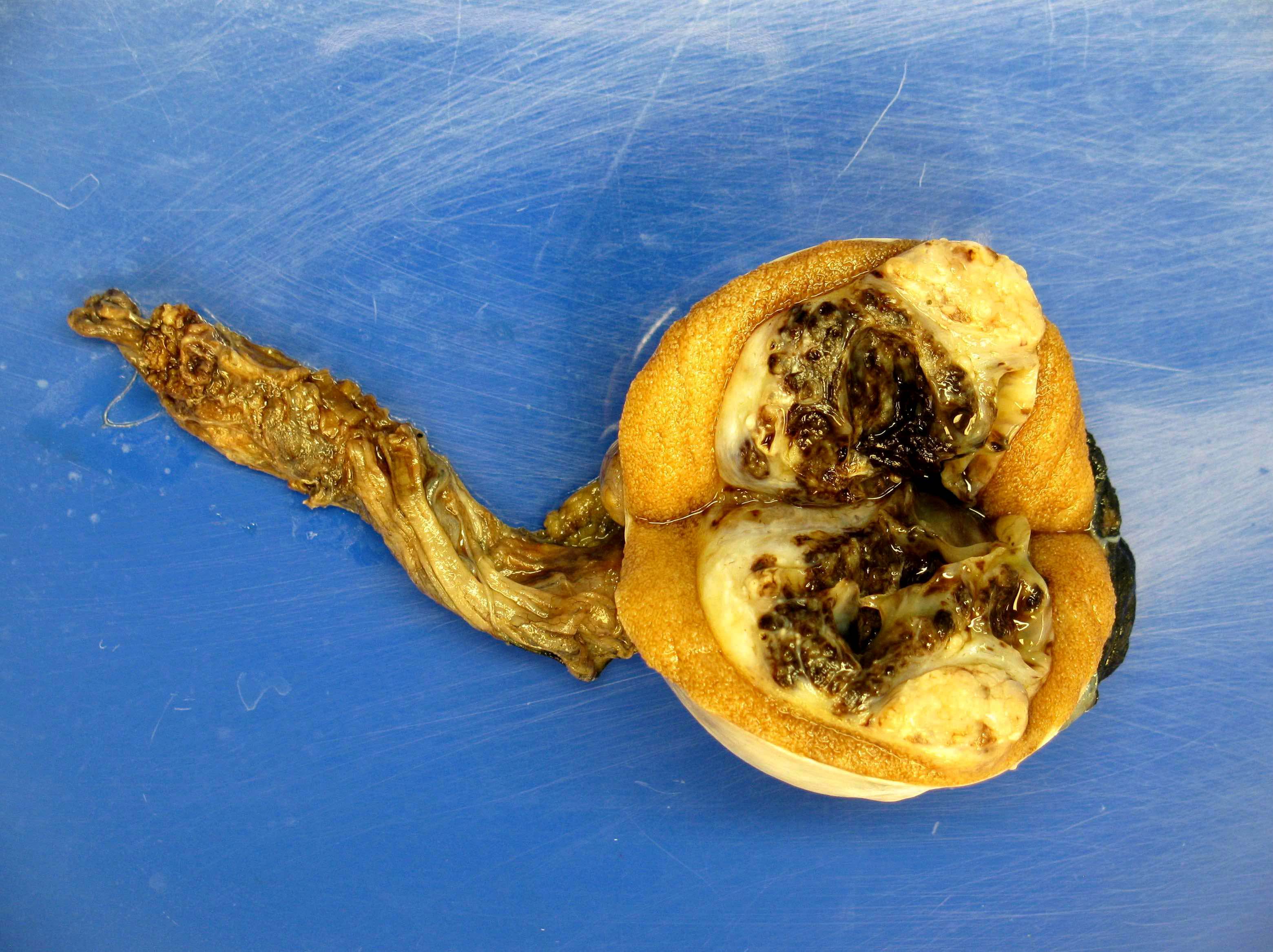
- Prepubertal type:
- More homogeneous, gray to yellow, solid or mucoid, bulging and lobulated
- Hemorrhage and necrosis are uncommon
- References: Surg Pathol Clin 2015;8:687, Hum Pathol 2018;82:113
Gross images
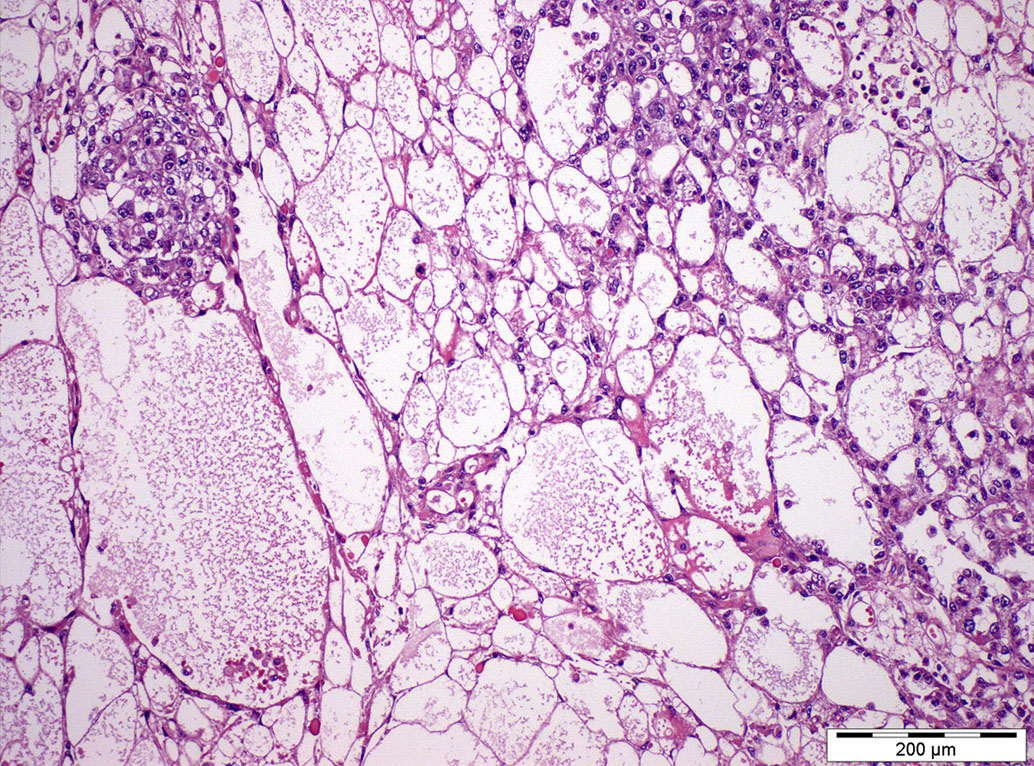
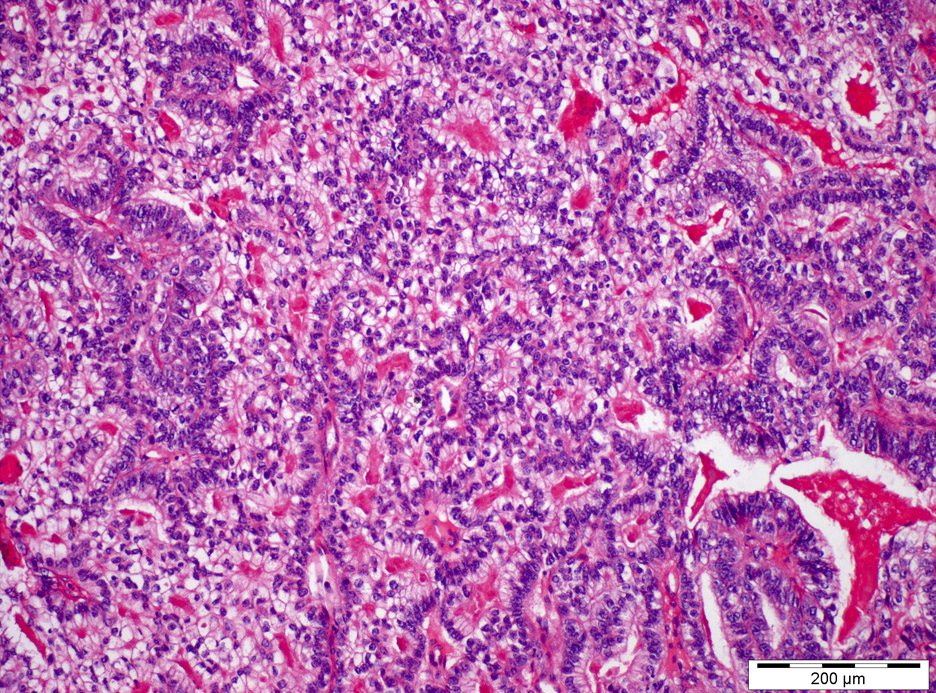
Microscopic (histologic) description
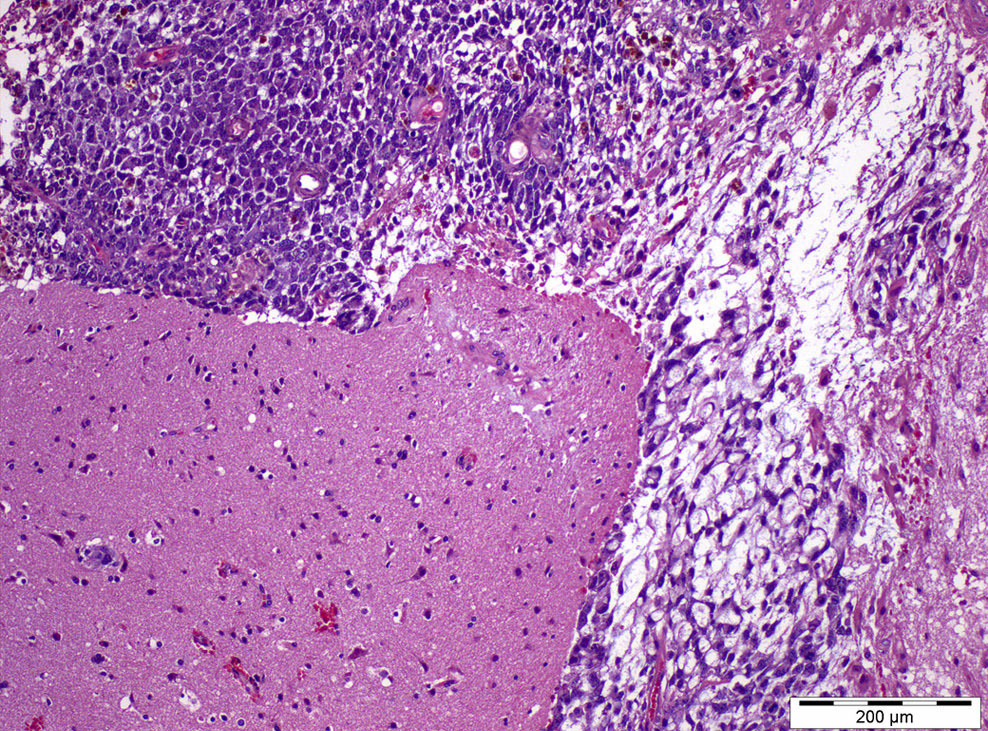
- Most overlooked subtypes of germ cell tumors (dedicated sampling is key) (Pathology 2018;50:88, Hum Pathol 2018;82:113)
- Remarkable heterogeneity, multiple patterns, often combined:
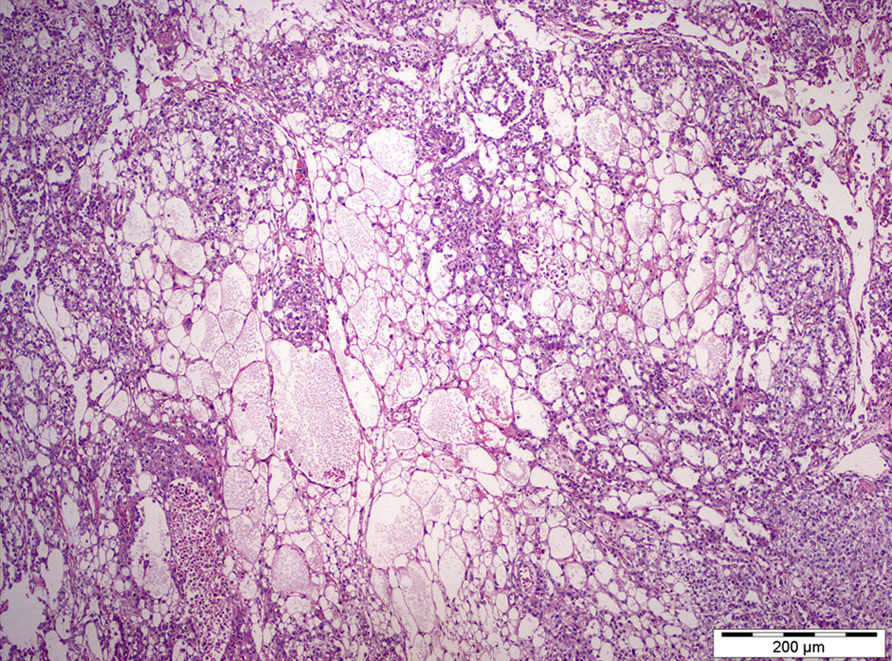
- Microcystic / reticular pattern:
- Most common
- Anastomosing cords of flattened cells forming a honeycomb / spider web meshwork, forming spaces enclosing mucoid / basophilic material
- Macrocystic pattern:
- Coalescence of smaller cystic spaces described in the microcystic pattern
- Myxomatous pattern:
- Resembles the microcystic pattern
- Sparse dispersed spindled / stellate cells in a myxoid background that often contains a prominent capillary network (also called angioblastic pattern)
- Focal differentiation into cartilage or skeletal muscle may be rarely seen and should not elicit diagnosis of teratoma
- Sarcomatoid / spindle cell pattern:
- Progression of the myxomatous pattern (increase in number / density of spindled / stellate cells retaining cytokeratin positivity)
- Some cases regarded as somatic malignancy in the form of sarcoma may actually represent sarcomatoid pattern of yolk sac tumor (Am J Surg Pathol 2014;38:1396)
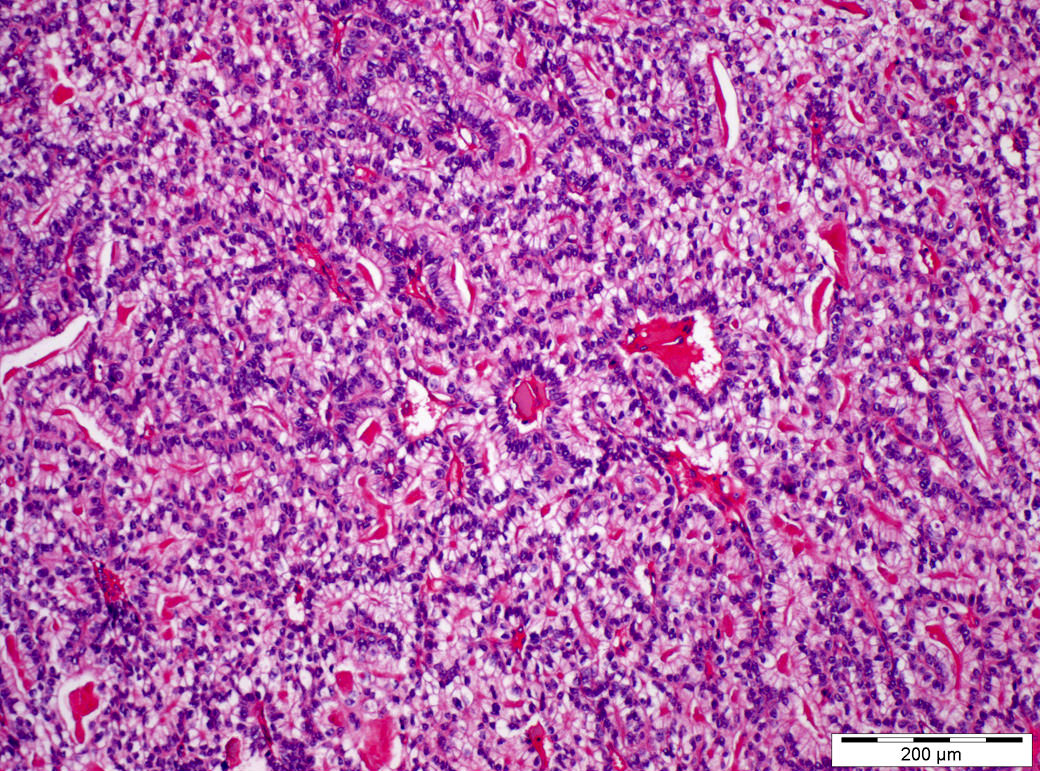
- Solid pattern:
- Sheets of polygonal clear to amphophilic cells form nodules
- Distinct cell membrane (like in seminoma)
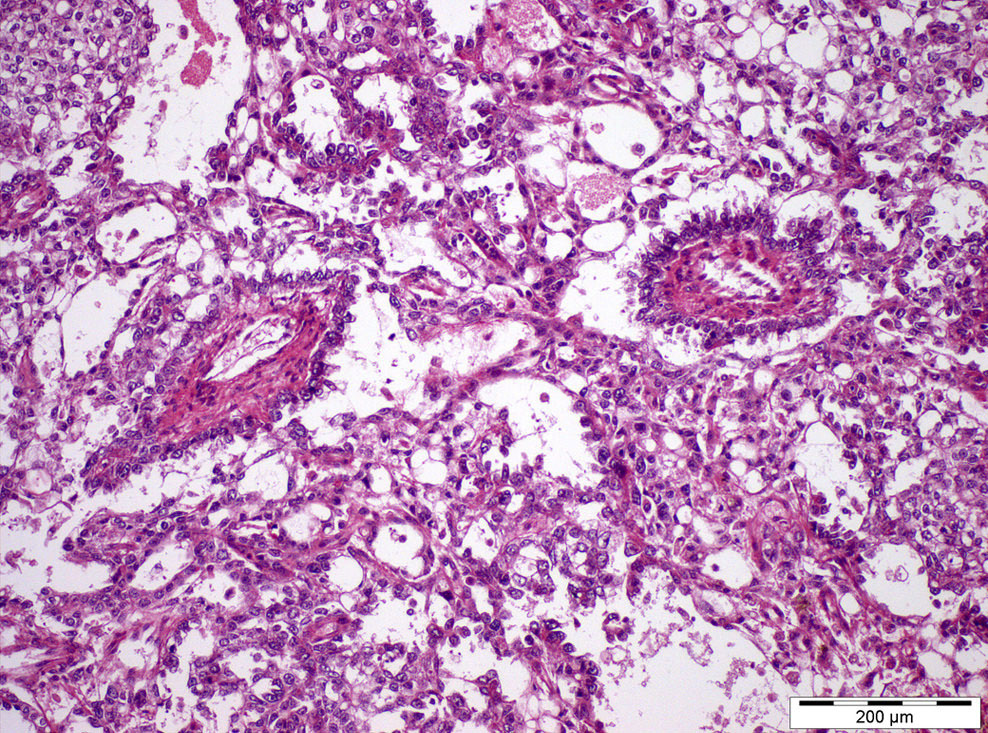
- Glandular / alveolar pattern:
- Glands or tubular structures, sometimes with subnuclear vacuoles resembling secretory endometrium or resembling intestinal epithelium
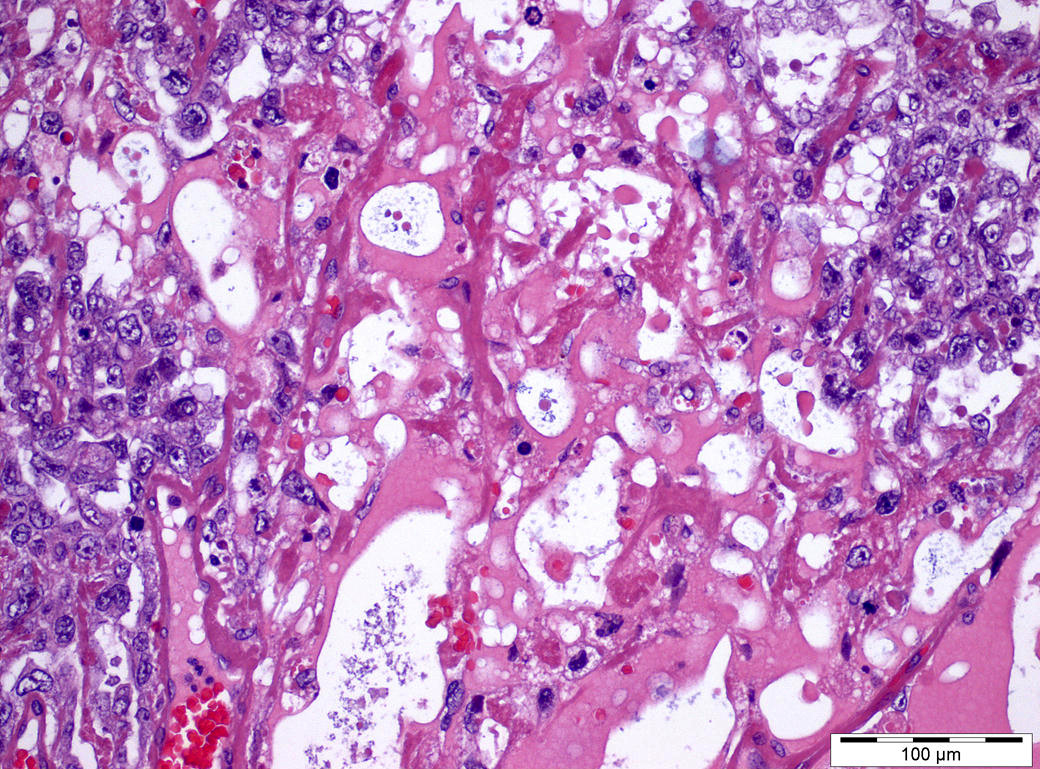
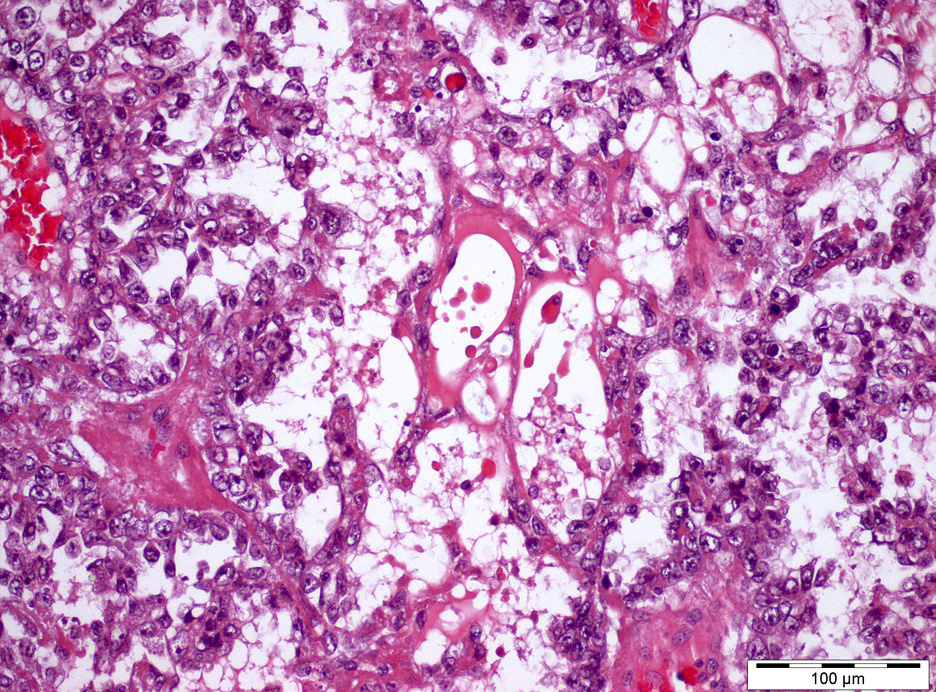
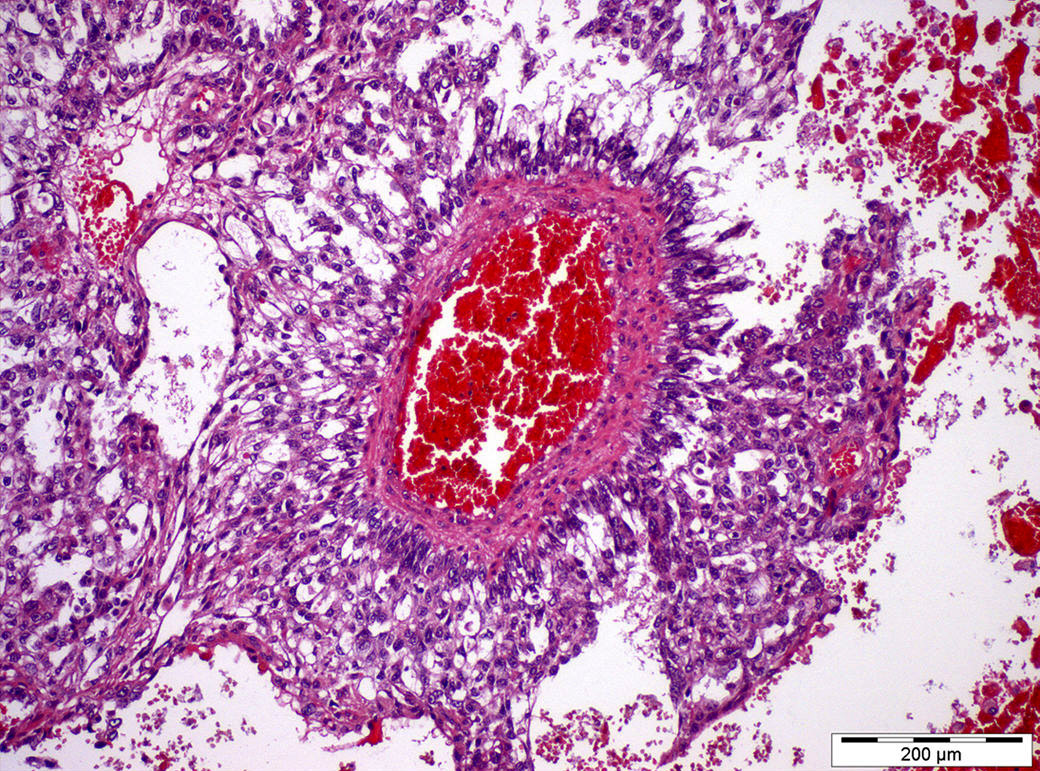
- Endodermal sinus / perivascular pattern:
- Predominance of Schiller-Duval bodies (see below)
- Hepatoid pattern:
- Sheets, solid nests or trabeculae of polygonal eosinophilic cells, with large nuclei and prominent nucleoli, resembling hepatocytes
- Strongly alpha fetoprotein positive
- Papillary pattern:
- Papillary formations with or without fibrovascular cores projecting into cysts
- Often hobnail morphology, high nuclear to cytoplasmic ratio
- Exfoliated cells forming clusters may be seen in the vicinity
- Parietal pattern:
- Abundant, dense, eosinophilic basement membrane material surrounding tumor cells
- Polyvesicular vitelline pattern:
- Always occurs with other patterns
- Vesicles, sometimes with constrictions (forming figure 8 shaped structures), in an edematous to fibrous stroma
- Microcystic / reticular pattern:
- Cytological atypia is variable across patterns; it is most significant in solid, sarcomatoid and glandular patterns
- 50% of tumors contain Schiller-Duval bodies (papillary structures within cystic spaces, lined by cuboidal to columnar cells with a distinct central vessel)
- Characteristic (but not pathognomonic) hyaline globules (within and outside cytoplasm), refractile and eosinophilic, which are alpha-1 antitrypsin+ and PAS diastase+
Microscopic (histologic) images
Cytology description
- Variably sized cell clusters, occasional glomeruloid structures, metachromatic basement membrane material, large cells with high nuclear to cytoplasmic ratio, mucoid background, PAS positive hyaline globules (J Cytol 2015;32:53)
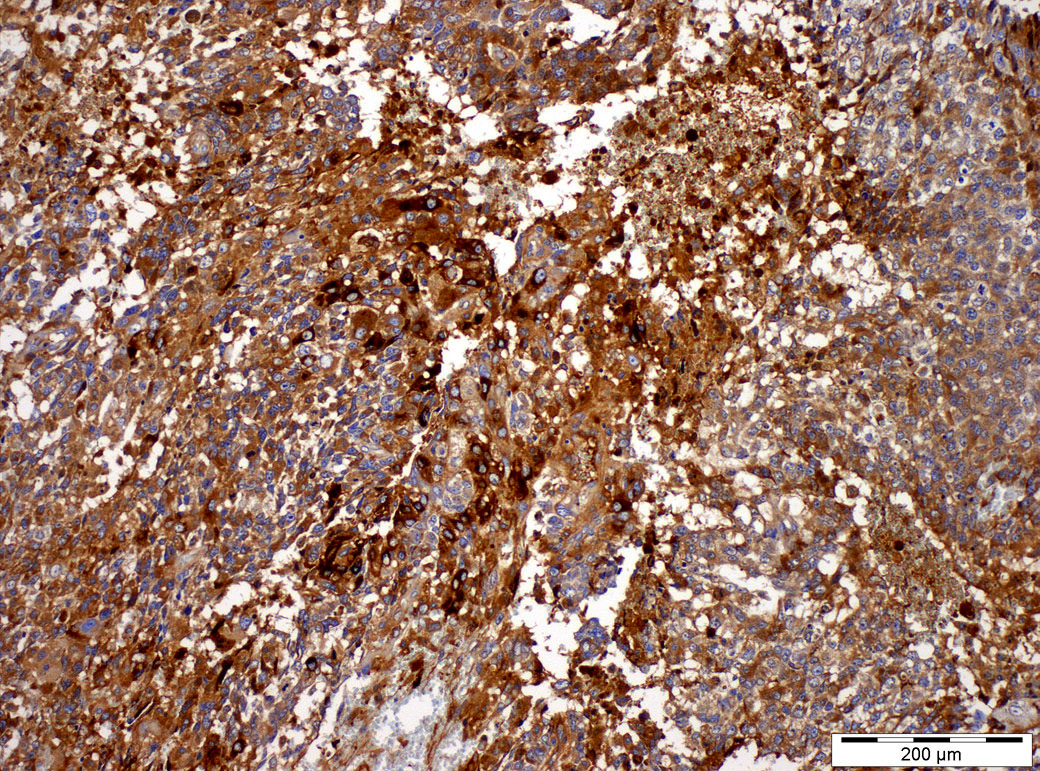
Positive stains
- Alpha fetoprotein (more specific but less sensitive: variable, often focal, in many cases entirely absent) (Histopathology 2014;65:51)
- Glypican 3 (more sensitive, staining almost all cases but less specific) (Am J Surg Pathol 2006;30:1570, Histopathology 2011;58:312)
- SALL4 (sensitive and specific for most germ cell tumor types including yolk sac tumor) (Am J Surg Pathol 2009;33:1065)
- Pancytokeratin (uniformly positive)
- Villin (usually positive)
- CK7 (84%) (Hum Pathol 2019;84:254)
Negative stains
- OCT 3/4
- CD30 (weak / focal staining can be seen)
- CD117 (usually negative but variable; 60% in solid patterns) (Am J Surg Pathol 2012;36:360)
- PLAP and podoplanin (D2-40) (rarely positive)
- Inhibin (Hum Pathol 2019;84:254)
- p63 (Hum Pathol 2019;84:254)
- GATA3 (12%, may be expressed in primitive patterns) (Hum Pathol 2016;48:18, Histopathology 2016;68:613)
Electron microscopy description
- Epithelial cells with tight junctional complexes, apical microvilli, extracellular deposits of basal lamina, glycogen
Molecular / cytogenetics description
- Postpubertal type:
- Gain of 12p (usually isochromosome 12p)
- Erased genomic imprinting
- Prepubertal type:
- Multiple gains (1q, 20q, 22) and losses (1p, 4, 6q) but no 12p gain
- Biparental genomic imprinting
- References: Int J Mol Sci 2019;20:258, Nat Rev Cancer 2019;19:522
Sample pathology report
- Right testicle, right radical orchiectomy:
- Mixed germ cell tumor, postpubertal type, 40% teratoma, 30% embryonal carcinoma, 15% yolk sac tumor and 15% seminoma (see synoptic report)
Differential diagnosis
- Teratoma (versus glandular pattern of yolk sac tumor):
- Encircling smooth muscle layer, no evidence of glandular branching
- Seminoma (versus solid pattern of yolk sac tumor):
- Presence of fibrovascular septa filled with prominent lymphocytic infiltrate
- OCT 3/4+, CD117 diffusely +; AFP-, glypican 3-, AE1 / AE3- to weak / focal
- Embryonal carcinoma (some patterns, such as papillary or glandular):
- Higher cytological atypia, extensive necrosis, brisk mitotic activity
- OCT 3/4+, CD30 diffusely +; AFP-, glypican 3- (Pathology 2018;50:88)
- Rete testis hyperplasia with hyaline globules:
- Lacks atypia and mitoses
- SALL4-, AFP-, glypican 3- (Am J Surg Pathol 1991;15:66)
- Microcystic Leydig cell tumor:
- No recurrence or metastasis
- Calretinin+, inhibin+, MelanA+; AFP-, glypican 3-; cytokeratin and CAM 5.2 focal (Am J Surg Pathol 1999;23:546)
Practice question #1
A 2 year old boy is submitted to orchiectomy. In the figure you may appreciate a histological section of the lesion. Which sentence is true?
- Most likely the tumor has gain of 12p
- Most likely this tumor is present in its pure form and not mixed with other subtypes
- Negative AFP excludes a yolk sac tumor
- OCT 3/4 staining will be positive in this tumor
- Schiller-Duval bodies are present and are definitive proof of a postpubertal type yolk sac tumor
Practice answer #1
B. The picture represents a prepubertal type yolk sac tumor. This entity does not show 12p gain (contrarily to postpubertal type tumors of the testis). Schiller-Duval bodies are present but may be seen (or not) in both prepubertal type and postpubertal type yolk sac tumors. AFP is variably positive (but many times negative) in yolk sac tumors and OCT 3/4 is invariably negative. Prepubertal type yolk sac tumor is almost always seen in its pure form (contrarily to postpubertal type tumors, which are frequently mixed).
Comment Here
Reference: Yolk sac tumor
Comment Here
Reference: Yolk sac tumor
Practice question #2
Which of the following is true regarding yolk sac tumor of the testis?
- Differential diagnosis with seminoma is particularly problematic in the microcystic pattern
- Glypican 3 is the most specific marker of this tumor subtype
- Can be germ cell neoplasia related or unrelated depending on pubertal status
- Only occurs in adolescents
- Serum AFP levels are not relevant for the diagnosis
Practice answer #2
C. Yolk sac tumor of the testis can be germ cell neoplasia in situ related (postpubertal type) or germ cell neoplasia in situ unrelated (prepubertal type), showing a bimodal epidemiology (young adults and young children, respectively). Glypican 3 is sensitive but not a specific marker of this tumor type. Differential diagnosis with seminoma occurs when a solid pattern is extensive. There is a strong correlation between the finding of elevated serum AFP and presence of yolk sac tumor elements.
Comment Here
Reference: Yolk sac tumor
Comment Here
Reference: Yolk sac tumor